How does the blue bottle experiment work What

How does the blue bottle experiment work?

What is the blue bottle experiment?

Hypothesis �Transparent Shake Blue Transparent �Cause: Dioxygen

Calculations �m=n*M �Mglucose = 180, 096 g/mol �Glucose mass = 180, 096*8, 0*10 -3 = 1, 44 g �V= n/C �Csodium hydroxide = 0, 50 mol/L �Sodium hydroxide volume = 2, 5*10 -2 / 0, 50 = 0, 05 L or 50 m. L

Protocol �Pour 50 m. L of sodium hydroxide solution (0. 5 mol/L) into an erlenmeyer flask. �Add 1. 44 g of Glucose into the Erlenmeyer flask. �Shake the erlenmeyer flask �Add three drops of methylene blue to the Erlenmeyer flask. �Place the cap on the flask. �Shake the flask and let it stand for about one minute until the color disappears. �Repeat last step until no more color appears it the flask. �Open the flask to add new air, this should make the experiment repeatable again.

Identification of the gas �mgas = 0, 00266 g �ngas = 8, 31 x 10 -31 mol �M= m/n M= 32 g/mol �Mdinitrogen= 28, o g/mol �Mdioxygen= 32, o g/mol �Mcarbon dioxyde= 44, 0 g/mol �So the gas which causes the coloration is the dioxygen

Results Before After The dioxygen reacts with the incolor methylene blue to create blue methylene blue, wich makes become blue the solution
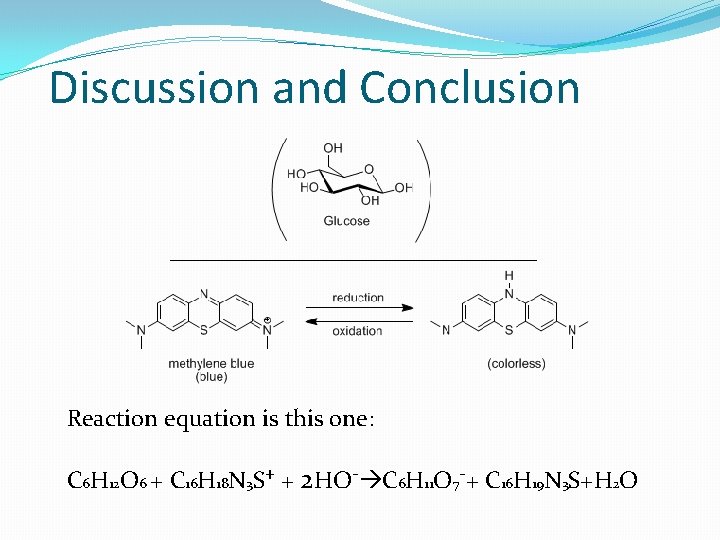
Discussion and Conclusion Reaction equation is this one: C 6 H 12 O 6 + C 16 H 18 N 3 S+ + 2 HO- C 6 H 11 O 7 -+ C 16 H 19 N 3 S+H 2 O
- Slides: 8