Unit 5 Chemical Kinetics Day 5 Begin Kinetics

Unit 5: Chemical Kinetics Day 5: Begin Kinetics Lab

Warm Up If you are given 15 M stock solution and you need to make ONLY 20 m. L of a 6 M solution, how much of the stock solution should you use?

Agenda Demonstration 2. Begin Lab Set Up 1. Purpose Hypothesis Materials Pre-Lab Questions 4. If Time: Begin Introductory Activity 3.
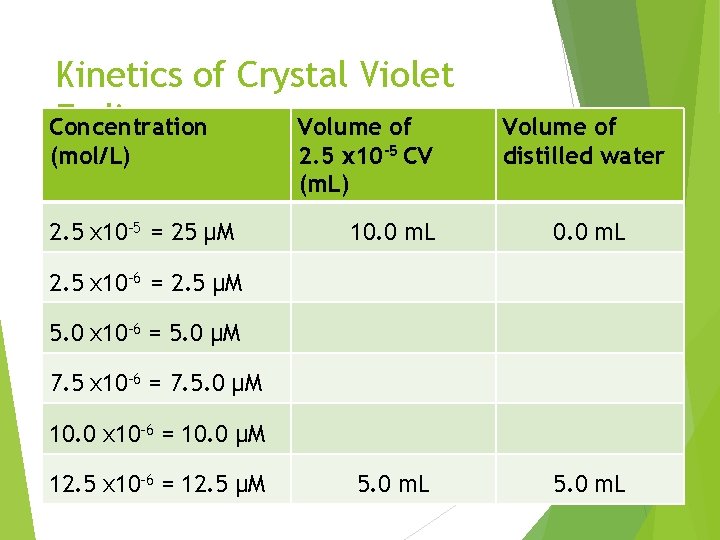
Kinetics of Crystal Violet Pre-Lab Fading Concentration Volume of 1. (mol/L) 2. 5 x 10 -5 = 25 μM 2. 5 x 10 -5 CV (m. L) Volume of distilled water 10. 0 m. L 5. 0 m. L 2. 5 x 10 -6 = 2. 5 μM 5. 0 x 10 -6 = 5. 0 μM 7. 5 x 10 -6 = 7. 5. 0 μM 10. 0 x 10 -6 = 10. 0 μM 12. 5 x 10 -6 = 12. 5 μM

Kinetics of Crystal Violet Fading Guided Inquiry Design and Procedure Part A CV+ + OH- CVOH What percent of OH- ions will remain at the end of a reaction if the initial concentrations of each reactant were equal (1: 1 ratio)?
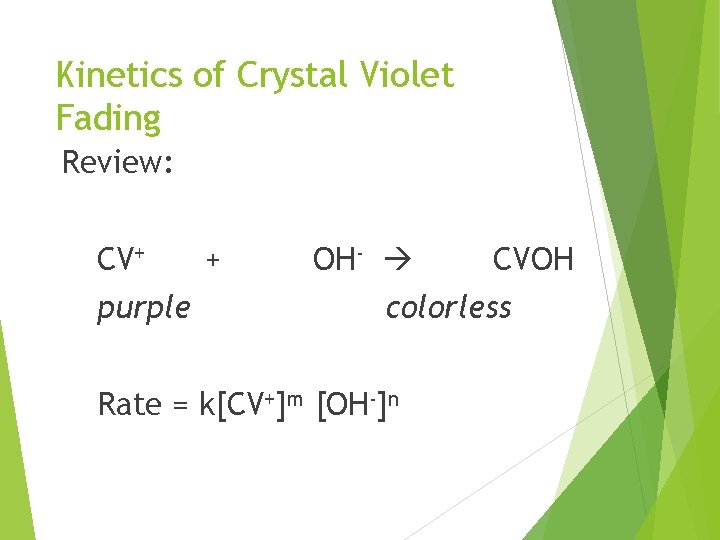
Kinetics of Crystal Violet Fading Review: CV+ purple + OH- CVOH colorless Rate = k[CV+]m [OH-]n
![Kinetics of Crystal Violet Fading Rate Law: Rate = k’ [CV+]m where k’ = Kinetics of Crystal Violet Fading Rate Law: Rate = k’ [CV+]m where k’ =](http://slidetodoc.com/presentation_image/f29355676e3a7d1af80a655a86d7bec2/image-7.jpg)
Kinetics of Crystal Violet Fading Rate Law: Rate = k’ [CV+]m where k’ = k [OH-]n The constant k’ is a new “pseudo” rate constant. The new pseudo rate law is valid when the concentration of OH- ions is much greater than the concentration of CV+ ions. Under these conditions, the [OH-] term in the original rate law will not change much over the course of the reaction and may be treated as a constant in the rate equation.
Kinetics of Crystal Violet Fading
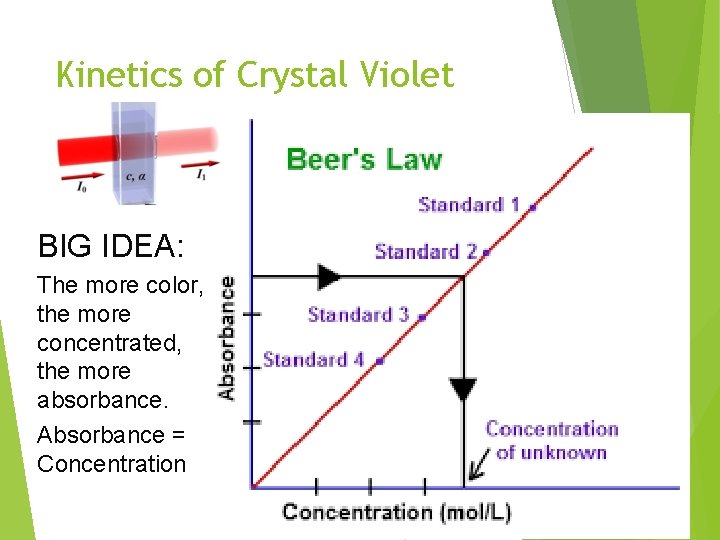
Kinetics of Crystal Violet Fading BIG IDEA: The more color, the more concentrated, the more absorbance. Absorbance = Concentration

Part B: Procedure Outline 1. 2. 3. 4. 5. Na. OH conc. needs to be changed, not CV+, so that the effect on k’ can be calculated and the order of reaction with respect to OH- determined Colorimeter must be zeroed with a blank, equal volumes 0. 01 M Na. OH and water To make 10 m. L of 0. 01 M Na. OH, measure 5. 0 m. L of 0. 02 M Na. OH in a serological pipet and… Clean the pipet with small portions of crystal violet solution. Measure 10 m. L of 25 u. M crystal violet solution… After the absorbance vs. time data are recorded, the absorbance values need to be converted into concentration, then separate data columns for ln[CV+] and 1/[CV+] need to be created analyzed to determine k’ for the new concentration of Na. OH, 0. 01 M.
- Slides: 10