Clicker Questions Chapter 18 Electrochemistry Allison Soult University























- Slides: 31

Clicker Questions Chapter 18 Electrochemistry Allison Soult University of Kentucky © 2014 Pearson Education, Inc.

What is the coefficient of Fe 2+ when the following redox equation is balanced? Fe 2+ (aq) + Mn. O 4– (aq) → Fe 3+ (aq) + Mn 2+ (aq) a) b) c) d) e) 1 2 3 4 5 © 2014 Pearson Education, Inc.

What is the coefficient of Fe 2+ when the following redox equation is balanced? Fe 2+ (aq) + Mn. O 4– (aq) → Fe 3+ (aq) + Mn 2+ (aq) a) b) c) d) e) 1 2 3 4 5 © 2014 Pearson Education, Inc.

The following reaction occurs in a lead storage battery. How many moles of electrons are transferred in the following balanced redox equation per reaction cycle? Pb (s) + Pb. O 2 (aq) + 2 HSO 4– (aq) + 2 H+ (aq) → 2 Pb. SO 4 (s) + 2 H 2 O (l) a) b) c) d) 2. 34 1. 35 1. 30 2. 41 © 2014 Pearson Education, Inc.

The following reaction occurs in a lead storage battery. How many moles of electrons are transferred in the following balanced redox equation per reaction cycle? Pb (s) + Pb. O 2 (aq) + 2 HSO 4– (aq) + 2 H+ (aq) → 2 Pb. SO 4 (s) + 2 H 2 O (l) a) b) c) d) 2. 34 1. 35 1. 30 2. 41 © 2014 Pearson Education, Inc.

How many water molecules will there be when Mn. O 4– + CN– Mn. O 2 + CNO– is balanced in base? a) b) c) d) 4 on the reactant side 4 on the product side 2 on the reactant side 2 on the product side © 2014 Pearson Education, Inc.

How many water molecules will there be when Mn. O 4– + CN– Mn. O 2 + CNO– is balanced in base? a) b) c) d) 4 on the reactant side 4 on the product side 2 on the reactant side 2 on the product side © 2014 Pearson Education, Inc.

Which transformation could take place at the anode of an electrochemical cell? a) b) c) d) NO → NO 3– VO 2+ → VO 2+ O 2 → H 2 O 2 All of the above would function as cathodes. e) All of the above would function as anodes. © 2014 Pearson Education, Inc.

Which transformation could take place at the anode of an electrochemical cell? a) b) c) d) NO → NO 3– VO 2+ → VO 2+ O 2 → H 2 O 2 All of the above would function as cathodes. e) All of the above would function as anodes. © 2014 Pearson Education, Inc.

The purpose of the salt bridge in an electrochemical cell is a) to maintain electrical neutrality in the half-cells via migration of ions. b) to provide a source of ions to react at the anode and cathode. c) to provide oxygen to facilitate oxidation at the anode. d) to provide a means for electrons to travel from the anode to the cathode. e) to provide a means for electrons to travel from the cathode to the anode. © 2014 Pearson Education, Inc.

The purpose of the salt bridge in an electrochemical cell is a) to maintain electrical neutrality in the half-cells via migration of ions. b) to provide a source of ions to react at the anode and cathode. c) to provide oxygen to facilitate oxidation at the anode. d) to provide a means for electrons to travel from the anode to the cathode. e) to provide a means for electrons to travel from the cathode to the anode. © 2014 Pearson Education, Inc.

What is the reducing agent in the following electrochemical cell? Ni (s) | Ni 2+ (aq) || Ag+ (aq) | Ag (s) a) b) c) d) e) Ni Ni 2+ Ag Ag+ There is no reducing agent in an electrochemical cell. © 2014 Pearson Education, Inc.

What is the reducing agent in the following electrochemical cell? Ni (s) | Ni 2+ (aq) || Ag+ (aq) | Ag (s) a) b) c) d) e) Ni Ni 2+ Ag Ag+ There is no reducing agent in an electrochemical cell. © 2014 Pearson Education, Inc.

Which of the following is the best reducing agent? a) b) c) d) Ni 2+ Ni Zn Ag © 2014 Pearson Education, Inc. Ni 2+ (aq) + 2 e– → Ni (s) – 0. 23 V Zn 2+ (aq) + 2 e– → Zn (s) – 0. 76 V Ag+ (aq) + e– → Ag (s) 0. 80 V

Which of the following is the best reducing agent? a) b) c) d) Ni 2+ Ni Zn Ag © 2014 Pearson Education, Inc. Ni 2+ (aq) + 2 e– → Ni (s) – 0. 23 V Zn 2+ (aq) + 2 e– → Zn (s) – 0. 76 V Ag+ (aq) + e– → Ag (s) 0. 80 V

Calculate the cell potential for the following reaction under standard conditions: Mg (s) + Cu 2+ (aq) → Cu (s) + Mg 2+ (aq) a) b) c) d) e) +2. 04 – 2. 04 +2. 71 – 1. 36 +1. 36 © 2014 Pearson Education, Inc. Cu 2+ (aq) + 2 e– → Cu (s) 0. 34 V Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 V

Calculate the cell potential for the following reaction under standard conditions: Mg (s) + Cu 2+ (aq) → Cu (s) + Mg 2+ (aq) a) b) c) d) e) +2. 04 – 2. 04 +2. 71 – 1. 36 +1. 36 © 2014 Pearson Education, Inc. Cu 2+ (aq) + 2 e– → Cu (s) 0. 34 V Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 V

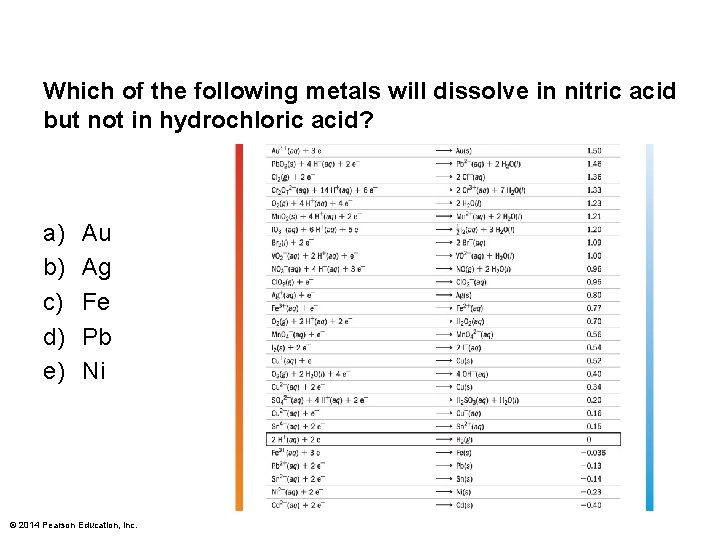
Which of the following metals will dissolve in nitric acid but not in hydrochloric acid? a) b) c) d) e) Au Ag Fe Pb Ni © 2014 Pearson Education, Inc.

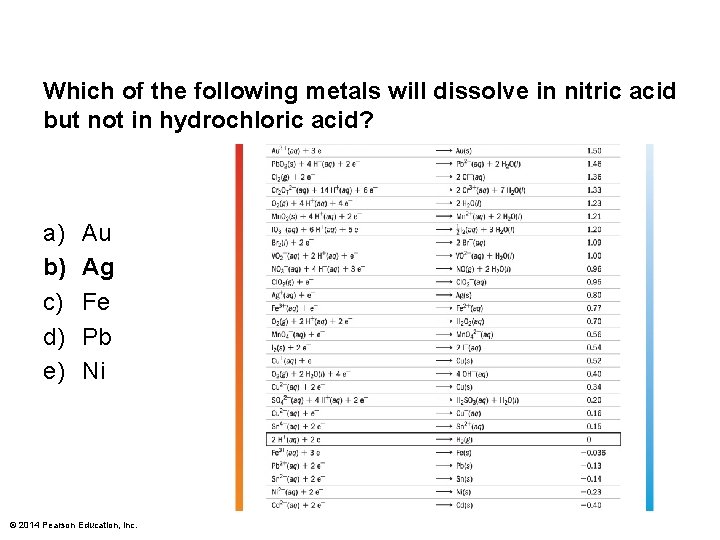
Which of the following metals will dissolve in nitric acid but not in hydrochloric acid? a) b) c) d) e) Au Ag Fe Pb Ni © 2014 Pearson Education, Inc.

Consider a cell that employs the following overall reaction: 2 Al (s) + 3 I 2 (s) → 2 Al 3+ (aq) + 6 I– (aq) What is ΔG° for the cell? a) b) c) d) e) I 2 (s) + 2 e– → 2 I– (aq) 0. 54 Al 3+ (aq) + 3 e– → Al (s) – 1. 66 636 k. J/mol – 648 k. J/mol – 216 k. J/mol – 636 k. J/mol – 1270 k. J/mol © 2014 Pearson Education, Inc.

Consider a cell that employs the following overall reaction: 2 Al (s) + 3 I 2 (s) → 2 Al 3+ (aq) + 6 I– (aq) What is ΔG° for the cell? a) b) c) d) e) I 2 (s) + 2 e– → 2 I– (aq) 0. 54 Al 3+ (aq) + 3 e– → Al (s) – 1. 66 636 k. J/mol – 648 k. J/mol – 216 k. J/mol – 636 k. J/mol – 1270 k. J/mol © 2014 Pearson Education, Inc.

What is the value of the equilibrium constant when the cell potential is found to be − 0. 29 V for a transfer of 2 moles of electrons? a) b) c) d) e) 1. 6 × 10– 10 0. 56 0. 72 0. 87 6. 4 × 10– 2 © 2014 Pearson Education, Inc.

What is the value of the equilibrium constant when the cell potential is found to be − 0. 29 V for a transfer of 2 moles of electrons? a) b) c) d) e) 1. 6 × 10– 10 0. 56 0. 72 0. 87 6. 4 × 10– 2 © 2014 Pearson Education, Inc.

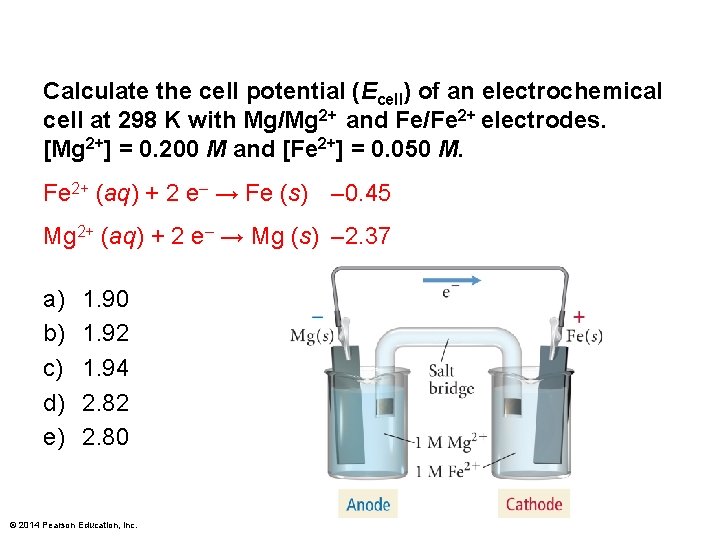
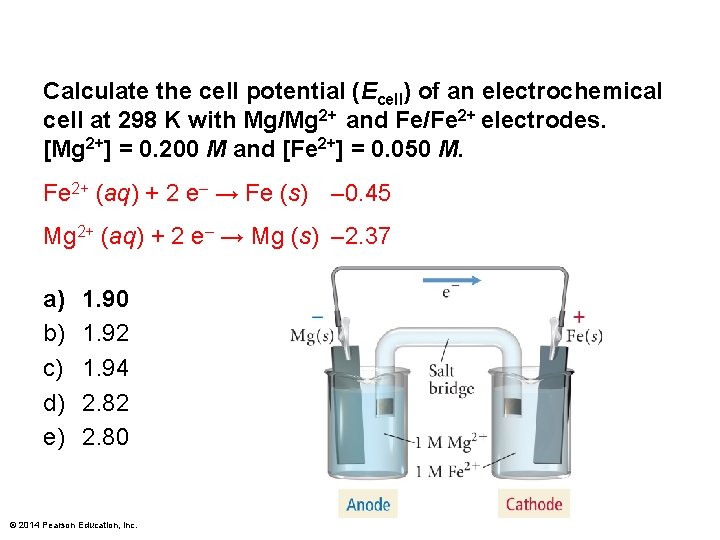
Calculate the cell potential (Ecell) of an electrochemical cell at 298 K with Mg/Mg 2+ and Fe/Fe 2+ electrodes. [Mg 2+] = 0. 200 M and [Fe 2+] = 0. 050 M. Fe 2+ (aq) + 2 e– → Fe (s) – 0. 45 Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 a) b) c) d) e) 1. 90 1. 92 1. 94 2. 82 2. 80 © 2014 Pearson Education, Inc.

Calculate the cell potential (Ecell) of an electrochemical cell at 298 K with Mg/Mg 2+ and Fe/Fe 2+ electrodes. [Mg 2+] = 0. 200 M and [Fe 2+] = 0. 050 M. Fe 2+ (aq) + 2 e– → Fe (s) – 0. 45 Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 a) b) c) d) e) 1. 90 1. 92 1. 94 2. 82 2. 80 © 2014 Pearson Education, Inc.

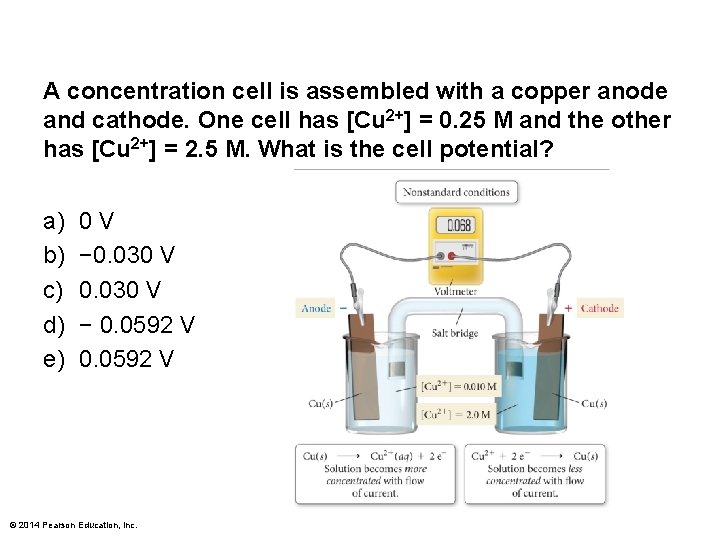
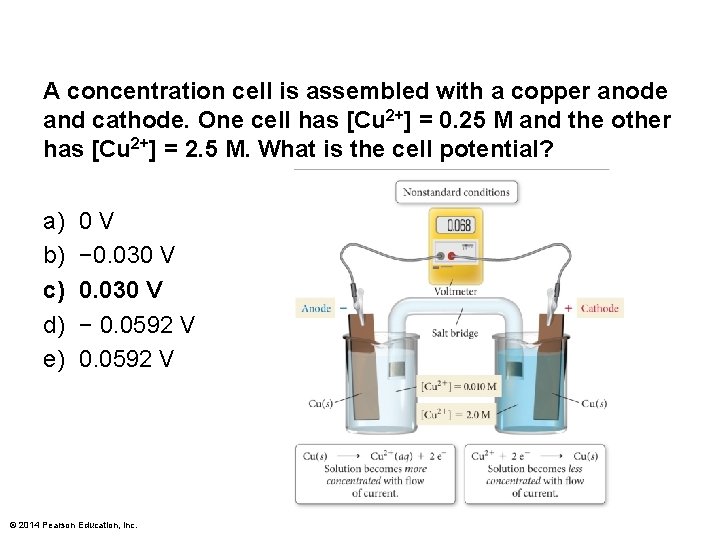
A concentration cell is assembled with a copper anode and cathode. One cell has [Cu 2+] = 0. 25 M and the other has [Cu 2+] = 2. 5 M. What is the cell potential? a) b) c) d) e) 0 V − 0. 030 V − 0. 0592 V © 2014 Pearson Education, Inc.

A concentration cell is assembled with a copper anode and cathode. One cell has [Cu 2+] = 0. 25 M and the other has [Cu 2+] = 2. 5 M. What is the cell potential? a) b) c) d) e) 0 V − 0. 030 V − 0. 0592 V © 2014 Pearson Education, Inc.

What mass of nickel can be plated from a solution containing Ni 2+ with a current of 3. 6 A for 14 minutes? a) b) c) d) e) 0. 92 g 1. 8 g 3. 7 g 1. 0 g 0. 071 g © 2014 Pearson Education, Inc.

What mass of nickel can be plated from a solution containing Ni 2+ with a current of 3. 6 A for 14 minutes? a) b) c) d) e) 0. 92 g 1. 8 g 3. 7 g 1. 0 g 0. 071 g © 2014 Pearson Education, Inc.

Which of the following is the best sacrificial electrode for iron? a) b) c) d) e) Au Cu Ni Pb Mg Au 3+ (aq) + 3 e– → Au (s) 1. 50 V Cu 2+ (aq) + 2 e– → Cu (s) 0. 34 V Fe 3+ (aq) + 3 e– → Fe (s) – 0. 036 V Pb 2+ (aq) + 2 e– → Pb (s) – 0. 13 V Ni 2+ (aq) + 2 e– → Ni (s) – 0. 23 V Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 V © 2014 Pearson Education, Inc.

Which of the following is the best sacrificial electrode for iron? a) b) c) d) e) Au Cu Ni Pb Mg Au 3+ (aq) + 3 e– → Au (s) 1. 50 V Cu 2+ (aq) + 2 e– → Cu (s) 0. 34 V Fe 3+ (aq) + 3 e– → Fe (s) – 0. 036 V Pb 2+ (aq) + 2 e– → Pb (s) – 0. 13 V Ni 2+ (aq) + 2 e– → Ni (s) – 0. 23 V Mg 2+ (aq) + 2 e– → Mg (s) – 2. 37 V © 2014 Pearson Education, Inc.