Clicker Questions Chapter 14 Chemical Equilibrium Allison Soult

Clicker Questions Chapter 14 Chemical Equilibrium Allison Soult University of Kentucky © 2014 Pearson Education, Inc.

Which of the following best describes a reaction at equilibrium? a) Reactants are being consumed and products are being formed. b) The rate of the reaction is zero. c) The rate of the forward and reverse reactions are equal. d) Only products are present. e) The rate of the forward reaction is greater than the rate of the reverse reaction. © 2014 Pearson Education, Inc.

Which of the following best describes a reaction at equilibrium? a) Reactants are being consumed and products are being formed. b) The rate of the reaction is zero. c) The rate of the forward and reverse reactions are equal. d) Only products are present. e) The rate of the forward reaction is greater than the rate of the reverse reaction. © 2014 Pearson Education, Inc.

What is the equilibrium expression for the given reaction? 2 A(aq) + 3 B(aq) ⇌ C(aq) + 2 D(aq) a) b) c) d) © 2014 Pearson Education, Inc.

What is the equilibrium expression for the given reaction? 2 A(aq) + 3 B(aq) ⇌ C(aq) + 2 D(aq) a) b) c) d) © 2014 Pearson Education, Inc.
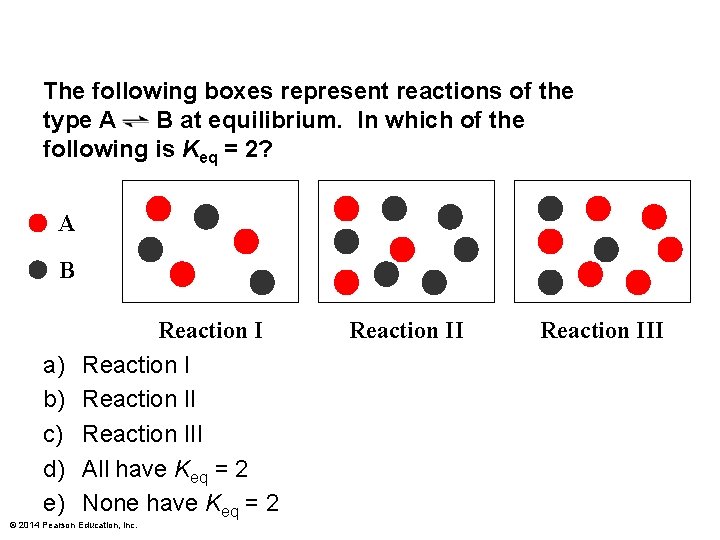
The following boxes represent reactions of the type A B at equilibrium. In which of the following is Keq = 2? A B a) b) c) d) e) Reaction III All have Keq = 2 None have Keq = 2 © 2014 Pearson Education, Inc. Reaction III
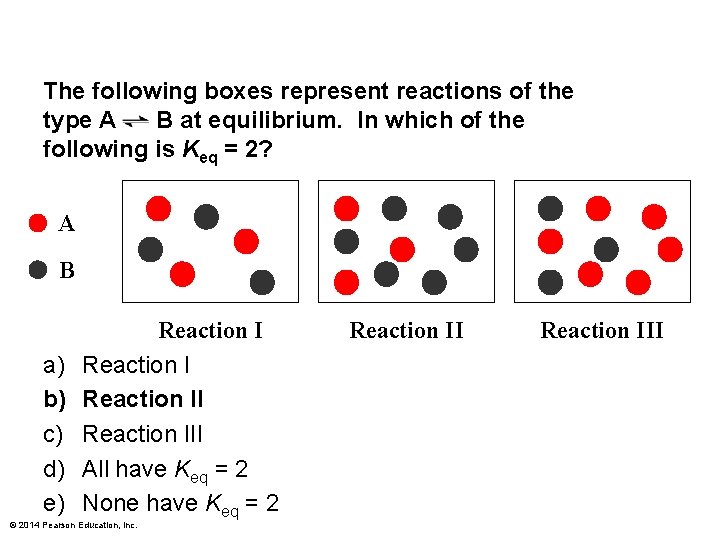
The following boxes represent reactions of the type A B at equilibrium. In which of the following is Keq = 2? A B a) b) c) d) e) Reaction III All have Keq = 2 None have Keq = 2 © 2014 Pearson Education, Inc. Reaction III

Which of the following is a true statement? a) If a chemical equation is reversed, K is inverted. b) If the coefficients in a chemical equation are multiplied by a factor, K is raised to the same factor. c) If two equations are added, the corresponding K values are multiplied to obtain the overall equilibrium constant. d) Statements 1– 3 are ALL true. e) None of the above statements are true. © 2014 Pearson Education, Inc.

Which of the following is a true statement? a) If a chemical equation is reversed, K is inverted. b) If the coefficients in a chemical equation are multiplied by a factor, K is raised to the same factor. c) If two equations are added, the corresponding K values are multiplied to obtain the overall equilibrium constant. d) Statements 1– 3 are ALL true. e) None of the above statements are true. © 2014 Pearson Education, Inc.

The equilibrium constant is given for two of the reactions below. Determine the value of the missing equilibrium constant. a) b) c) d) e) A (g) + 2 B (g) ⇌ AB 2 (g) Kc = 59 AB 2 (g) + B (g) ⇌ AB 3 (g) Kc = ? A (g) + 3 B (g) ⇌ AB 3 (g) Kc = 478 3. 5 × 10‒ 5 2. 8 × 104 8. 1 0. 12 89 © 2014 Pearson Education, Inc.

The equilibrium constant is given for two of the reactions below. Determine the value of the missing equilibrium constant. a) b) c) d) e) A (g) + 2 B (g) ⇌ AB 2 (g) Kc = 59 AB 2 (g) + B (g) ⇌ AB 3 (g) Kc = ? A (g) + 3 B (g) ⇌ AB 3 (g) Kc = 478 3. 5 × 10‒ 5 2. 8 × 104 8. 1 0. 12 89 © 2014 Pearson Education, Inc.

Methane reacts with water to form CO and H 2 as follows: CH 4 (g) + H 2 O (g) ⇌ CO (g) + 3 H 2 (g) The value of Kp at 298 K for the reaction is 7. 7 × 1024, calculate Kc. a) b) c) d) e) 4. 60 × 1027 6. 22 × 1021 3. 15 × 1023 1. 83 × 1024 1. 29 × 1022 © 2014 Pearson Education, Inc.

Methane reacts with water to form CO and H 2 as follows: CH 4 (g) + H 2 O (g) ⇌ CO (g) + 3 H 2 (g) The value of Kp at 298 K for the reaction is 7. 7 × 1024, calculate Kc. a) b) c) d) e) 4. 60 × 1027 6. 22 × 1021 3. 15 × 1023 1. 83 × 1024 1. 29 × 1022 © 2014 Pearson Education, Inc.

The reaction below has a Kc value of 61. What is the Kp for this reaction at 500. K? N 2 (g) + 3 H 2 (g) ⇌ 2 NH 3 (g) a) b) c) d) e) 1. 9 × 10– 2 3. 6 × 10– 2 28 61 15 © 2014 Pearson Education, Inc.

The reaction below has a Kc value of 61. What is the Kp for this reaction at 500. K? N 2 (g) + 3 H 2 (g) ⇌ 2 NH 3 (g) a) b) c) d) e) 1. 9 × 10– 2 3. 6 × 10– 2 28 61 15 © 2014 Pearson Education, Inc.

What is the equilibrium constant for the following reaction? 2 KCl. O 3 (s) ⇌ 2 KCl (s) + 3 O 2 (g) a) Keq = c) Keq = © 2014 Pearson Education, Inc. [KCl]2[O 2]3 [KCl. O 3]2 [2 KCl][3 O 2] [2 KCl. O 3] ΔH < 0 b) Keq = d) [KCl][O 2] [KCl. O 3] Keq = [O 2]3

What is the equilibrium constant for the following reaction? 2 KCl. O 3 (s) ⇌ 2 KCl (s) + 3 O 2 (g) a) Keq = c) Keq = © 2014 Pearson Education, Inc. [KCl]2[O 2]3 [KCl. O 3]2 [2 KCl][3 O 2] [2 KCl. O 3] ΔH < 0 b) Keq = [KCl][O 2] [KCl. O 3] d) Keq = [O 2]3
![Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr] Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr]](http://slidetodoc.com/presentation_image_h2/3c45d094b9cc3904ea4d9e8aa15e8d17/image-18.jpg)
Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr] = 0. 20 M, [H 2] = 0. 10 M, [Br 2] = 0. 12 M 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) a) b) c) d) e) 6. 0 × 10– 3 0. 30 16. 7 3. 33 7. 2 × 10– 4 © 2014 Pearson Education, Inc.
![Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr] Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr]](http://slidetodoc.com/presentation_image_h2/3c45d094b9cc3904ea4d9e8aa15e8d17/image-19.jpg)
Determine the Kc value for the following reaction if the equilibrium concentrations are: [HBr] = 0. 20 M, [H 2] = 0. 10 M, [Br 2] = 0. 12 M 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) a) b) c) d) e) 6. 0 × 10– 3 0. 30 16. 7 3. 33 7. 2 × 10– 4 © 2014 Pearson Education, Inc.

PCl 5 decomposes to PCl 3 and Cl 2. If the initial concentration of PCl 5 is 0. 200 M and the final concentration is 0. 120 M, what is the equilibrium constant for the reaction? a) b) c) d) e) 0. 0533 0. 0133 0. 0720 0. 0320 0. 00800 © 2014 Pearson Education, Inc.

PCl 5 decomposes to PCl 3 and Cl 2. If the initial concentration of PCl 5 is 0. 200 M and the final concentration is 0. 120 M, what is the equilibrium constant for the reaction? a) b) c) d) e) 0. 0533 0. 0133 0. 0720 0. 0320 0. 00800 © 2014 Pearson Education, Inc.

The equilibrium constant for the reaction 2 A ⇌ 3 B is 2. 3. If [A] = 0. 10 M and [B] = 0. 050 M, which of the following statements is true? a) b) c) d) e) The system is at equilibrium. The reaction will proceed to the left because Q < K. The reaction will proceed to the right because Q < K. The reaction will proceed to the left because Q > K. The reaction will proceed to the right because Q > K. © 2014 Pearson Education, Inc.

The equilibrium constant for the reaction 2 A ⇌ 3 B is 2. 3. If [A] = 0. 10 M and [B] = 0. 050 M, which of the following statements is true? a) b) c) d) e) The system is at equilibrium. The reaction will proceed to the left because Q < K. The reaction will proceed to the right because Q < K. The reaction will proceed to the left because Q > K. The reaction will proceed to the right because Q > K. © 2014 Pearson Education, Inc.

At 425 °C, Kp = 4. 18 × 10– 9 for the following reaction: 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) In a given experiment 0. 50 atm of HBr is placed in a container. What is the equilibrium pressure for H 2? a) b) c) d) 2. 1 × 10– 9 atm 4. 6 × 10– 5 atm 0. 25 atm 0. 50 atm © 2014 Pearson Education, Inc.

At 425 °C, Kp = 4. 18 × 10– 9 for the following reaction: 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) In a given experiment 0. 50 atm of HBr is placed in a container. What is the equilibrium pressure for H 2? a) b) c) d) 2. 1 × 10– 9 atm 4. 6 × 10– 5 atm 0. 25 atm 0. 50 atm © 2014 Pearson Education, Inc.

At 425 °C, Keq = 4. 18 × 10– 9 for the following reaction: 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) In a given experiment 0. 20 atm of HBr, 0. 010 atm of H 2, and 0. 010 atm of Br 2 are introduced into a container. What is the equilibrium concentration of HBr? a) b) c) d) e) 1. 4 × 10– 5 atm 0. 010 atm 0. 19 atm 0. 22 atm None of the above are correct © 2014 Pearson Education, Inc.

At 425 °C, Keq = 4. 18 × 10– 9 for the following reaction: 2 HBr (g) ⇌ H 2 (g) + Br 2 (g) In a given experiment 0. 20 atm of HBr, 0. 010 atm of H 2, and 0. 010 atm of Br 2 are introduced into a container. What is the equilibrium concentration of HBr? a) b) c) d) e) 1. 4 × 10– 5 atm 0. 010 atm 0. 19 atm 0. 22 atm None of the above are correct © 2014 Pearson Education, Inc.
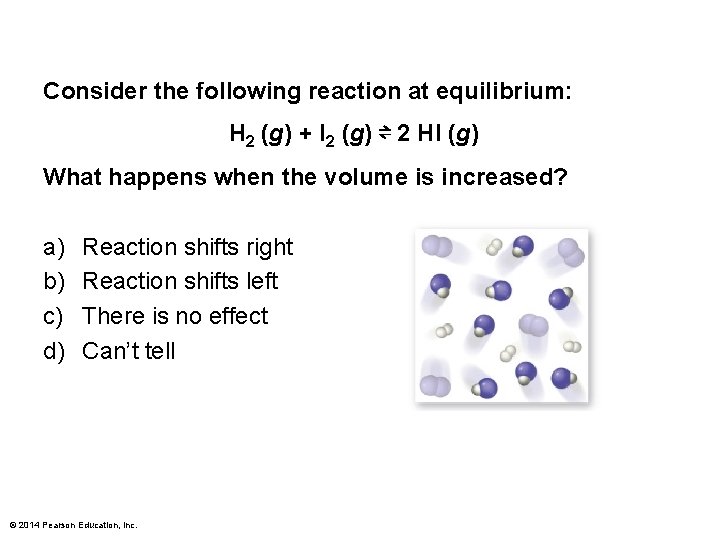
Consider the following reaction at equilibrium: H 2 (g) + I 2 (g) ⇌ 2 HI (g) What happens when the volume is increased? a) b) c) d) Reaction shifts right Reaction shifts left There is no effect Can’t tell © 2014 Pearson Education, Inc.
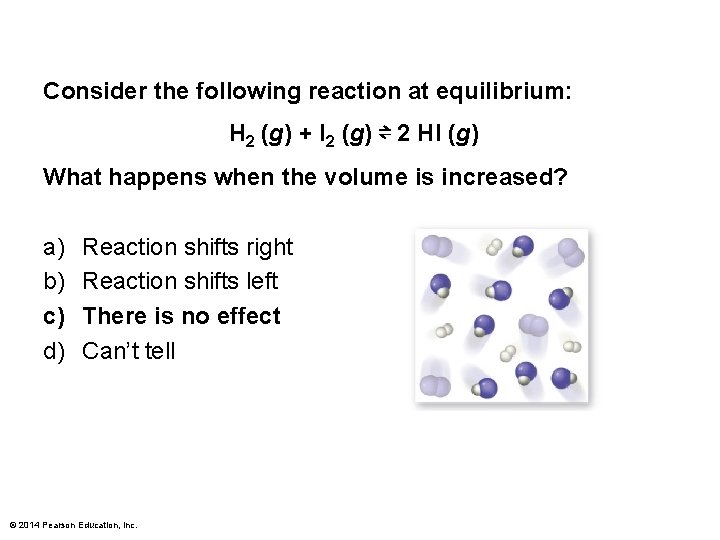
Consider the following reaction at equilibrium: H 2 (g) + I 2 (g) ⇌ 2 HI (g) What happens when the volume is increased? a) b) c) d) Reaction shifts right Reaction shifts left There is no effect Can’t tell © 2014 Pearson Education, Inc.
Consider the following reaction at equilibrium: 2 KCl. O 3 (s) ⇌ 2 KCl (s) + 3 O 2 (g) ΔH < 0 What happens when the reaction mixture is heated? a) b) c) d) Reaction shifts right Reaction shifts left There is no effect Can’t tell © 2014 Pearson Education, Inc.
Consider the following reaction at equilibrium: 2 KCl. O 3 (s) ⇌ 2 KCl (s) + 3 O 2 (g) ΔH < 0 What happens when the reaction mixture is heated? a) b) c) d) Reaction shifts right Reaction shifts left There is no effect Can’t tell © 2014 Pearson Education, Inc.

Given the following reaction, how could you increase the concentration of products? N 2 (g) + 3 H 2 (g) ⇌ 2 NH 3 (g) a) b) c) d) e) ΔH = – 46. 19 k. J Increase volume, add N 2, or increase temperature Increase volume, add N 2, or decrease temperature Decrease volume, remove N 2, or increase temperature © 2014 Pearson Education, Inc.

Given the following reaction, how could you increase the concentration of products? N 2 (g) + 3 H 2 (g) ⇌ 2 NH 3 (g) a) b) c) d) e) ΔH = – 46. 19 k. J Increase volume, add N 2, or increase temperature Increase volume, add N 2, or decrease temperature Decrease volume, remove N 2, or increase temperature © 2014 Pearson Education, Inc.
- Slides: 33