Overview of electrochemistry 1 Equilibrium electrochemistry Dynamic electrochemistry
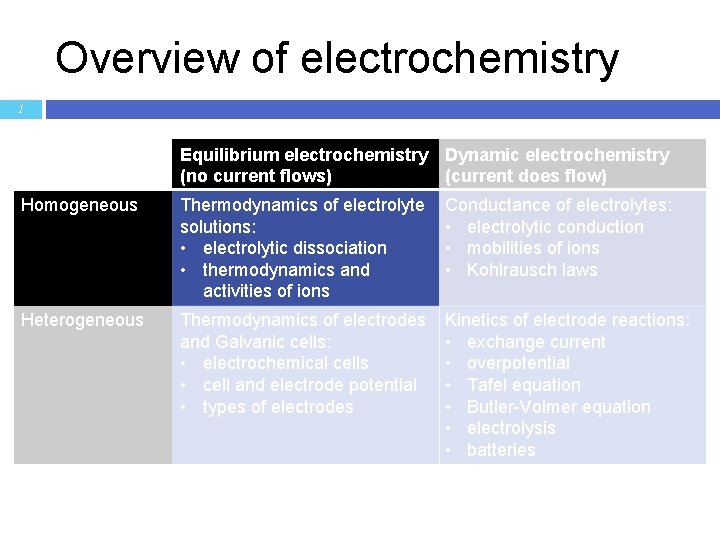
Overview of electrochemistry 1 Equilibrium electrochemistry Dynamic electrochemistry (no current flows) (current does flow) Homogeneous Thermodynamics of electrolyte solutions: • electrolytic dissociation • thermodynamics and activities of ions Conductance of electrolytes: • electrolytic conduction • mobilities of ions • Kohlrausch laws Heterogeneous Thermodynamics of electrodes and Galvanic cells: • electrochemical cells • cell and electrode potential • types of electrodes Kinetics of electrode reactions: • exchange current • overpotential • Tafel equation • Butler-Volmer equation • electrolysis • batteries

HOMOGENEOUS EQUILIBRIUM ELECTROCHEMISTRY: THERMODYNAMICS OF ELECTROLYTES I. Thermodynamics of electrolytes. A. Characterization of electrolytes B. Thermodynamic formation functions of ions C. Activities of ions in solution, the Debye–Hückel theory

I. Thermodynamics of electrolytes A. Characterization of electrolytes 3 Electrolytes are special types of solutions: homogeneous, multicomponent systems Ionic compounds dissociate into anion(s) and cation(s) in polar solvents (Svante Arrhenius, Nobel prize in 1903) Mostly the same general properties as for other (dilute) solutions: � colligative properties (freezing point depression, boiling point elevation, osmosis, distribution…) � may both be ideal and real Unique property: � conduction of electricity

I. Thermodynamics of electrolytes A. Characterization of electrolytes 4 Electroneutrality – there are no solutions containing cation only, or anion only! The electric conduction is an ion conduction where κ increases with increasing temperature (versus metals). Molten electrolytes (e. g. Na. Cl(l)) also conduct electricity.

I. Thermodynamics of electrolytes A. Characterization of electrolytes 5 Acids, bases, salts are electrolytes: � Salts: practically complete dissociation � Acids and bases: weak or strong Dissociation is an equilibrium: � Kd dissociation constant or � α degree of dissociation Electrolytic dissociation: HA(aq) + H 2 O(l) � (acid-base A–(aq) + H 3 O+(aq) equilibria, p. H, buffers, p. H-titration, p. Ka � complexation) indicators,
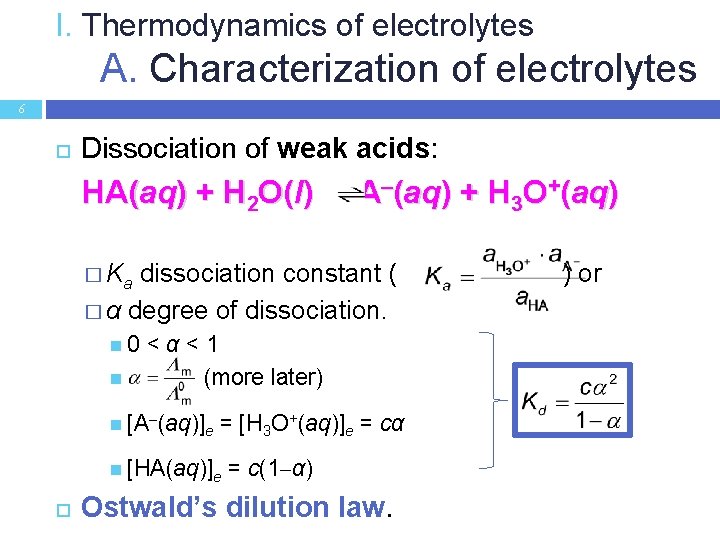
I. Thermodynamics of electrolytes A. Characterization of electrolytes 6 Dissociation of weak acids: HA(aq) + H 2 O(l) A–(aq) + H 3 O+(aq) � Ka dissociation constant ( � α degree of dissociation. 0 <α<1 (more later) [A–(aq)]e = [H 3 O+(aq)]e = cα [HA(aq)]e = c(1 α) Ostwald’s dilution law. ) or

I. Thermodynamics of electrolytes A. Characterization of electrolytes 7 Complete dissociation in the saturated solutions of solid salts. Ions are in equilibrium with a solid phase rather than individual molecules: KA(s) K+(sol) + A– (sol) Exception: e. g. Hg. Cl 2 (S = 1. 3 10 5 mol/dm 3) Hg. Cl 2(s) Hg. Cl 2(aq) Hg 2+(aq) + 2 Cl– (aq) 0. 1 M Hg. Cl 2

B. Thermodynamic formation functions of ions 8 Thermodynamic formalism: is close to ideal until ca. 10 3 mol/dm 3, � above: real solutions. � Activity (a) is used instead of concentration (c or m). � behavior

B. Thermodynamic formation functions of ions 9 Problem: solutions never contain one type of ion (e. g. cation) only, there is always at least one other oppositely charged ion. Arbitrary conventions (but widely agreed on): � Δf H (H+, aq) = 0 J/mol and � Δf G (H+, aq) = 0 J/mol. What’ more: � Δf S (H+, aq) = 0 J/(K mol) at every temperature. This entropy is measured on a relative scale! � Absolute entropy (third law of thermodynamics) Δf S (H+, aq) = -21 J/(K mol).

B. Thermodynamic formation functions of ions 10 Example: Ag(s) + ½Cl 2(g) → Ag+(aq) + Cl– (aq) § but Δr H for the processes Ag(s) → Ag+(aq) + e– and ½Cl 2(g) + e– → Cl– (aq) are not measurable separately. Solution: start from H+(aq) and proceed to other ions: § ½H 2(g) + ½Cl 2(g) → H+(aq) + Cl– (aq) Δr H = Δf H (H+, aq) + Δf H (Cl , aq) = 167 k. J/mol, and thus Δf H (Cl , aq) = 167 k. J/mol. § Now Δf H (Ag+, aq) is availabe. § Δr H = Δf H (Ag+, aq) + Δf H (Cl , aq) = 61. 5 k. J/mol,

B. Thermodynamic formation functions of ions 11 Example: Born Haber cycle for ion formation: ½H 2(g) + ½Cl 2(g) → H+(aq) + Cl– (aq) ΔG (k. J/mol) dissociation of H 2: ½ H 2(g) → H(g) +203 ionization of H: H(g) → H+(g) + e +1318 hydration of H+: H+ (g) → H+(aq) x=0 dissociation of Cl 2: ½ Cl 2(g) → Cl(g) +106 electron affinity of Cl: Cl(g) + e → Cl (g) -355 hydration of Cl : Cl (g) → Cl (aq) y Δf G (Cl , aq) = x + y + 1272 k. J/mol. This is an indirect method for determining the directly not measurable Gibbs free energy of hydration (or solvation).
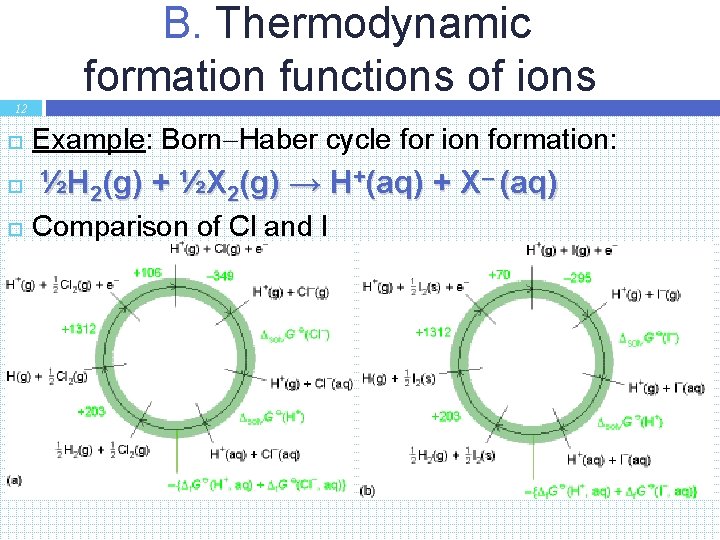
B. Thermodynamic formation functions of ions 12 Example: Born Haber cycle for ion formation: ½H 2(g) + ½X 2(g) → H+(aq) + X– (aq) Comparison of Cl and I

B. Thermodynamic formation functions of ions 13 Δhyd G cannot be measured experimentally, but can be calculated from the Born Haber cycle. The Born equation can be used for estimating the electrostatic component of Gibbs free energy of solvation of an ion: � z: charge of ion � e: elementary charge, 1. 6022× 10− 19 C � 0: vacuum permittivity � r: dielectric constant of the solvent � ri: effective radius of ion

C. Activities of ions in solution 14 Solutions containing ions(electrolytes) are not ideal because of electrostatic interactions. Therefore, activities are used instead of concentrations (e. g. equilibria or electrode potentials). So far: � = + RTlna � a = ·m/m � where m is molality. = + RTlnm/m + RTln = 0 + RTln where 0 is the chemical potentia of an ideal dilute solution with the same molality.
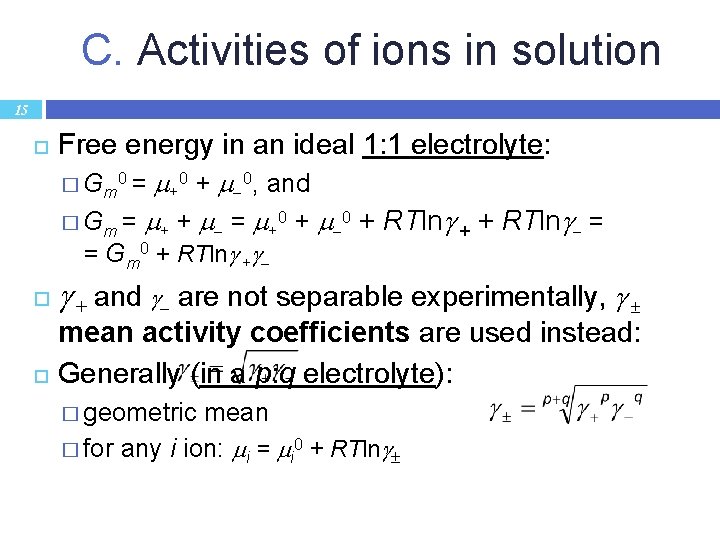
C. Activities of ions in solution 15 Free energy in an ideal 1: 1 electrolyte: +0 + 0, and � Gm = + + = +0 + RTln + + RTln = � G m 0 = = Gm 0 + RTln + + and are not separable experimentally, mean activity coefficients are used instead: Generally (in a p: q electrolyte): � geometric mean � for any i ion: i = i 0 + RTln

C. Activities of ions in solution 16 Questions: � Value of . � Can it be measured? � YES. � Can it be calculated? � YES. See Nernst equation. Debye–Hückel theory
C. Debye–Hückel theory 17 Debye–Hückel limiting law: Non-ideal behavior is caused by the electrostatic interaction between ions and it is sufficient to consider this one only in theory. � Values of can be obtained. Model: � Around any given ion, ions form a spherical excess. � This ion cloud reduces the chemical potential of the center. counter cloud and are in ion in

C. Debye–Hückel theory 18 based on the Debye–Hückel theory: Result: a combination of constants (in water at 25 C, 0. 509 (mol/kg)½ � I: ionic strength of the solution: � I involves all ions! � A: This formula is only valid at low concentrations, so, it is called the Debye–Hückel limiting law. An extension can be used at higher concentrations:
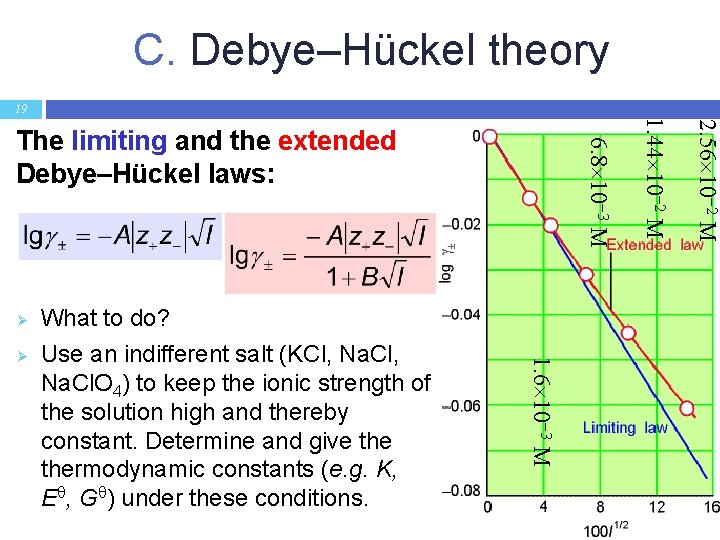
C. Debye–Hückel theory 19 2. 56 10 2 M 1. 6 10 3 M Ø What to do? Use an indifferent salt (KCl, Na. Cl. O 4) to keep the ionic strength of the solution high and thereby constant. Determine and give thermodynamic constants (e. g. K, E , G ) under these conditions. 1. 44 10 2 M Ø 6. 8 10 3 M The limiting and the extended Debye–Hückel laws:

Overview of electrochemistry 20 Equilibrium electrochemistry (no current flows) Dynamic electrochemistry (current does flow) Homogeneous Thermodynamics of electrolyte solutions: • electrolytic dissociation • thermodynamics and activities of ions Conductance of electrolytes: • electrolytic conduction • mobilities of ions • Kohlrausch laws Heterogeneous Thermodynamics of Galvanic cells and electrodes: • electrochemical cells • cell and electrode potential • types of electrodes Kinetics of electrode reactions: • exchange current • overpotential • Tafel equation • Butler-Volmer equation • electrolysis • batteries

HETEROGENEOUS EQUILIBRIUM ELECTROCHEMISTRY: Thermodynamics of electrodes and Galvanic cells I. Galvanic vs. electrolytic cells II. Galvanic cells: A. B. C. D. E. F. G. chemistry and thermodynamics Parts and usual notations of Galvanic cells Potential of a Galvanic cell Electrolyte and electrode concentration cells Measuring cell potential Galvanic cells (chemical sources of electricity) in practice Fuel cells

HETEROGENEOUS EQUILIBRIUM ELECTROCHEMISTRY: Thermodynamics of Galvanic cells and electrodes I. Galvanic vs. electrolytic cells III. Electrodes H. I. J. K. IV. Dependence of the electrode potentials on the composition Measuring electrode potentials The scale of electrode potentials Types of electrodes Summary

Thermodynamics of Galvanic cells and electrodes 23 Electrochemical cells: Galvanic cells vs. electrolysis Galvanic cells : � � connection between standard cell potential and standard reaction Gibbs free energy: –νFEcell = Δr. G connection between cell potential and composition (reaction quotient): Ecell = Ecell – (RT/νF) ln. Q (Nernst equation) Galvanic cell in equilibrium: RT ln. K = νFEcell types of Galvanic cells, practical applications Electrodes: � Nernst equation of the electrode: E + (RT/νF) lnai electrode reactions, types of electrodes E=

Thermodynamics of Galvanic cells and electrodes 24 In electrochemical cells, redox reactions proceed in a spatially separated arrangement. [Electrolytic dissociation is also a chemical change, but does not involve changes in oxidation states, only charge separation in a single solution. Acid-base reactions cannot be separated in space. ] � Parts of the cell (heterogeneous system): � two (metal) electrodes (anode and cathode) immersed into the solutions of their own ions (electrolytes) Electrode and electrolyte together form the electrode compartment. To connect them electrically, a salt bride or diaphragm is needed. Two different approaches: � � spontaneous Galvanic cell vs. forced electrolysis.
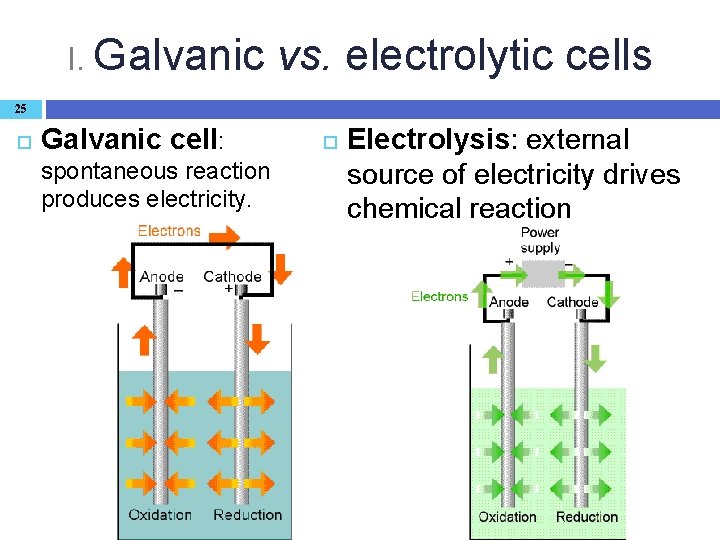
I. Galvanic vs. electrolytic cells 25 Galvanic cell: spontaneous reaction produces electricity. Electrolysis: external source of electricity drives chemical reaction

I. Galvanic vs. electrolytic cells 26 Anode: Spontaneous oxidation Increase in oxidation number Substance loses electrons Negative (-) Forced oxidation Increase in oxidation number Substance loses electrons Positive (+) Spontaneous reduction Cathode: forced reduction Decrease in oxidation number Substance gains electrons Positive (+) Negative (-)

I. Galvanic vs. electrolytic cells 27 Neither the Galvanic cells nor the electrolytic cells are in thermodynamic equilibrium, but if we allow the electron transition, the cell will go towards and finally reach the chemical equilibrium. There are chemical (redox) reactions in the cell: � this is always e transfer, sometimes also atom transfer, � reduction (e– gain) and oxidation (e– loss) � reduction and oxidation is separated in space � there is ionic conduction in the electrolyte solution � and metallic conduction in the outer circuit (wire). Note: The oxidizing agent oxidizes another substance while itself is reduced and vice versa.

II. A. Galvanic cells: chemistry and thermodynamics 28 Simple experiment: red elemental copper deposits on a zinc rod immersed into a solution of copper(II) sulfate, and – although this is not directly visible – zinc ions appear in the solution. The reduction of copper(II) and the oxidation of zinc proceeds in the same space: Zn(s) + Cu 2+(aq) → Zn 2+(aq) + Cu(s)
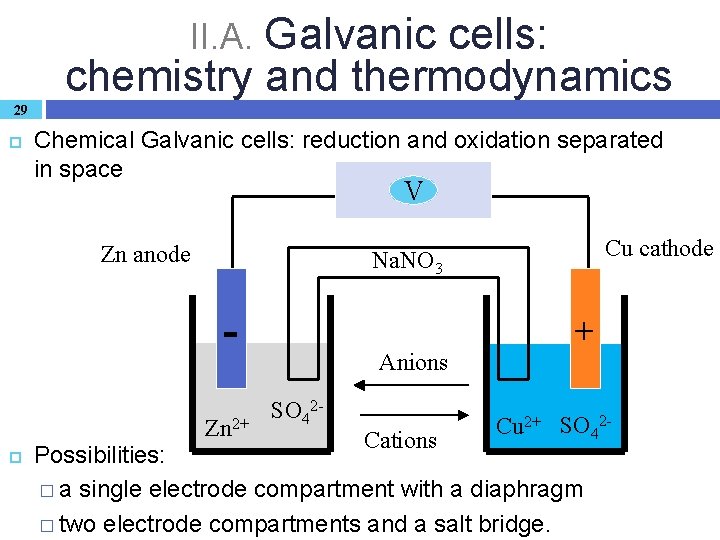
II. A. Galvanic cells: chemistry and thermodynamics 29 Chemical Galvanic cells: reduction and oxidation separated in space V Zn anode Zn 2+ Cu cathode Na. NO 3 Anions SO 42 - Cations + Cu 2+ SO 42 - Possibilities: � a single electrode compartment with a diaphragm � two electrode compartments and a salt bridge.

II. A. Galvanic cells: chemistry and thermodynamics 30 Separate the previous Zn(s) + Cu 2+(aq) → Zn 2+(aq) + Cu(s) reaction into oxidation and § Zn(s) → Zn 2+(aq) + 2 e– § red 1 → ox 1 + e– § oxidation: e– loss § anode: (-) electrode § anodic oxidation § notation: (left) or (1) reduction: § Cu 2+(aq) + 2 e– → Cu(s) § ox 2 + e– → red 2 § reduction: e– gain § cathode: (+) electrode § cathodic reduction § notation: (right) or (2) Both sides are redox pairs: ox + νe– → red

II. A. Galvanic cells: chemistry and thermodynamics 31 In the convention of electrochemistry, both processes are written in the direction of reduction: �– � {Zn 2+(aq) + 2 e– → Zn(s)} Cu 2+(aq) + 2 e– → Cu(s) The net reaction is now the difference of the two reduction processes: (right) – (left) This makes good common sense, as Ecell is always positive in agreement with the negative Δr. G of the spontaneous process: Ecell = Eright electrode – Eleft electrode

II. B. Parts and usual notations of Galvanic cells 32 Galvanic cell (or voltaic/electrochemical cell) = � 2 half cells or � 2 half reactions or � 2 electrodes Notation: Particularly: (–) Zn(s)│Zn. SO 4(aq) ║ Cu. SO 4(aq)│Cu(s) (+) phase boundary (solid/solution, solid/gas) � ║ surface on the boundary – no liquid junction potential (liquid junction potential is minimized with the use of an appropriate salt bridge. � ⋮ electrolytes in contact with each other �│

II. C. Potential of a Galvanic cell 33 In a Galvanic cell, there is electric potential (potential difference): it is called cell potential. Connection between the cell potential and reaction Gibbs energy: –νFEcell = Δr. G Derivation of the relationship: � the maximum useful work of the chemical reaction: Δ r. G � electric work (in physics): –νFEcell � In a Galvanic cell, the two must be the same.

II. C. Potential of a Galvanic cell 34 In a reversible reaction, the actual value of Δr. G depends on the reaction quotient Q: � Δ r. G = Δr. G + RT ln. Q, For the cell reaction: = Δr. G + RT ln. Q � Ecell = –(Δr. G /νF) – (RT/νF) ln. Q � Let –(Δr. G /νF) = Ecell , the standard cell potential � Finally, the Nernst equation for the cell : � –νFEcell = Ecell – ( RT / v. F ) ln Q

II. C. Potential of a Galvanic cell 35 In equilibrium (i. e. when Ecell = 0), Q = K. From this, the connection between standard cell potential and equilibrium constant can be written: Ecell = (RT/νF) ln. K Relationship between cell potential (Ecell ), � equilibrium constant (K) and � standard reaction Gibbs free energy (Δr. G ): � standard νF Ecell = RT ln K = – Δr G So, equilibrium electrochemistry gives the connection between equilibrium chemistry and thermodynamics.

II. C. Potential of a Galvanic cell 36 Connections between cell potential and othermodynamic functions (H, S) : � Basis: temperature dependence of Ecell and Ecell. � Well-known � from this: –Δr. S = (∂(–νFEcell)/∂T)p � since from thermodynamics: –S = (∂G/∂T)p Δr. G = Δr. H – T·Δr. S, Δr. H = Δr. G + T·Δr. S = This is also valid for the standard values. So, Δr. G , Δr. S and Δr. H can be determined based on electrochemical measurements.
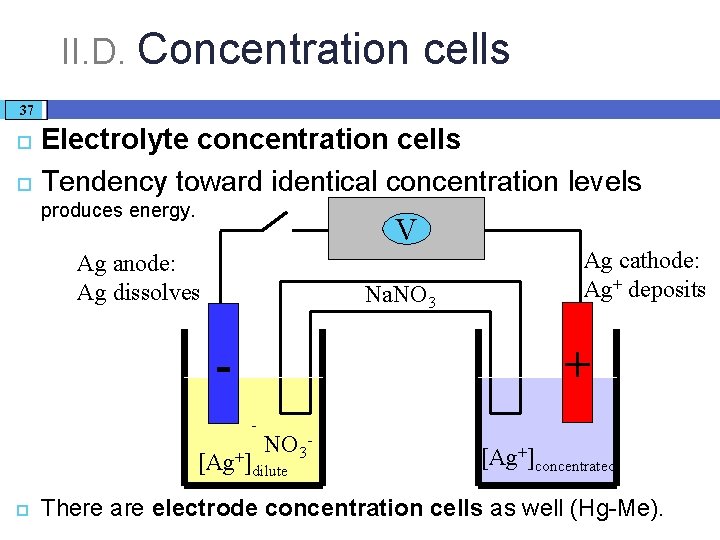
II. D. Concentration cells 37 Electrolyte concentration cells Tendency toward identical concentration levels produces energy. V Ag anode: Ag dissolves Na. NO 3 + - NO 3 - [Ag+]dilute Ag cathode: Ag+ deposits [Ag+]concentrated There are electrode concentration cells as well (Hg-Me).

II. D. Concentration cells 38 In an electrolyte concentration Galvanic cell: �a difference of 1 order of magnitude in concentration (or activity) yields 59 m. V of cell potential (at 25 °C). � Across a biological membrane, typically there is a 20 -30 times difference in [K+] yielding ca. 77 m. V of potential difference – also an electrolyte concentration cell. � This membrane potential has a role in nerve signal transmission.

II. Galvanic cells 39 Galvanic cells with melted salts: � Such Galvanic cells are seldom used. � Melt electrolysis is important in industry: e. g. electrolysis of Al 2 O 3 (alumina). Galvanic cells without solvents: � The use of solvent should be avoided if possible: it may be sensitive to heat and/or environmentally pollutant. � Practical examples will be shown later (batteries).

II. Galvanic cells 40 With no current flowing (I = 0), the potential is called: electromotive force (e. m. f. ). The Galvanic cell is able to do work (maintain current), when it is far from chemical equilibrium. When it is in use the voltage is lower and it is called: output voltage. When (the redox reaction) reaches the chemical equilibrium, e. m. f. = 0, no more current flows (the battery is „dead”). The process is (theoretically) always reversible and the battery can be „recharged” with electrolysis: this is called rechargeable battery.

II. E. Measuring cell potentials 41 � electromotive force (no current flows): e. m. f. � with current (producing electric work): actual cell potential of a working electrode Measuring electromotive force (e. m. f. ): � compensation method (fully current-free), classical: Du. Bois–Reymond–Poggendorf � voltmeter with large internal resistance (R >1010 Ω). Today this is a common piece of laboratory equipment (e. g. p. H-meter). � In practice, voltages of 0 ≤ 3 V have to be measured with a precision of 0. 1 m. V (10 -4 V).

II. F. Galvanic cells (chemical sources of electricity) in practice 42 Galvanic cell (primary battery): non-rechargeable (when it reaches thermodynamic equilibrium) Accumulator (secondary or rechargeable battery): rechargeable many (hundreds of) times (cars, hearing aids, etc. ) Fuel cell: production of electricity from combustion –with continuous input of reactants Significance: (small) source of electricity anytime anywhere without access to the grid or a large generator, � operation is very easy (no training necessary), � portability (radio, watch, cell phone, pacemaker etc. ). �

II. F. Galvanic cells (chemical sources of electricity) in practice 43 Qualification: voltage (can be increased by using several cells in a series) � capacity (scalable) � current output (can be increased by using several parallel cells) � power/mass ratio � efficiency � voltage stability (under current and in time) � fast/slow current output change � recharge cycles (for accumulators) � temperature range � environmental pollution (Hg, Cd free!) � price �

II. F. Galvanic cells (chemical sources of electricity) in practice 44 Galvanic cells in practice: � Leclanché cell � Alkaline manganese oxide � Lithium battery � Mercury oxide battery � (Daniell cell) � (Volta’s battery / Voltaic pile)
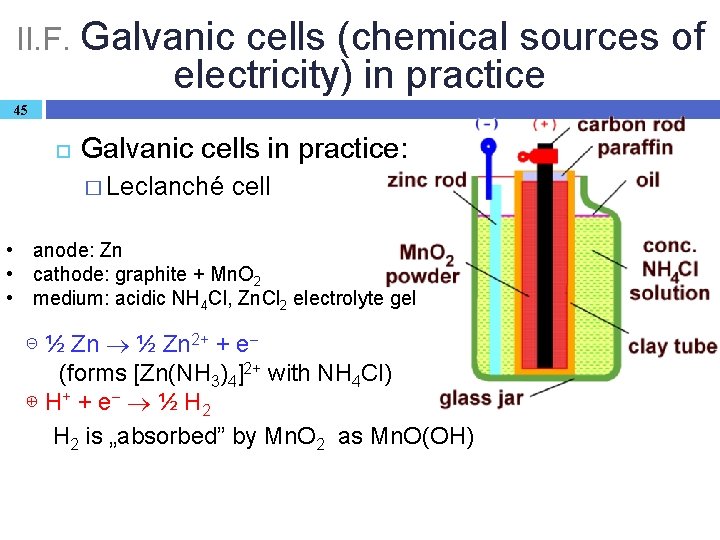
II. F. Galvanic cells (chemical sources of electricity) in practice 45 Galvanic cells in practice: � Leclanché cell • anode: Zn • cathode: graphite + Mn. O 2 • medium: acidic NH 4 Cl, Zn. Cl 2 electrolyte gel ⊝ ½ Zn 2+ + e (forms [Zn(NH 3)4]2+ with NH 4 Cl) ⊕ H+ + e ½ H 2 is „absorbed” by Mn. O 2 as Mn. O(OH)

G. Galvanic cells (chemical sources of electricity) in practice 46 Galvanic cells in practice: � Alkaline manganese oxide (alkaline battery, 1. 5 V) • anode: Zn • cathode: graphite + Mn. O 2 • medium: KOH, Zn. Cl 2 electrolyte gel ⊝ ½ Zn + OH ½ Zn(OH)2 + e ⊕ H+ + e ½ H 2 is „absorbed” by Mn. O 2 as Mn. O(OH) Most common battery today, 5 -7 years of storage time. AA size: 2000 m. Ah

G. Galvanic cells (chemical sources of electricity) in practice 47 Galvanic cells in practice: � 9 V battery
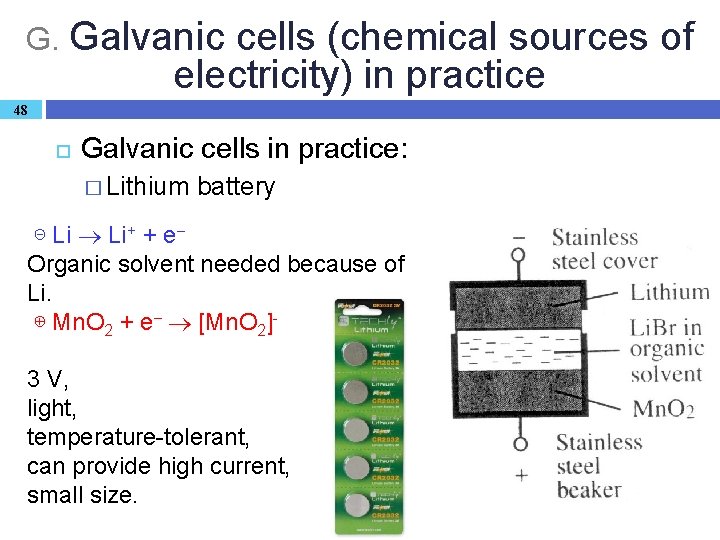
G. Galvanic cells (chemical sources of electricity) in practice 48 Galvanic cells in practice: � Lithium battery ⊝ Li Li+ + e– Organic solvent needed because of Li. ⊕ Mn. O 2 + e– [Mn. O 2]3 V, light, temperature-tolerant, can provide high current, small size.

G. Galvanic cells (chemical sources of electricity) in practice 49 Galvanic cells in practice: � Mercury oxide battery (Ruben–Mallory) net reaction: Zn + Hg. O Zn. O + Hg 1. 25 V, constant! Hearing aids, pacemakers Hg – environmental concerns!
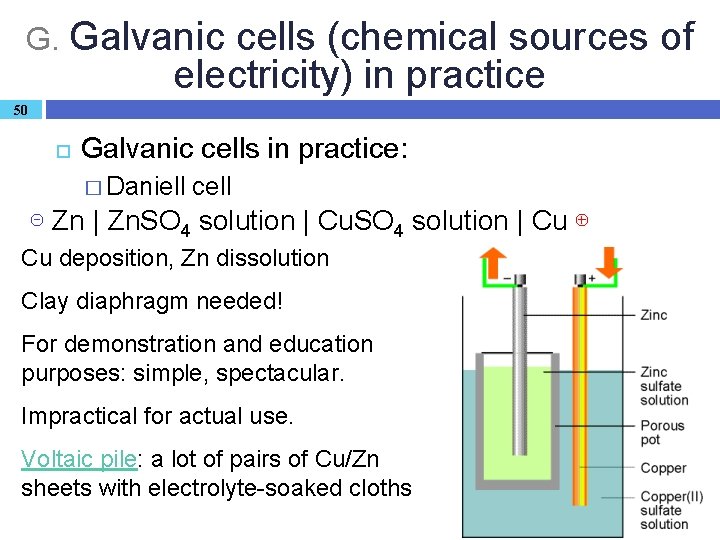
G. Galvanic cells (chemical sources of electricity) in practice 50 Galvanic cells in practice: � Daniell cell ⊝ Zn | Zn. SO 4 solution | Cu ⊕ Cu deposition, Zn dissolution Clay diaphragm needed! For demonstration and education purposes: simple, spectacular. Impractical for actual use. Voltaic pile: a lot of pairs of Cu/Zn sheets with electrolyte-soaked cloths

II. G. Fuel cells 51 Chemical energy → electricity: indirect in a power plant (two steps with heat as the intermediate): maximum efficiency about 40% Fuel cell: special Galvanic cell that produces electricity directly based on combustion (75 -90%). The hydrogen + oxygen reaction (in KOH) is separated in space. Catalyst needed, T = 70– 140 °C � anode reaction: 2 H 2(g) + 4 OH– → 4 H 2 O + 4 e– � cathode reaction: O 2(g) + 2 H 2 O(l) + 4 e– → 4 OH– Other fuels (methane, methanol) can also be used in a similar fashion.
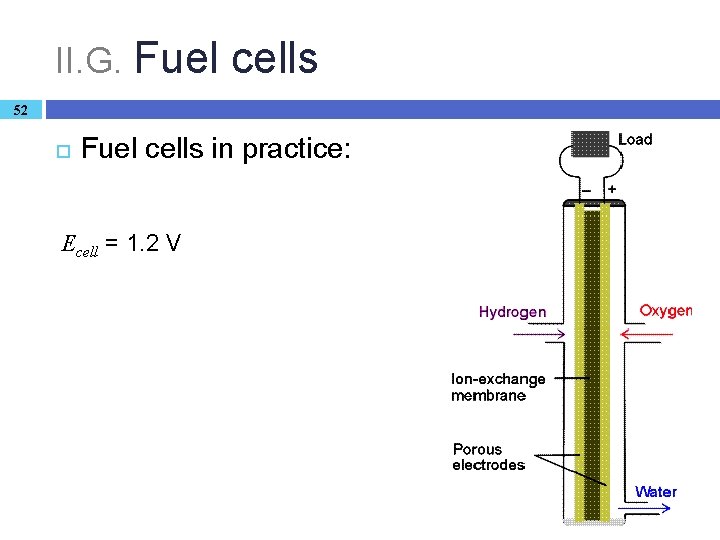
II. G. Fuel cells 52 Fuel cells in practice: Ecell = 1. 2 V

III. Electrodes 53 Connection between cell potentials and electrode potentials, a formal interpretation of electrode potentials: � the cell potential is the difference of two electrode potentials: Ecell = Eright – Eleft � The (absolute) potential of a single electrode can never be measured, neither it can be calculated. � A successful solution: preparation of a Galvanic cell in which the left hand electrode is the standard hydrogen electrode (SHE).

III. H. Dependence of the electrode potential on the composition: the Nernst equation 54 The principle of the definition is SHE-reference: Pt | H 2(g) | H+(aq) ║Ag+(aq) | Ag(s) Cell reaction: ½ H 2(g) + Ag+(aq) → H+(aq) + Ag(s) The Nernst equation of the cell expressed with Q (as ν = 1 and ESHE = 0): Ecell = Ecell – RT/F ln. Q = E (Ag+/Ag) – RT/F ln. Q as Q = 1/a(Ag+) is valid: E(Ag+/Ag) = E (Ag+/Ag) + RT/F ln a(Ag+) Nernst equation for the (Ag+/Ag) electrode. Generally (metal/metal ion electrodes): Ei = Ei + (RT/νF) lnai
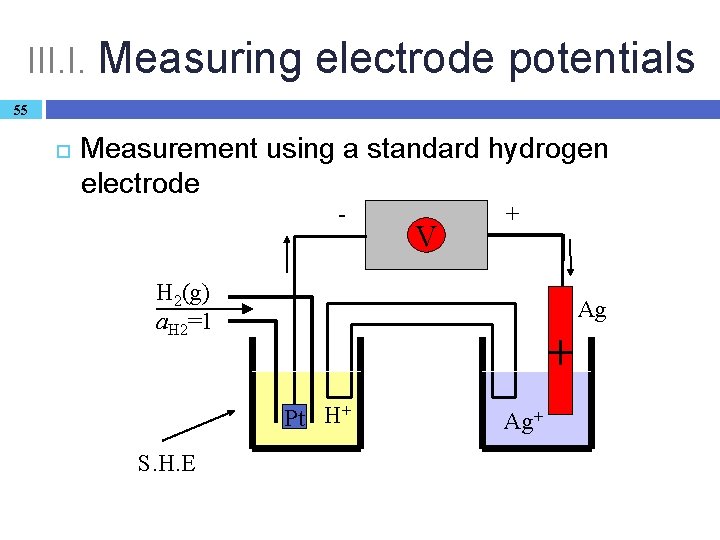
III. I. Measuring electrode potentials 55 Measurement using a standard hydrogen electrode - V + H 2(g) a. H 2=1 Ag + Pt H+ S. H. E Ag+

III. I. Measuring electrode potentials 56 Reference point SHE: E (H+/H 2) = 0. 000 V � complicated, impractical, dangerous (H 2 gas!) Practical reference electrodes: � stable potential independent of many external conditions (metal/insoluble salt electrode) � Ag(s) | Ag. Cl(s) | Cl–(aq) E = + 0. 22 V (silver/silver chloride electrode) � Pt|Hg(l)|Hg 2 Cl 2(s)|Cl–(aq) E = + 0. 27 V (saturated calomel electrode)

III. J. Scales of electrode potentials 57 As the potential of SHE is 0. 000 V at each temperature by convention. . . the electromotive force of the Galvanic cell is the same as the potential of the electrode on the right. Result: � Positive and negative electrode potentials in the range from +3 V to – 2 V � The electrode potential can be arranged as a series

III. J. Scales 58 Oxidizing agent Reducing agent of electrode potentials

59 Electrochemical series of metals Highly reducing Reacts readily with cold water Reacts slowly with water vapor Reacts readily with acids Reacts slowly with acids No reaction with acids Almost non-reducing
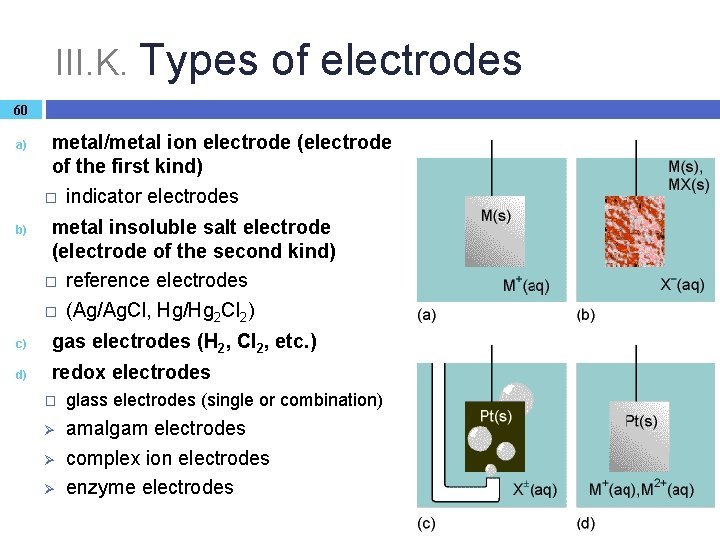
III. K. Types of electrodes 60 a) b) metal/metal ion electrode (electrode of the first kind) � indicator electrodes metal insoluble salt electrode (electrode of the second kind) � reference electrodes � (Ag/Ag. Cl, Hg/Hg 2 Cl 2) c) gas electrodes (H 2, Cl 2, etc. ) d) redox electrodes � glass electrodes (single or combination) Ø amalgam electrodes complex ion electrodes enzyme electrodes Ø Ø

III. K. Types of electrodes 61 a) Electrodes of the first kind � Electrode potential is determined primarily by the metal ion activity (so these electrodes are often used to determine activity values): Ei = Ei + (RT/νF) lnai b) Electrodes of the second kind � Electrode potential is determined by the counter ion concentration of the weakly soluble salt: MX(s) M+(aq) + X–(aq) Ksol = a(M+) a(X–) � Using a high and constant [X-], the electrode potential can be kept constant, so, these electrodes are used as reference electrodes. � Ksol solubility product can be determined by measuring the potential value.
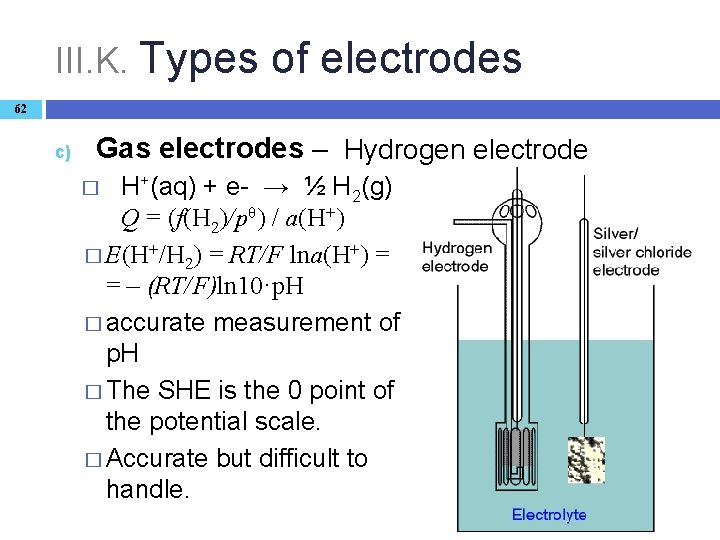
III. K. Types of electrodes 62 c) Gas electrodes – Hydrogen electrode H+(aq) + e- → ½ H 2(g) Q = (f(H 2)/pθ) / a(H+) � E(H+/H 2) = RT/F lna(H+) = = – (RT/F)ln 10·p. H � accurate measurement of p. H � The SHE is the 0 point of the potential scale. � Accurate but difficult to handle. �
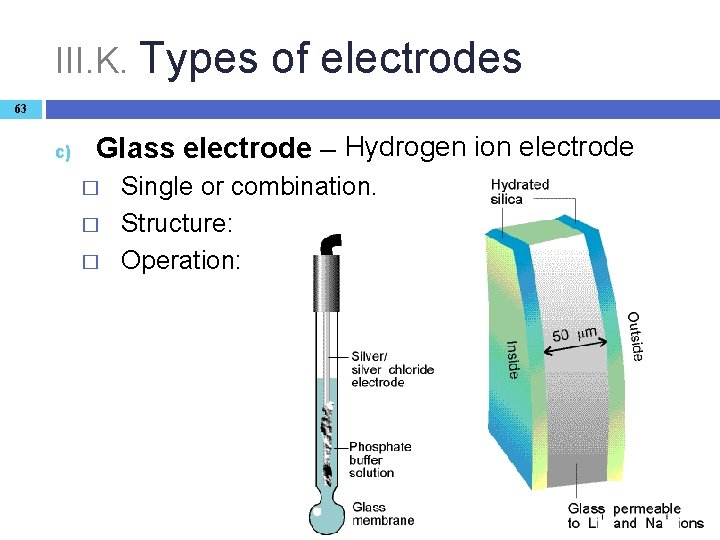
III. K. Types of electrodes 63 c) Glass electrode – Hydrogen ion electrode � � � Single or combination. Structure: Operation:
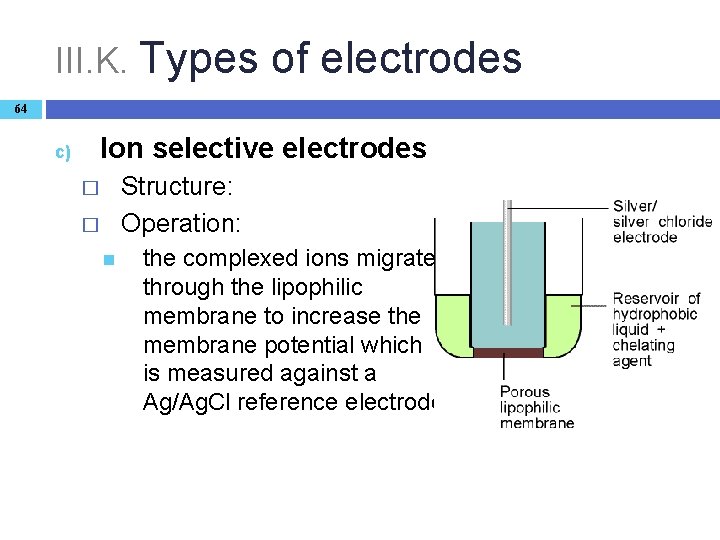
III. K. Types of electrodes 64 Ion selective electrodes c) Structure: Operation: � � the complexed ions migrate through the lipophilic membrane to increase the membrane potential which is measured against a Ag/Ag. Cl reference electrode.

III. K. Types of electrodes 65 d) Redox electrodes � � � e) „Old” nomenclature Ei = Eiθ + (RT/νF) ln(aox/ared) There are two different oxidation states of the same (metal) ion in a solution (e. g. Fe 3+/Fe 2+) and an inert Pt electrode. Amalgam electrodes � Metal mercury amalgams can also be electrodes: their potential depends on the metal concentration of the amalgam. (Example: electrolysis of Na. Cl. )

IV. Summary 66 Practical use of electrochemistry: � to find the spontaneous direction of a chemical reaction using standard electrode potentials, � (from these) determining the value of K equilibrium constant, � determining solubility products (Ksol), � measuring p. H and determining p. Ka’s, � determining complex stability constant values, � measuring ion activities and mean activity coefficients experimentally, � determining thermodynamic functions (G, S, H).

IV. Summary 67 In galvanic cells: �A non-equilibrium, redox-type chemical process can perform electrical work when the two half-reactions are separated in space. � The two electrodes thus formed – individually – has potential, and the potential difference is called cell potential. As a result, and external current flow (work) is generated. � To calculate e. m. f. : Ecell = Ecathode, right – Eanode, left � E depends of the chemical identities and the activities of the substance. E also depends on the temperature and solvent. (see: Nernst equations)

IV. Summary 68 In galvanic cells: � The reaction proceeds until the equilibrium is reached. � In equilibrium (when Ecell = 0) Q equals K. In this stage, there is no more work, no more chemical reaction. � Eθ describes the equilibrium state when every reactants and products are in standard state (ai = 1).

IV. Summary 69 Methods of utilizing chemical energy: � Chemical reactions (e. g. combustion, neutralization, etc. ) often produce heat (these are called exothermic reactions). In heat engines, chemical energy is transformed to mechanical energy (work) through heat. � In power plants, they generate electrical work from chemical energy through mechanical work. � In a Galvanic cell, chemical energy is directly transferred to electrical work.
- Slides: 69