Chapter 18 The Second Law of Thermodynamics Directions












- Slides: 25

Chapter 18: The Second Law of Thermodynamics Directions of a thermodynamic process Reversible processes: Thermodynamic processes which can be reversed with a small change in conditions. Requires near equilibrium conditions (quasi-equilibrium) that take place slowly (quasi-statically). Irreversible processes generally non-equilibrium processes have a preferred direction (towards increasing “disorder, increasing entropy). 203/4 c 18: 1

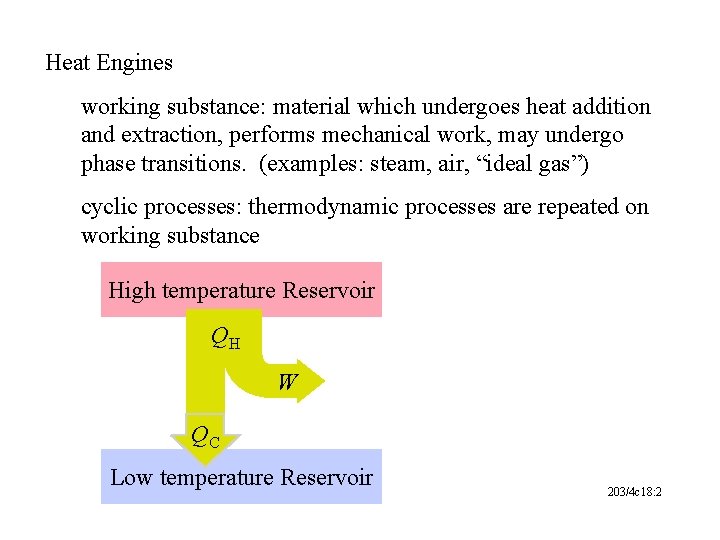
Heat Engines working substance: material which undergoes heat addition and extraction, performs mechanical work, may undergo phase transitions. (examples: steam, air, “ideal gas”) cyclic processes: thermodynamic processes are repeated on working substance High temperature Reservoir QH W QC Low temperature Reservoir 203/4 c 18: 2

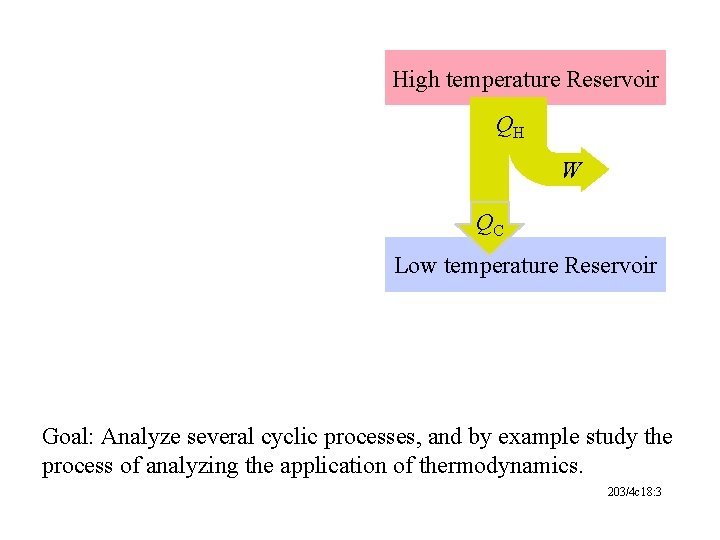
High temperature Reservoir QH W QC Low temperature Reservoir Goal: Analyze several cyclic processes, and by example study the process of analyzing the application of thermodynamics. 203/4 c 18: 3

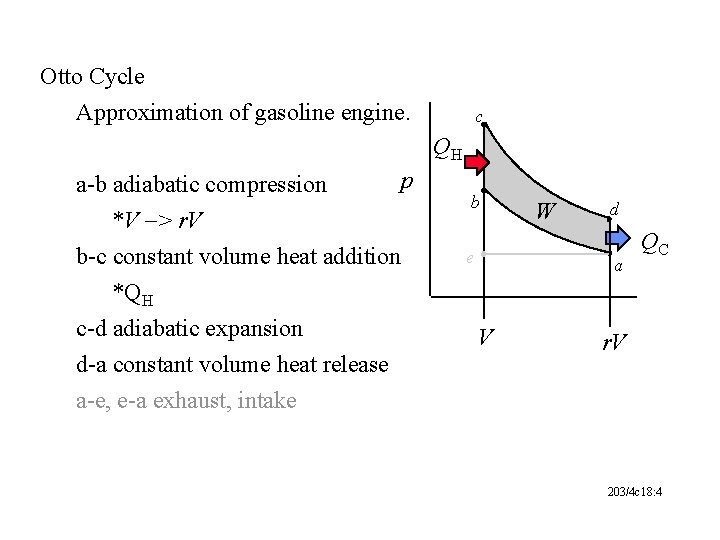
Otto Cycle Approximation of gasoline engine. c QH a-b adiabatic compression *V -> r. V p b-c constant volume heat addition *QH c-d adiabatic expansion d-a constant volume heat release a-e, e-a exhaust, intake b e W d a V QC r. V 203/4 c 18: 4

203/4 c 18: 5

203/4 c 18: 6
203/4 c 18: 7

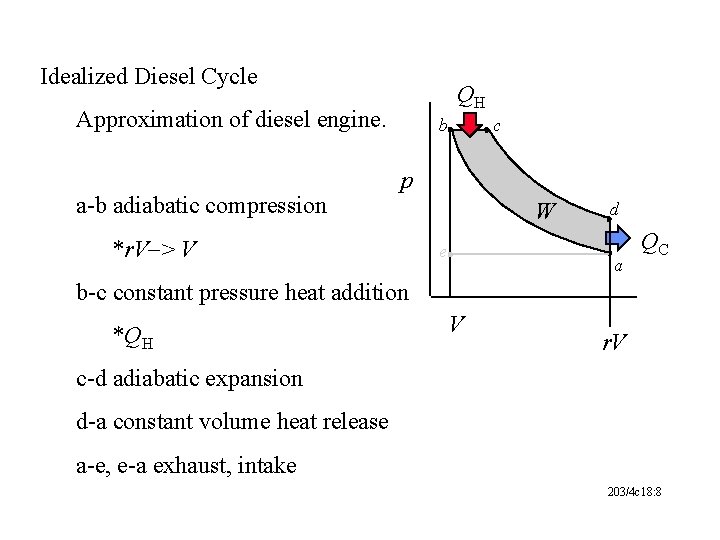
Idealized Diesel Cycle QH Approximation of diesel engine. a-b adiabatic compression b c p *r. V-> V W e d a QC b-c constant pressure heat addition *QH V r. V c-d adiabatic expansion d-a constant volume heat release a-e, e-a exhaust, intake 203/4 c 18: 8

Problem 38: Consider a Diesel cycle that starts with 1. 20 L of air at 300 K and a pressure of 1. 00 E 5 Pa. If Tc is 1200 K, derive an expression for the efficiency in terms of the compression ratio r. 203/4 c 18: 9

Refrigerators Heat Engines run backwards High temperature Reservoir QH W QC Low temperature Reservoir 203/4 c 18: 10

High temperature Reservoir QH W QC Low temperature Reservoir 203/4 c 18: 11

The Second Law of Thermodynamics “It is impossible for any system to undergo a cyclic process in which it absorbs heat from a reservoir and converts the heat completely into mechanical work. ” High temperature Reservoir QH W Low temperature Reservoir 203/4 c 18: 12

The Second Law of Thermodynamics, equivalent form for refrigeration: “It is impossible for any process to have as its sole result the transfer of heat from cooler body to a warmer one. ” High temperature Reservoir QC QC Low temperature Reservoir 203/4 c 18: 13

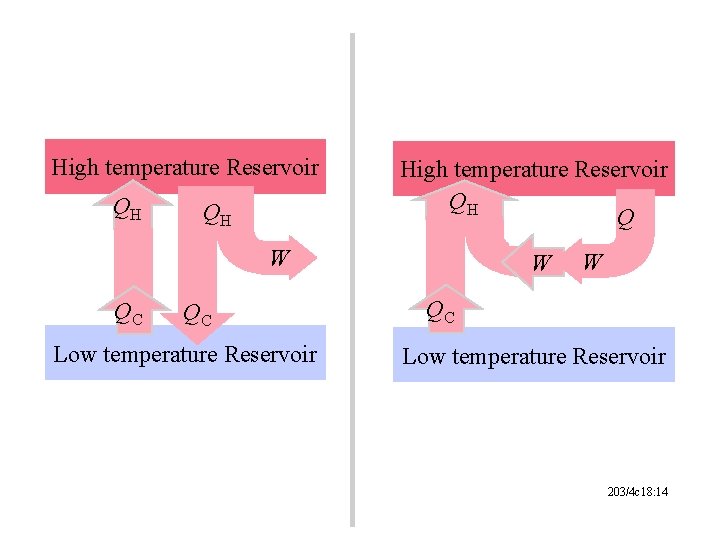
High temperature Reservoir QH QH High temperature Reservoir QH Q W QC QC Low temperature Reservoir W W QC Low temperature Reservoir 203/4 c 18: 14

Entropy S A quantitative measure of disorder. A new thermodynamic variable. (DScyclic = 0) For reversible processes (T may or may not be constant) Adiabatic Process => d. S = 0 203/4 c 18: 15

Melt ice at 0°C: DS = ? (LF = 3. 34 x 105 J/Kg) Heat water from 0°C to 100°C : DS = ? (c = 4190 J/Kg-°C) Boil water at 100°C: DS = ? (LV = 2. 256 x 106 J/Kg) 203/4 c 18: 16

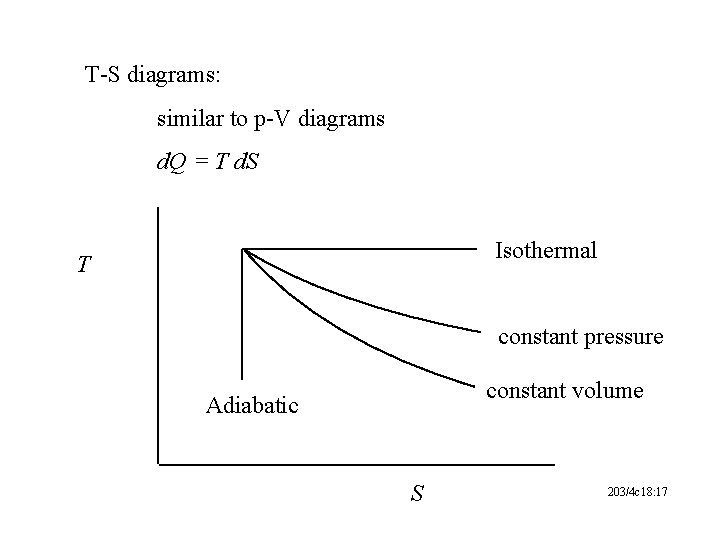
T-S diagrams: similar to p-V diagrams d. Q = T d. S Isothermal T constant pressure constant volume Adiabatic S 203/4 c 18: 17

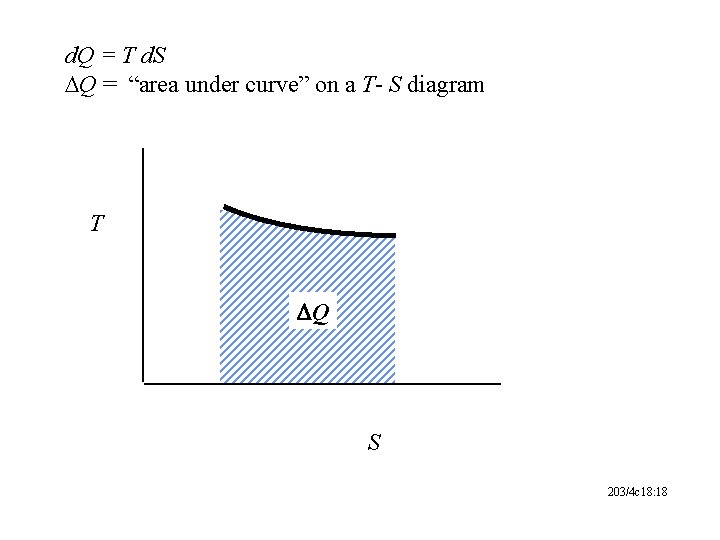
d. Q = T d. S DQ = “area under curve” on a T- S diagram T DQ S 203/4 c 18: 18

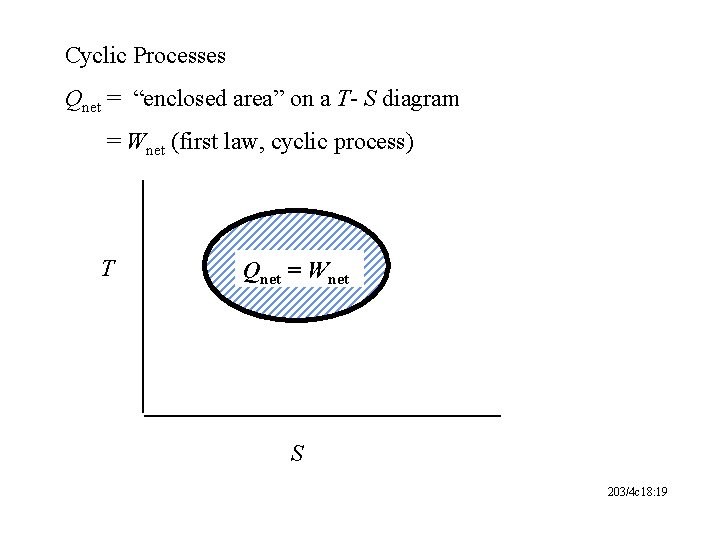
Cyclic Processes Qnet = “enclosed area” on a T- S diagram = Wnet (first law, cyclic process) T Qnet = Wnet S 203/4 c 18: 19

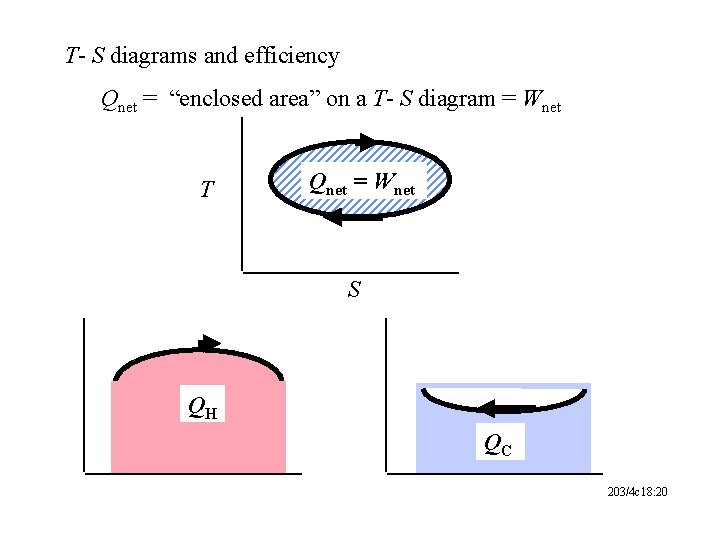
T- S diagrams and efficiency Qnet = “enclosed area” on a T- S diagram = Wnet T Qnet = Wnet S QH QC 203/4 c 18: 20

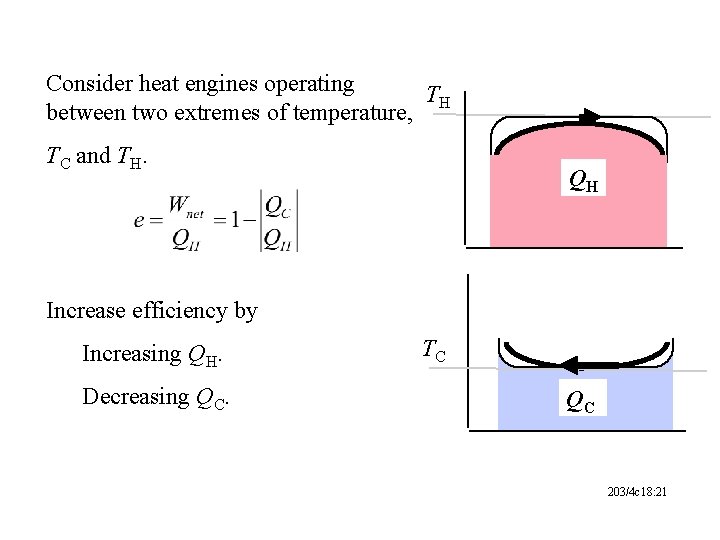
Consider heat engines operating TH between two extremes of temperature, TC and TH. QH Increase efficiency by Increasing QH. Decreasing QC. TC QC 203/4 c 18: 21

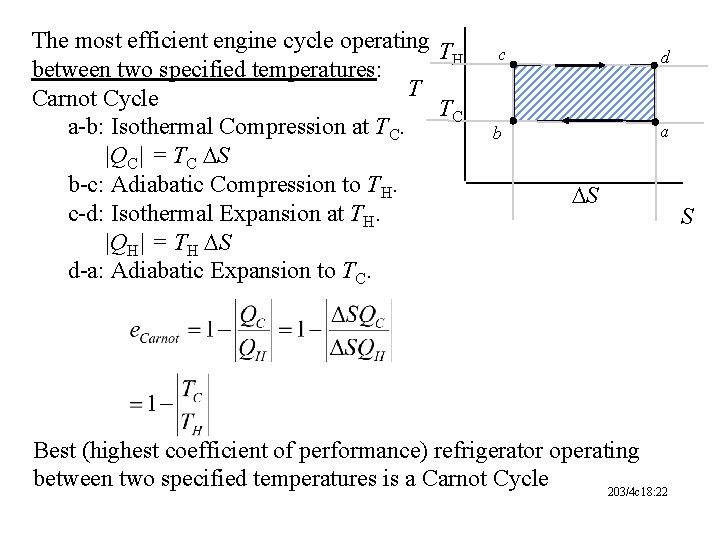
The most efficient engine cycle operating T H between two specified temperatures: T Carnot Cycle TC a-b: Isothermal Compression at TC. |QC| = TC DS b-c: Adiabatic Compression to TH. c-d: Isothermal Expansion at TH. |QH| = TH DS d-a: Adiabatic Expansion to TC. c d b a DS Best (highest coefficient of performance) refrigerator operating between two specified temperatures is a Carnot Cycle 203/4 c 18: 22 S

Example 18 -2: A Carnot engine takes 2000 J of heat from a reservoir at 500 K. does some work, and discards the remainder to a heat reservoir at 300 K. How much work does it do, how much heat is discarded, and what is thermal efficiency? 203/4 c 18: 23

Example 18 -3: . 200 moles of an ideal gas (g = 1. 40) undergoes a Carnot cycle operating between 400 K and 300 K. The initial pressure (just before the isothermal expansion) is 10. 0 E 5 Pa, and during the isothermal expansion, the volume doubles. 203/4 c 18: 24

Best (highest coefficient of performance) refrigerator operating between two specified temperatures is a Carnot Cycle (reversed Carnot Engine) 203/4 c 18: 25