Chapter 11 Chemical Reactions Writing Chemical Equations Word







































- Slides: 41

Chapter 11 Chemical Reactions

Writing Chemical Equations • Word Equations – Names of reactants on the left of an arrow separated by plus signs – Names of products to the right of the arrow separated by plus signs – Ex: flour + water + yeast + salt bread - Ex: carbon + oxygen carbon dioxide

Word equation • Methane + Oxygen Carbon dioxide + Water

Chemical Reaction • When one or more substances (reactants) are changed into one or more new substances (products), a CHEMICAL REACTION has occurred • represented as a chemical equation Reactants products

If you mix two things together how do you know a chemical reaction has occurred?

Evidence a reaction has occurred: • • • Color change Gas released Precipitate formed Temperature change (endo or exothermic) Odor produced Smoke Light Flames p. H change Flammable to nonflammable or vice versa

Chemical Equations • Replaces words with chemical formulas skeleton equations- does not indicate the relative amounts of reactants and products- UNBALANCED –Here is the skeleton equation for rusting: –Fe + O 2 Fe 2 O 3


Symbols for reactions • • • + separates 2 reactants or 2 products (s) solid (l) liquid (g) gas (aq) aqueous – solid dissolved in a liquid usually water (c) crystal precipitate produced gas produced Δ energy is needed yields or produces- separates reactants from products

Symbols for reactions • A catalyst is a substance that speeds up the reaction but is not used up in the reaction • identified above the yield sign KMn. O 4 H 2 O 2 H 2 + O 2

Law of Conservation of Mass • Demo match • Silver nitrate

Law of Conservation of Mass • Mass is never created or destroyed-ALL must be conserved and accounted for during a chemical reaction • The same number of atoms of reactant elements must equal the atoms of product elements • By balancing equations, we satisfy the Law of Conservation of Mass

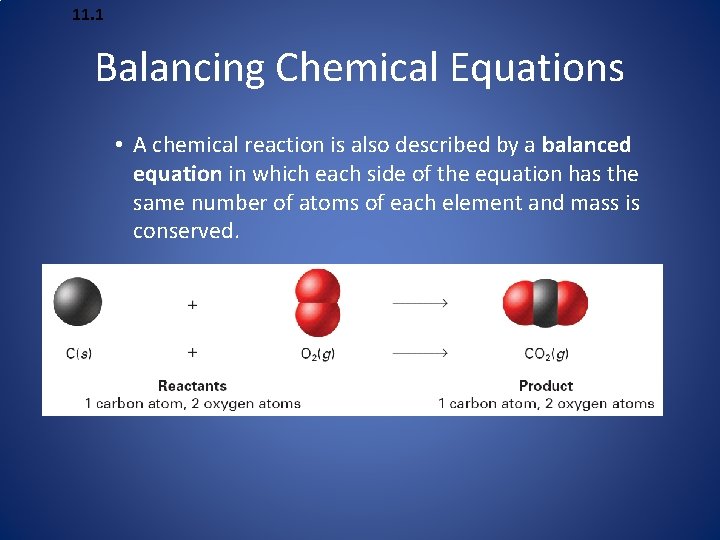
11. 1 Balancing Chemical Equations • A chemical reaction is also described by a balanced equation in which each side of the equation has the same number of atoms of each element and mass is conserved.

11. 1 Balancing Chemical Equations • This is a balanced equation for making a bicycle. The numbers are called coefficients—small whole numbers that are placed in front of the formulas in an equation in order to balance it.

11. 1 Balancing Chemical Equations – To write a balanced chemical equation, first write the skeleton equation. Then use coefficients to balance the equation so that it obeys the law of conservation of mass.

Rules for balancing equations: • Write correct skeleton formula • Determine number of atoms of each element of reactants and products. COUNT POLYATOMIC ION AS A SINGLE UNIT if it appears unchanged on both sides of the equation • Balance elements one at a time by using coefficients. NEVER change subscripts • Begin with the easiest elements first • Check both sides to see if they match • Make sure coefficients are in the lowest possible ratio

Remember diatomics!! • When you write the skeleton equation, remember these elements must be written as 2 atoms when they are not involved in a compound…. • Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2



Types of Reactions

Types of Chemical Reactions • • • Synthesis (combination) Decomposition Single Replacement Double Replacement Combustion

1. Synthesis • Synthesis (composition, combination) reaction • two or more substances react to form a single new substance. A + B AB

1. Synthesis Rules for synthesis reactions • Element + element → binary compound – Metal + nonmetal → ionic compound – Nonmental + nonmetal → covalent compound • Compound + compound → ternary compound ex: CO 2 + H 2 O → H 2 CO 3

Practice • Predict the products. Write and balance the following synthesis reaction equations. • Sodium metal reacts with chlorine gas Na(s) + Cl 2(g) • Solid Magnesium reacts with fluorine gas Mg(s) + F 2(g) • Aluminum metal reacts with fluorine gas Al(s) + F 2(g)

2. Decomposition reaction • a single compound breaks down into two or more simpler products. – AB A + B

2. Decomposition Reactions Rules for Decomposition Reactions: • All binary compounds (ex: Mg. Cl 2) will break down into their elements • All carbonates (CO 32 -) break down into oxide and carbon dioxide • Chlorates (Cl. O 3 -) break down into binary salt and oxygen • Bases (OH group) and compounds with an H and an O will break down into water and an oxide

Practice • Predict the products. Then, write and balance the following decomposition reaction equations: • Solid Lead (IV) oxide decomposes Pb. O 2(s) • Aluminum nitride decomposes Al. N(s)

• http: //educationportal. com/academy/lesson/decompositionand-synthesis-reactions. html#lesson

3. Single Replacement – • one element replaces a second element in a compound. M + AB N + AB MB + A AN + B

Activity Series lists metals in order of decreasing reactivity. As a general rule, more reactive metals replace less reactive metals in a compound Li K Ba Ca Na Mg Al Zn Fe Cd Ni Sn Pb H Cu Hg Ag Au

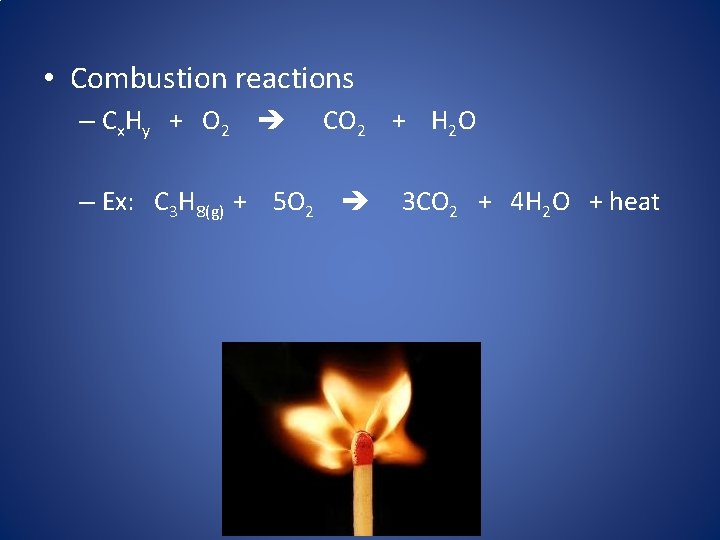
4. Combustion Reactions • Combustion reactions occur when a fuel reacts with oxygen gas, which produces heat! Fuel + O 2 (+ Heat) Product

• Combustion reactions – C x Hy + O 2 CO 2 + H 2 O – Ex: C 3 H 8(g) + 5 O 2 3 CO 2 + 4 H 2 O + heat

Hydrocarbon Combustion Reactions • Products in combustion are ALWAYS carbon dioxide and water. (although incomplete burning does cause some by-products like carbon monoxide) • Combustion is used to heat homes (CH 4)and run automobiles (octane: C 8 H 18)

Carbon Monoxide Effects Edgar Allen Poe’s drooping eyes and mouth are potential signs of CO poisoning.

Combustion • Example • C 5 H 12 + O 2 CO 2 + H 2 O 6 8 5 • Write the products and balance the following combustion reaction: • C 10 H 22 + O 2

5. Double Replacement (precipitation) • Double replacement- also known as precipitation reaction, (and sometimes neutralization reaction) AB + CD AD + CB • The attractive forces between t oppositely charged ions is greater than the forces of attraction between the water molecules and the ions

1. Double Replacement cont Colors of precipitants Precipitant formed • Pb. I 2 • Cd. S • Pb. S • Ag 2 S • Ni(OH)2 • Al(OH)3 • Pb. SO 4 • Ba. SO 4 • Ca 3(PO 4)2 • Ag. Cl Color Bright yellow Dark yellow Black Green White white

Double Replacement Reactions • Think about it like “foil”ing in algebra, first and last ions go together + inside ions go together • Example: Ag. NO 3(aq) + Na. Cl(s) Ag. Cl(s) + Na. NO 3(aq) 2 • Another example: K 2 SO 4(aq) + Ba(NO 3)2(aq) KNO 3(aq) + Ba. SO 4(s)

Solubility rules

Practice • Predict the products. Balance the equation – HCl(aq) + Ag. NO 3(aq) – Ca. Cl 2(aq) + Na 3 PO 4(aq) – Pb(NO 3)2(aq) + Ba. Cl 2(aq) – Fe. Cl 3(aq) + Na. OH(aq) – H 2 SO 4(aq) + Na. OH(aq) – KOH(aq) + Cu. SO 4(aq)

Mixed Practice • State the type, predict the products, and balance the following reactions: 1. Ba. Cl 2 + H 2 SO 4 2. C 6 H 12 + O 2 3. Zn + Cu. SO 4 4. Cs + Br 2 5. Fe. CO 3