Chemical Reactions Writing Equations Balancing Equations Classifying Equations

Chemical Reactions Writing Equations Balancing Equations Classifying Equations Predicting Products

Writing Equations • Chemical equations represent changes that occur in a chemical reaction. • Reactants – starting substances – Always on the left of the arrow • Products – substances formed in a reaction – Always on the right of the arrow • means “yields” “forms” “produces”
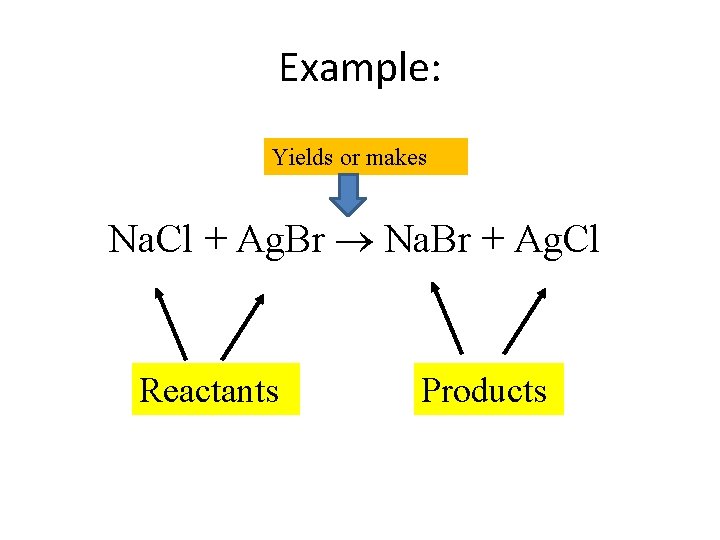
Example: Yields or makes Na. Cl + Ag. Br Na. Br + Ag. Cl Reactants Products

State Symbols • • • cr – crystalline solid s - solid g – gas l – liquid aq – aqueous solution
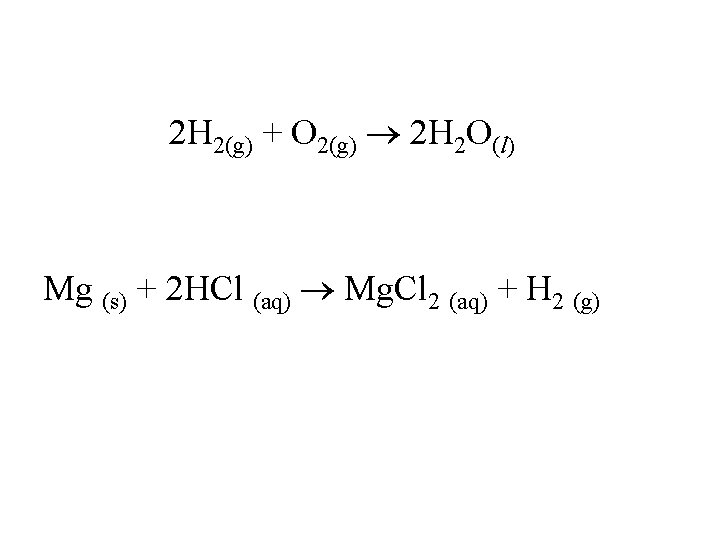
2 H 2(g) + O 2(g) 2 H 2 O(l) Mg (s) + 2 HCl (aq) Mg. Cl 2 (aq) + H 2 (g)

Writing and Balancing Equations • Determine reactants and products. • Write correct formulas for each. • Add coefficients to balance. – the number of atoms of each element must be equal on each side of the equation. – Law of Conservation of Matter YOU CANNOT EVER CHANGE SUBSCRIPTS! TO BALANCE EQUATIONS, YOU CAN ONLY ADD COEFFICIENTS!!!!

When zinc reacts with hydrochloric acid, zinc chloride and hydrogen are formed.
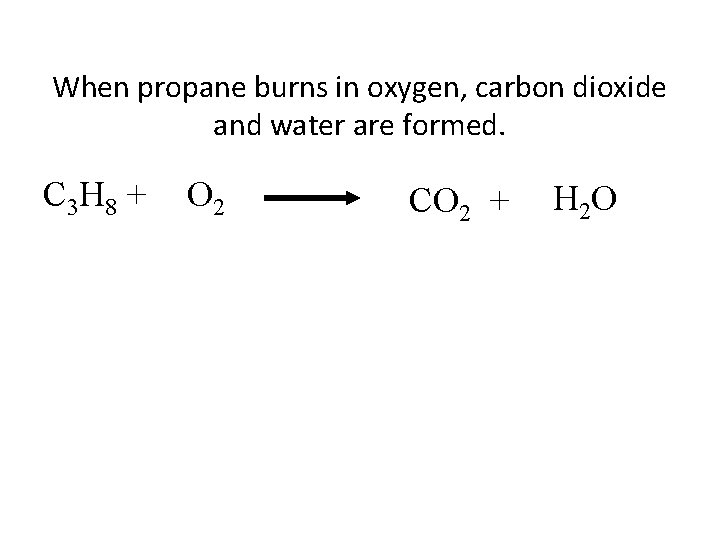
When propane burns in oxygen, carbon dioxide and water are formed. C 3 H 8 + O 2 CO 2 + H 2 O

Classifying Chemical Reactions 1. Single Displacement – One element replaces another in a compound – A + BC AC + B – Element + Compounds Element + Compound – + ions replace + ions, – ions replace – ions
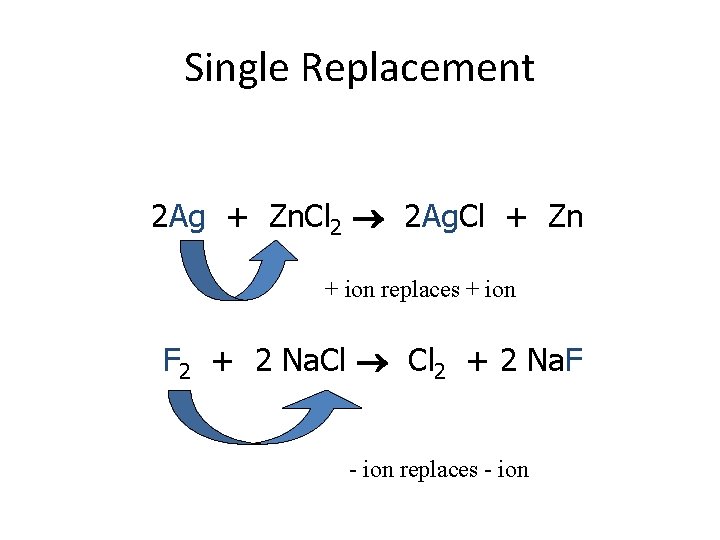
Single Replacement 2 Ag + Zn. Cl 2 2 Ag. Cl + Zn + ion replaces + ion F 2 + 2 Na. Cl 2 + 2 Na. F - ion replaces - ion

2. Double Displacement: Two compounds swap ions • AB + CD AD + CB • Compound + Compound
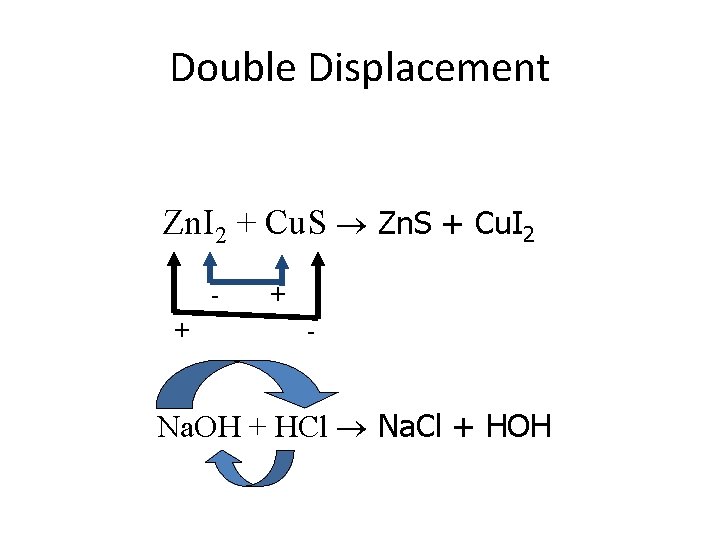
Double Displacement Zn. I 2 + Cu. S Zn. S + Cu. I 2 + + - Na. OH + HCl Na. Cl + HOH

Activity Series of Metals Lithium Potassium Calcium Sodium Magnesium Aluminum Zinc Chromium Iron Nickel Lead Hydrogen Bismuth Copper Mercury Silver Platinum Gold *Metals above other metals will replace them in replacement reactions. 2 Fe (s) + Pb(SO 4)2(aq) + 2 Fe(SO 4)(aq) + Pb(s) *All metals above H will replace H in acids. 2 Al(s) + 6 HCl(aq) → 2 Al. Cl 3(aq) + 3 H 2(g) *All metals above Na will replace H in water. Ca(s) + H 2 O → Ca. O(aq) + H 2(g)

Activity Series for Halogens REMEMBER: Halogens are elements in Group 17 Elements above others will replace them. Cl 2(g) + 2 Na. I(aq) → 2 Na. Cl(aq) + I 2(g)

3. Decomposition • 1 reactant breaks down to form several products (may be elements or compounds) • ABC A + BC • C E + E or E + C or C + C • Example: 2 KCl. O 3 2 KCl + 3 O 2

Decomposition Rules: • Metallic oxides form metals and oxygen. 2 Mg. O 2 Mg + O 2 • Metallic hydroxides form metallic oxides and water. Mg(OH)2 Mg. O + H 2 O • Metallic carbonates form metallic oxides and carbon dioxide. Mg. CO 3 Mg. O + CO 2

• Metallic chlorates form metallic chlorides and oxygen. Mg(Cl. O 3)2 Mg. Cl 2 + 3 O 2 • Oxyacids form nonmetallic oxides and water. H 2 CO 3 CO 2 + H 2 O • Compounds decomposed by electrolysis form elements. 2 Na. Cl 2 Na + Cl 2

4. Synthesis: • 2 or more substances combine to form 1 new substance • A + B AB or AB + C ABC • E+E C or C + E C • Opposite of decomposition meaning more than 1 reactant join to form 1 product
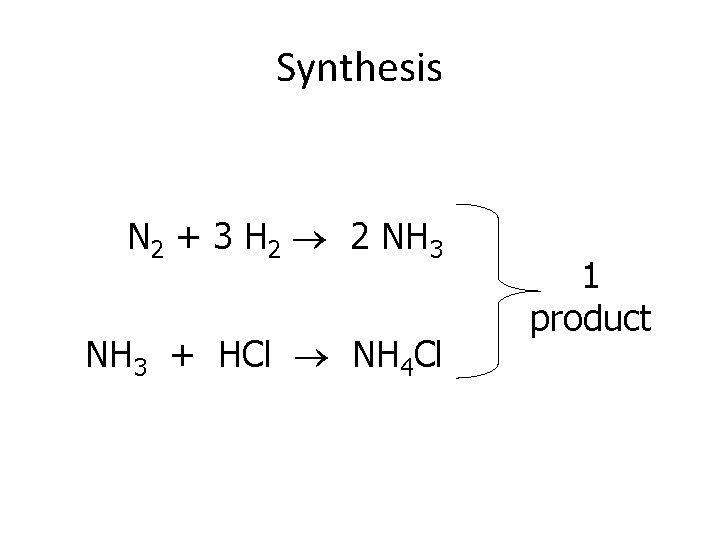
Synthesis N 2 + 3 H 2 2 NH 3 + HCl NH 4 Cl 1 product

5. Combustion • A hydrocarbon reacts with oxygen to form carbon dioxide and water • Cn. H(2 n+2) + O 2 CO 2 + H 2 O

Combustion Let’s balance these! CH 4 + O 2 CO 2 + H 2 O C 8 H 18 + O 2 CO 2 + H 2 O

Balance Equations and ID Rxn Type H 2 S + O 2 → H 2 O + S Cu(NO 3)2 + NH 4(OH) → Cu(OH)2 + NH 4(NO 3) NH 3 + O 2 → N 2 + H 2 O Hg 2 O + O 2 → Hg. O

Balance Equations and ID Rxn Type C 4 H 10 + O 2 → CO 2 + H 2 O H 2 O → H 2 + O 2 Ba(NO 3)2 + Na 2(Cr. O 4) → Ba(Cr. O 4) + Na(NO 3)

Predicting Products aluminum plus hydrochloric acid calcium hydroxide plus nitric acid zinc chloride plus hydrogen sulfide

Predicting Products sodium chlorate (heated) barium nitrate plus sodium chromate
- Slides: 25