Alcohols Foundation in Science Mahsa University College Chem





























- Slides: 44

Alcohols Foundation in Science Mahsa University College Chem 2 Topic: Alcohol Prepared by Mr. Daniel Philip 1

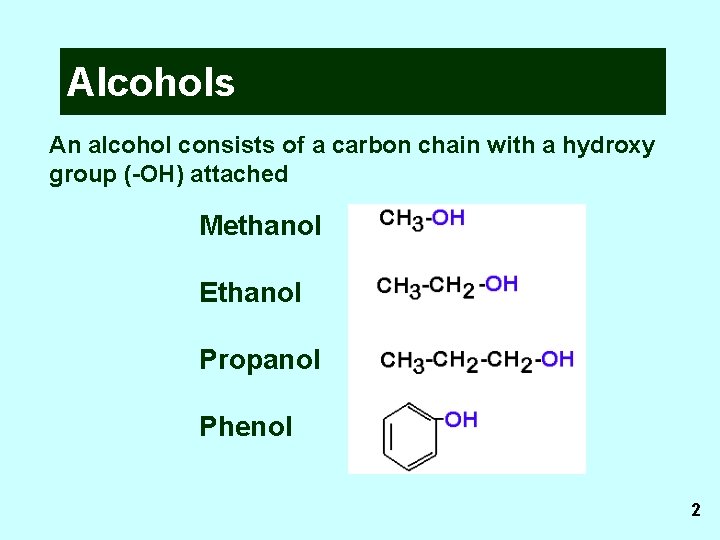
Alcohols An alcohol consists of a carbon chain with a hydroxy group (-OH) attached Methanol Ethanol Propanol Phenol 2

1. Naming and Drawing molecules Nomenclature of Alcohols a) Alcohols are named by dropping the e ending of the parent alkane and replacing with ol. The –OH group can also be named as a substituent using the group name, hydroxy. b) Still another nomenclature involves naming the alkyl group followed by a space and the word alcohol

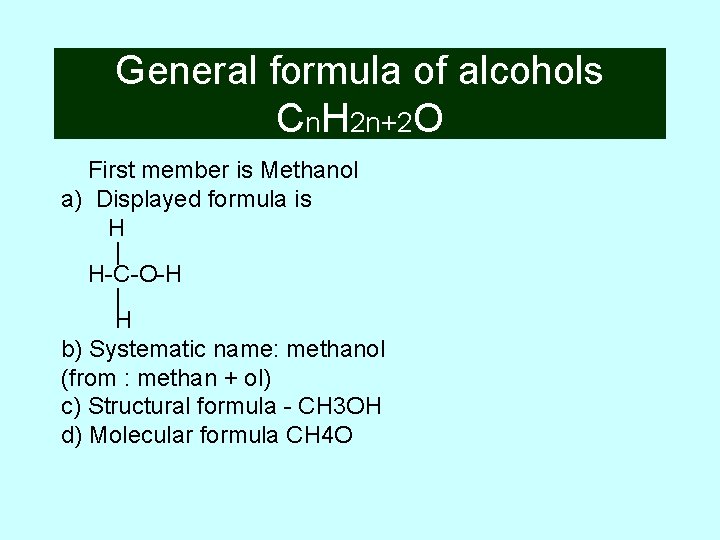
General formula of alcohols Cn. H 2 n+2 O First member is Methanol a) Displayed formula is H | H-C-O-H | H b) Systematic name: methanol (from : methan + ol) c) Structural formula - CH 3 OH d) Molecular formula CH 4 O

Names and Structural formula of common alcohols Examples: CH 3 OH Methanol CH 3 CH 2 OH Ethanol CH 3 CH 2 OH is called propan-1 -ol or 1 -propanol CH 3 CH(OH)CH 3 is called propan-2 -ol or 2 -propanol

if more than one hydroxyl is present, the prefixes di-, tri-, tetra-. . . are used the terminal "e" is retained on the parent alkane name. CH 2 OHCH(OH)CH 2 OH Common name is Glycerol and systematic name is 1, 2, 3 -propanetriol.

Exercise 1 Give the displayed formula, structural formula, molecular formula and empirical formula of the following alcohols a) b) c) d) 1 -butanol 2 -methyl-1 -propanol 2 -methyl-2 -propanol

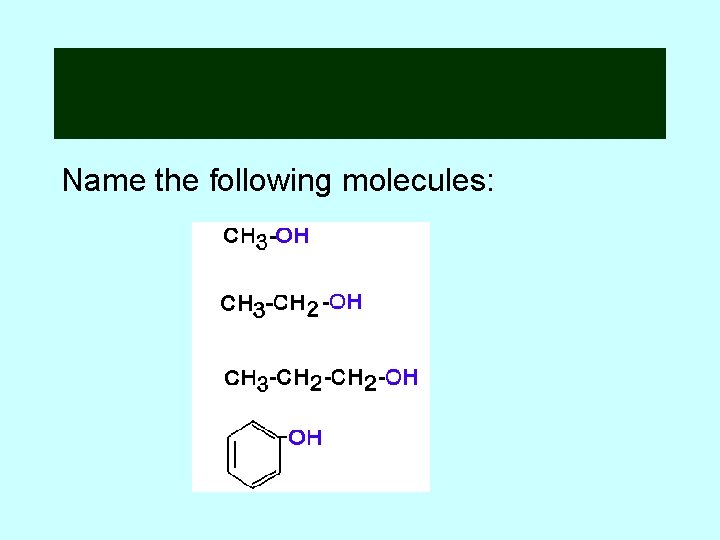
Name the following molecules:

Alcohol Classification Alcohols may be classified as - Primary - Secondary - Tertiary Depending on whether the carbon atom that is attached to the –OH group is surrounded by one, two or three other carbon atoms 9

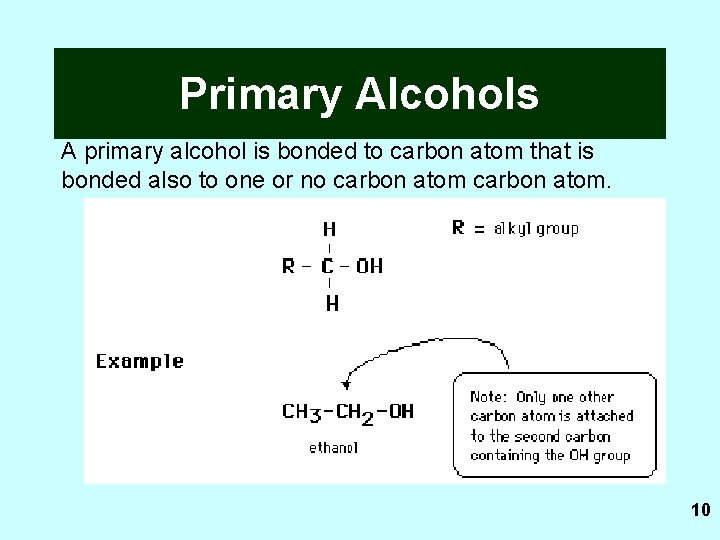
Primary Alcohols A primary alcohol is bonded to carbon atom that is bonded also to one or no carbon atom. 10

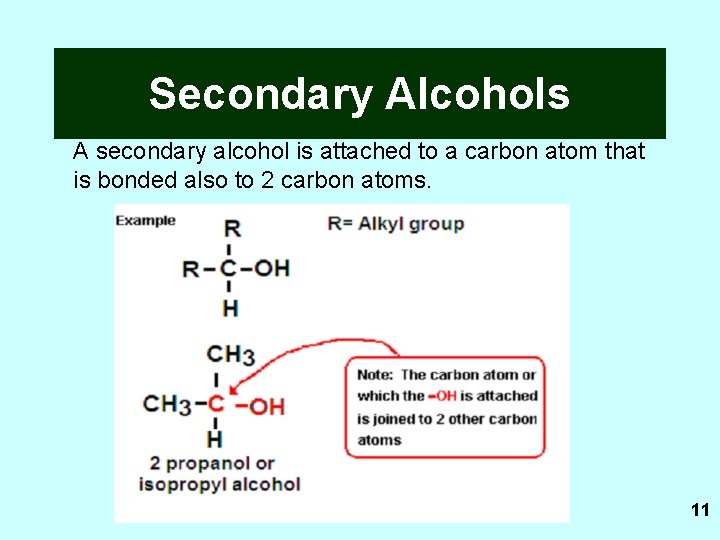
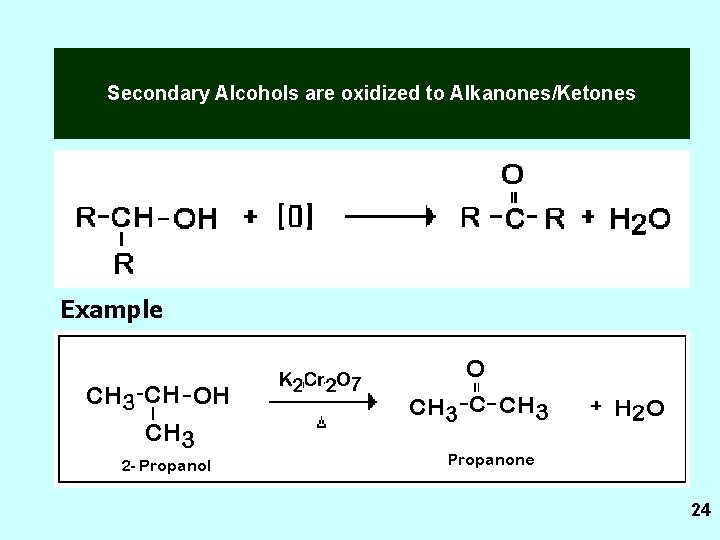
Secondary Alcohols A secondary alcohol is attached to a carbon atom that is bonded also to 2 carbon atoms. 11

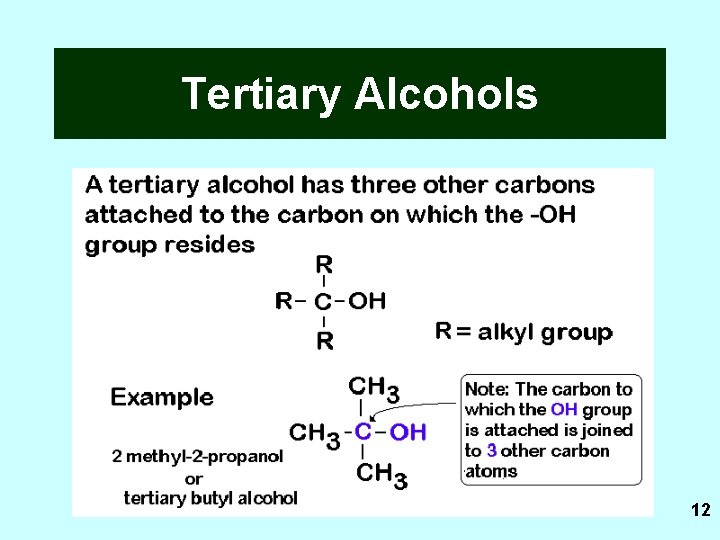
Tertiary Alcohols 12

Polyhydroxy alcohols Alcohols that have more than one OH group are known as polyhydroxyl alcohols. Two examples are shown below 13

Name and classify the following alcohols. • 2 -butanol (Secondary) • 2 -propene-1 -ol (Primary) • 2 -methyl-1 -butanol (Primary) • 2 -methyl-2 -butanol (Tertiary) 14

Properties of Alcohols The physical properties of alcohols are similar to those of both water and hydrocarbons The shorter chain alcohols such as methanol and ethanol are similar to water, in general they -- have higher boiling points than hydrocarbons but lower than water -- dissolve in water to some degree -- are more polar than hydrocarbons but less polar than water 15

Alcohols are polar molecules Reactivity: • The image shows the electrostatic potential for methanol. The more red an area is, the higher the electron density and the more blue an area is, the lower the electron density. • The alcohol O atom are a region of high electron density (red) due to the lone pairs.

3. Physical properties • 1. Boiling point is higher than hydrocarbon. (Due to intermolecular H-bond) • 2. Spirituous odour with burning taste. • 3. Short chain alcohols, up till 4 carbon atoms are soluble in water. Extra info: Phenol (at room temperature) is colourless liquid but tinged with pink oxidation product. It is moderately soluble in water. It is corrosive.

Exercise 1. Explain why ethanol liquid is soluble in water but ethane gas isn’t soluble in water. 4 marks 2. Explain why the boiling point of ethanol is greater than the boiling point of ethane. 4 marks

Reactions of Alcohols undergo several types of reactions including: §Oxidation §Dehydration §Reactions with active metals §Esterification §Substitution 19

4. Chemical reactions of Alcohols 4. 1 Alcohol with alkali metals → alkoxide (strong base!) 2 Na + 2 CH 3 OH → H 2(g) + 2 CH 3 O- + 2 Na+ (sodium methoxide) 4. 2. Alcohol substitution CH 3 CH 2 OH + HCl → CH 3 CH 2 Cl + H 2 O CH 3 CH 2 OH + PCl 5 CH 3 CH 2 Cl + HCl + POCl 3 4. 3. Alcohol dehydration CH 3 -CH 2 -OH → CH 2=CH 2 + H 2 O

Chemical reactions of Alcohols 4. 4 Alcohol oxidation (burning) * primary alcohols → aldehydes * secondary alcohols → ketones * tertiary alcohols → no reaction 4. 5 Alcohol with caboxylic acid for Esterification reaction alcohol + acid → ester + H 20

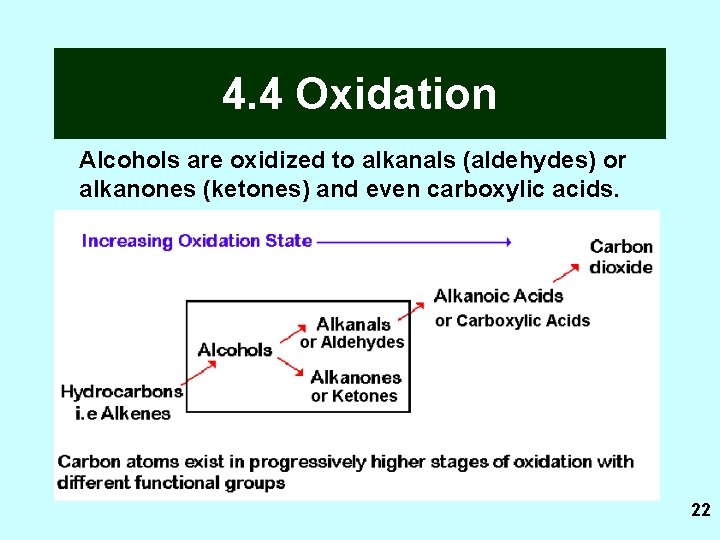
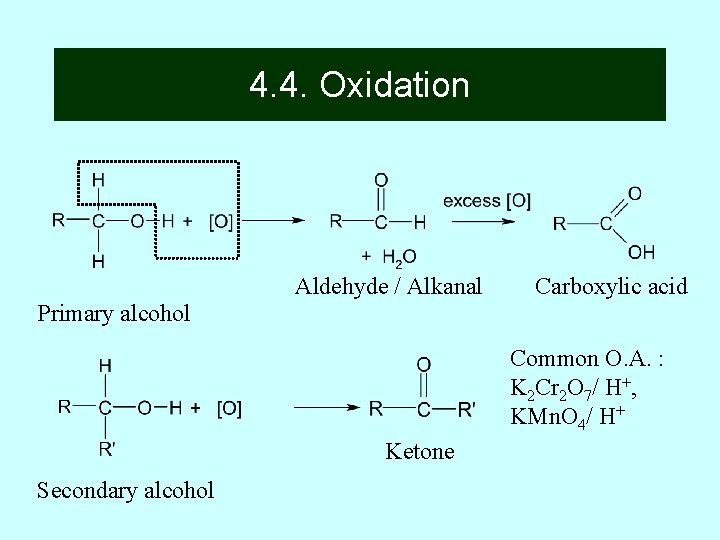
4. 4 Oxidation Alcohols are oxidized to alkanals (aldehydes) or alkanones (ketones) and even carboxylic acids. 22

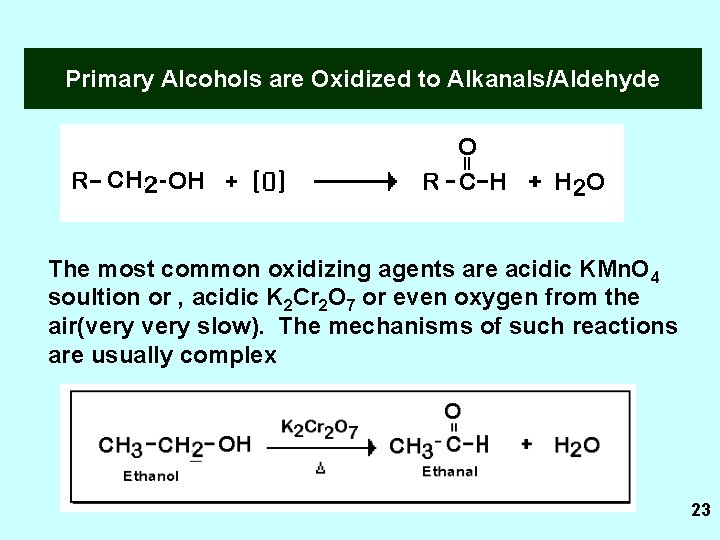
Primary Alcohols are Oxidized to Alkanals/Aldehyde The most common oxidizing agents are acidic KMn. O 4 soultion or , acidic K 2 Cr 2 O 7 or even oxygen from the air(very slow). The mechanisms of such reactions are usually complex 23

Secondary Alcohols are oxidized to Alkanones/Ketones Example 24

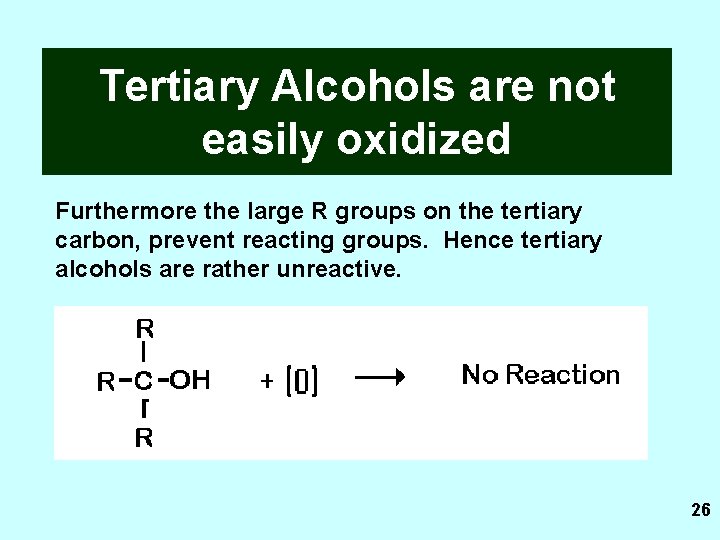
Tertiary Alcohols are not easily oxidized There is no hydrogen attached to the tertiary carbon. It is not possible for a carbonyl group on the tertiary carbon to be oxidixed. 25

Tertiary Alcohols are not easily oxidized Furthermore the large R groups on the tertiary carbon, prevent reacting groups. Hence tertiary alcohols are rather unreactive. 26

4. 4. Oxidation Aldehyde / Alkanal Carboxylic acid Primary alcohol Common O. A. : K 2 Cr 2 O 7/ H+, KMn. O 4/ H+ Ketone Secondary alcohol

Oxidizing agents are acidified KMn. O 4 or acidified K 2 Cr 2 O 7 Primary alcohol with acidic oxidizing agent solution, distilled, gives aldehyde Primary alcohol with acidic oxidizing agent solution, reflux boiling, gives carboxylic acid Secondary alcohol with acidic oxidizing agent solution, reflux boiling, gives ketone

Observation: a) acidified KMn. O 4 solution/acidified manganate(VII) solution : Purple to Colourless or purple is decolourised Or b) acidified K 2 Cr. O 7 solution/acidified dichromate(VI) solution : Orange to Green

Exercise Draw the displayed structure of the organic product formed, if any when the following alcohol was heated with acidified KMn. O 4 solution, reflux (boiling) a) b) c) d) e) f) g) h) Methanol Ethanol 1 -propanol 2 -propanol 1 -butanol 2 -methyl-1 -propanol 2 -methyl-2 -propanol

Draw the displayed structure of the organic product formed, if any when the following alcohol was mixed with acidified KMn. O 4 solution and the product is distilled off a) b) c) d) e) Methanol Ethanol 1 -propanol 1 -butanol 2 -methyl-1 -propanol

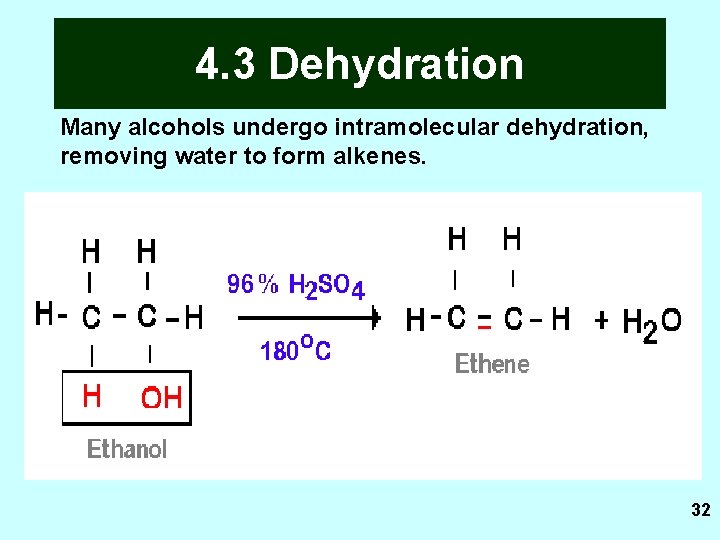
4. 3 Dehydration Many alcohols undergo intramolecular dehydration, removing water to form alkenes. 32

4. 3 Dehydration Definition: removal of water from a molecule. Extra info: H and OH are removed from two adjacent carbons. Reagent and conditions: Dehydrating agents Con H 2 SO 4, heat Con H 3 PO 4, heat Solid Si. O 2, heat Solid Al 2 O 3, heat Solid ceramic or solid pumice, heat

Exercise Write an equation and then give the displayed structure of the organic product formed, if any when the following alcohol was heated with con. H 2 SO 4 and heated(warm/boil or reflux boiling) a) b) c) d) e) f) g) Ethanol 1 -propanol 2 -propanol 1 -butanol 2 -methyl-1 -propanol 2 -methyl-2 -propanol

4. 1 Reactions with Metals • Alcohols are far less acidic than water but they react with highly reactive metals such as potassium and sodium. • The metal replaces the hydrogen atom in the alcohol group forming a salt and hydrogen gas 35

Note that the reaction between Na, sodium solid and alcohol at room temperature releases colourless gas of H 2. This indicates the presence of OH hydroxy group in alcohols.

Quick exercise Write an equation for reaction between Sodium metal at r. t. with a) Methanol b) Ethanol c) 1 -propanol d) 2 -propanol

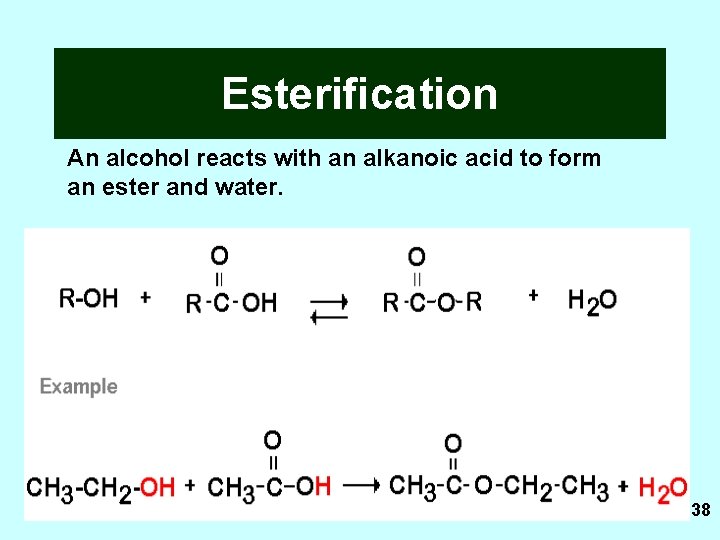
Esterification An alcohol reacts with an alkanoic acid to form an ester and water. 38

Write an equation and then give the displayed structure of the organic product formed, if any when the following alcohol was heated with ethanoic acid with drops of con. H 2 SO 4 and heated(warm/boil or reflux boiling) a) b) c) d) e) f) g) h) Methanol Ethanol 1 -propanol 2 -propanol 1 -butanol 2 -methyl-1 -propanol 2 -methyl-2 -propanol

Exercise Ethanol undergoes the following reactions: Reaction 1: Ethanol Ethene Reaction 2: Ethanol Ethanal Reaction 3: Ethanol Ethanoic acid Reaction 4: Ethanol CH 3 CH 2 ONa Reaction 5: Ethanol CH 3 CH 2 CO 2 CH 3 Reaction 6 : Ethanol to CH 3 CH 2 Cl a) Name the type of organic reaction in reaction labelled as: 1: ………………… 2 : ………………… 3 : ………………… 4 : ………………… 5 : ………………… 6: …………………

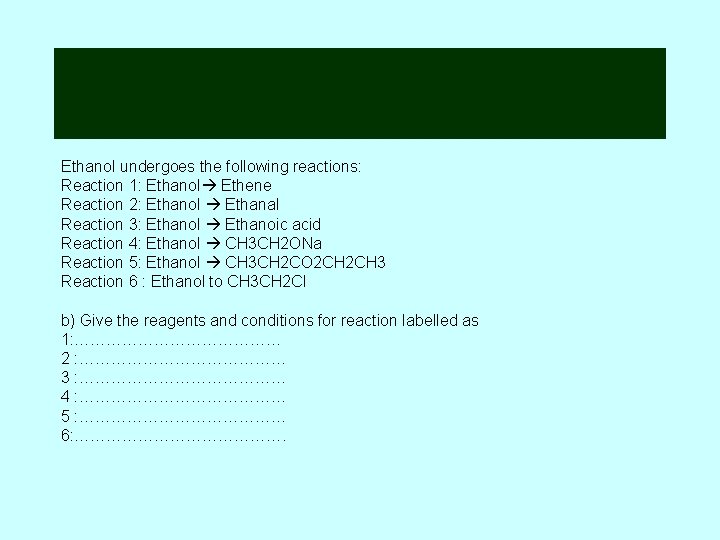
Ethanol undergoes the following reactions: Reaction 1: Ethanol Ethene Reaction 2: Ethanol Ethanal Reaction 3: Ethanol Ethanoic acid Reaction 4: Ethanol CH 3 CH 2 ONa Reaction 5: Ethanol CH 3 CH 2 CO 2 CH 3 Reaction 6 : Ethanol to CH 3 CH 2 Cl b) Give the reagents and conditions for reaction labelled as 1: ………………… 2 : ………………… 3 : ………………… 4 : ………………… 5 : ………………… 6: ………………….

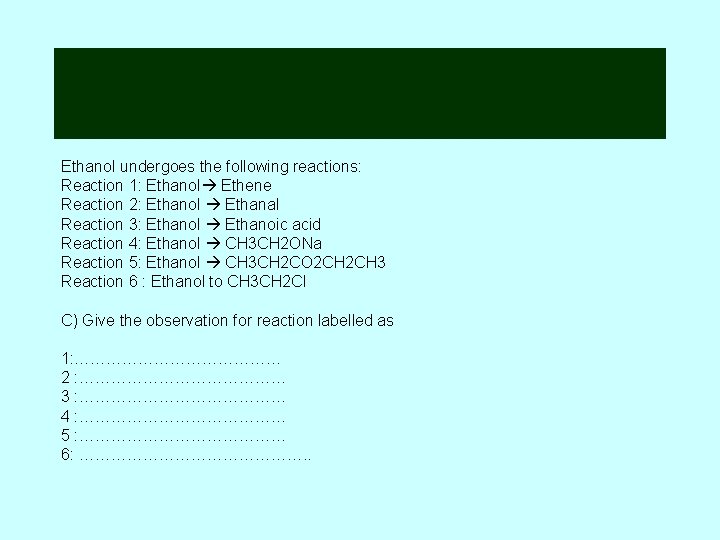
Ethanol undergoes the following reactions: Reaction 1: Ethanol Ethene Reaction 2: Ethanol Ethanal Reaction 3: Ethanol Ethanoic acid Reaction 4: Ethanol CH 3 CH 2 ONa Reaction 5: Ethanol CH 3 CH 2 CO 2 CH 3 Reaction 6 : Ethanol to CH 3 CH 2 Cl C) Give the observation for reaction labelled as 1: ………………… 2 : ………………… 3 : ………………… 4 : ………………… 5 : ………………… 6: …………………. .

Ethanol undergoes the following reactions: Reaction 1: Ethanol Ethene Reaction 2: Ethanol Ethanal Reaction 3: Ethanol Ethanoic acid Reaction 4: Ethanol CH 3 CH 2 ONa Reaction 5: Ethanol CH 3 CH 2 CO 2 CH 3 Reaction 6 : Ethanol to CH 3 CH 2 Cl d) Name the organic products of reaction labelled as 4: ……………. . 5: ……………. . 6: ……………. .

Ethanol undergoes the following reactions: Reaction 1: Ethanol Ethene Reaction 2: Ethanol Ethanal Reaction 3: Ethanol Ethanoic acid Reaction 4: Ethanol CH 3 CH 2 ONa Reaction 5: Ethanol CH 3 CH 2 CO 2 CH 3 Reaction 6 : Ethanol to CH 3 CH 2 Cl e) Give the displayed formula of organic prodct formed from reaction labelled as 1: 2: 3: