Alcohols IUPAC Nomenclature of Alcohols Nomenclature The longest





























- Slides: 31

Alcohols

IUPAC Nomenclature of Alcohols

Nomenclature • The longest C chain with the –OH group attached to it is chosen as the parent group. • The C atoms in the parent chain are numbered so that the C atom attached with the –OH group is given the lowest number possible. • The position of –OH group is indicated by the number of C atom to which it is attached. • The substituents and their positions in the parent chain are numbered from the C with the –OH group. The –OH group is given higher priority compared alkyl/halogen substituents in determining the direction of placements

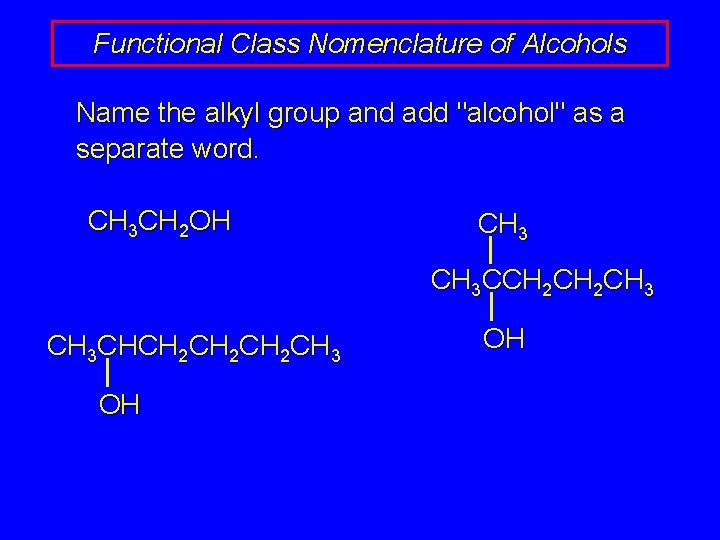
Functional Class Nomenclature of Alcohols Name the alkyl group and add "alcohol" as a separate word. CH 3 CH 2 OH CH 3 CCH 2 CH 3 CHCH 2 CH 2 CH 3 OH OH

Functional Class Nomenclature of Alcohols Name the alkyl group and add "alcohol" as a separate word. CH 3 CH 2 OH Ethyl alcohol CH 3 CHCH 2 CH 2 CH 3 OH 1 -Methylpentyl alcohol CH 3 CCH 2 CH 3 OH 1, 1 -Dimethylbutyl alcohol

Classes of Alcohols

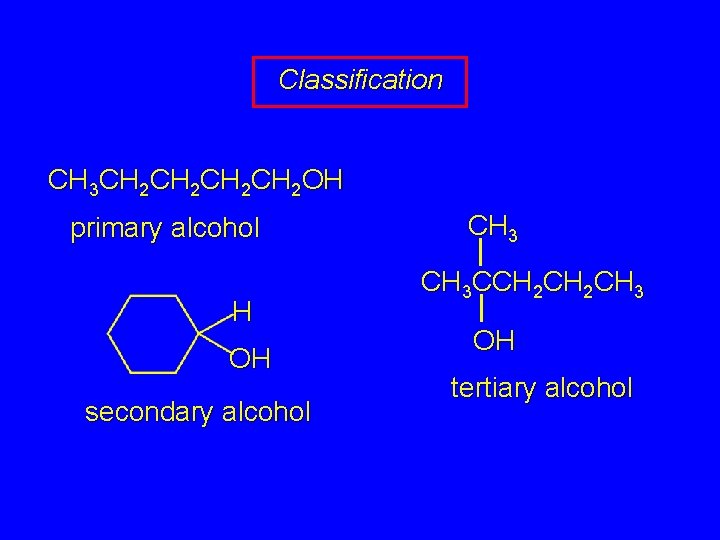
Classification Alcohols and alkyl halides are classified as primary secondary tertiary according to their "degree of substitution. " Degree of substitution is determined by counting the number of carbon atoms directly attached to the carbon that bears the halogen or hydroxyl group.

Classification CH 3 CH 2 CH 2 OH primary alcohol H OH secondary alcohol CH 3 CCH 2 CH 3 OH tertiary alcohol

Number of hydroxyl compound • Hydroxy compound that have only 1 OH group: monohydric alcohols. – Methanol, 2 -propanol • Have 2 –OH group: dihydric alcohols / diols. – 1, 2 -ethanediol, 1, 3 -propanediol. • Have 3 –OH group: trihydric alcohols / triols. – 1, 2, 3 -propanetriol.

Physical Properties of Alcohols

Boiling Points § Higher than other organic compounds with equivalent relative molecular mass. § Formation of hydrogen bond between –OH groups in alcohol molecule. § Boiling point increases as Molecular Mass of alcohol increase since the van der Waals forces of attraction increases with molecular size.

Boiling Points § Boiling point of branched chain alcohol is lower than straight chain, with same Molecular mass. § Small surface area, hence weaker van der Waals forces. § 3° alcohol < 2° alcohol < 1° alcohol boiling point increases

Solubility in Water § Lower members of alcohols are soluble in water; § Formation of H bond between water & alcohol. § Solubility in water decreases significantly: § Size of alkyl group, R § R is non-polar § Bigger influence when number of C (hence size) increases. § Order of solubility in water; § 3° alcohol < 2° alcohol < 1° alcohol solubility increases

• Due to stearic factor as alkyl, -R groups hinder the formation of H-bonds between the –OH groups and water molecules. • Polyhydric alcohols are more soluble in water than monohydric alcohols. • Triol > diol > monohydric alcohols Solubility in water decreases this is because the more –OH groups present in a molecule, the more hydrogen bonds are formed with water.

Reactions of Hydroxyl Compounds

Reactions § Divided into 2 groups: § Type 1: Cleavage of bond between O and H in –OH and H replaced by other groups. § Type 2: Cleavage of bond between C and O in –OH is replaced by other groups through nucleophilic substitution.

Type 1 Reactions • Hydroxyl reacts as acid. • Occurs for both aliphatic and aromatic alcohols • Example reactions: – Formation of alkoxides & phenoxides – Formation of ester – Oxidation of alcohol → carbonyl → carboxylic acid • Depends on class of alcohol

Type 2 Reactions • Hydroxyl react as base. • Occurs in aliphatic alcohols only. • Example reactions: – Rxn with hydrogen halides, phosphorus halide / thionyl chloride. – Dehydration → alkene / ethers.

T 1: Formation of alkoxides & Phenoxides • Alcohol & Phenol react with electropositive metals (Na/K) to form salt known as alkoxides/phenoxides & H 2 gas.

Application • Qualitative test for the presence of –OH group. – H 2 gas released when Na? K react with compound X. X could be alcohol/carboxylic acid • Quantitative test for the number of –OH groups. • To generate H 2 gas that is newly formed to carry out reduction reactions.

T 1: Esterification • Aliphatic alcohols + carboxylic acid → ester + water. • Aromatic compound → no rxn. • Acylation: – Both aliphatic & aromatic + acyl chloride → ester.

T 1: Oxidation • Alcohol can be oxidised to form carbonyl compound and carboxylic acid – depend on class of alcohol. • Involves removing 2 H atoms. • Hot acidified potassium dichromate (VI) / potassium manganate (VII) used. • 1° alcohol → aldehyde → carboxylic acid. • 2° alcohol → ketone: stable toward oxidizing agent. • 3° alcohol → resistance toward oxidation.

T 2: Rxn with PX 5/PX 3/SOX 2/HX • Involve fission of C-O bond in the hydroxy compound and the –OH group is replaced by halogen in nucleophilic substitution. • Application: – Conversion of alcohol → haloalkane • To convert –OH to –X in the preparation of RX from ROH. – Qualitative test for the presence of –OH group. • White fumes of HCl liberated when solid PCl 5 added to compound Y, then –OH is present in comp Y. Y maybe aliphatic hydrocyl, ROH or carboxylic acid, RCOOH. – Quantitative test to determine number of –OH group. • 1 mol of –OH group liberates 1 mol of hydrogen chloride gas.

• Application cont. : – In the rxn of thionyl chloride (sulfur dichloride oxide), SOCl 2 with alcohol, the chloroalkane produce can be easily isolated as the liquid as the rest of the by-products (SO 2 & HCl) are gases. – Alcohol react withconc. HCl / HBr to produce haloalkane. • Lucas Reagent: mixt of conc. HCl & Zn. Cl 2 • Distinguish class of alcohol, rate of reaction is different. – 1° alcohol: react very slowly, no cloudiness at room temperature. – 2° alcohol: react in 1 -5 min (solution turn cloudy after 5 min). – 3° alcohol: react almost instantaneously (immediate cloudiness)

T 2: Dehydration rxn • Two types of dehydration producing diff. product at diff. condition. – Intramolecular elimination of water. – Intermolecular elimination of water. • Intramolecular elimination of water from hydroxyl group & alpha H produce alkene. – α-H: H attached to C adjacent to –OH group. – By refluxing the alcohol with excess conc. H 2 SO 4 / H 3 PO 4 at temp. of 170 -180°C / heated with alumina. • Intermolecular elimination of water from two alcohol molecules to produce ether. – Conc. H 2 SO 4 and excess alcohol refluxed at temp. of 140°C.

Formation of Haloform • All alcohol with structure of RCH(OH)CH 3, where R is H/alkyl/aryl group, will produce haloform when heated with halogen & aqueous alkali. • Haloforms: iodoform, CHI 3 / chloroform, CHCl 3 • Iodoform test: iodomethane formed: yellow precipitate. – Used to identify a methyl group, -CH 3 adjacent to the carbonyl group or hydroxyl group in ethanol (1° alcohol) / 2° alcohol.

Reactions of the Benzene Ring in Phenol

• Since –OH group in ortho- and para- directing, phenol undergo electrophilic substitution reactions in the 2 -(ortho) and 4 -(para) positions of benzene ring under mild conditions. • The electrophilic substitutions ofphenol include: – Halogenation with chlorine / bromine water. – Nitration with conc. Nitric acid – Friedel-Crafts alkylation & acylation.

Preparation of Hydroxyl Compound.

Preparation: Aliphatic Alcohol 1. Hyration of alkenes. 2. Hydrolysis of haloalkanes. 3. Reaction between Grignard reagents & carbonyl compounds. 4. Reduction of carbonyl compound. 5. Fermentation of carbohydrate.

Preparation: Phenol 1. Hydrolysis of chlorobenzene. 2. Cumene process 3. Hydroysis of diazonium salt