A Nitzan Tel Aviv University ELECTRON TRANSFER AND











![I. Inoue et al, Journal of Physiology 541. 3, pp. 769 -778(2002) [Ca+2]=1 x I. Inoue et al, Journal of Physiology 541. 3, pp. 769 -778(2002) [Ca+2]=1 x](https://slidetodoc.com/presentation_image_h/2c87698b30cbab11ff06f7ee63192acf/image-21.jpg)














- Slides: 55

A. Nitzan, Tel Aviv University ELECTRON TRANSFER AND TRANSMISSION IN MOLECULES AND MOLECULAR JUNCTIONS AEC, Grenoble, Sept 2005 Lecture 4

Grenoble Sept 2005 (1) Relaxation and reactions in condensed molecular systems • Kinetic models • Transition state theory • Kramers theory and its extensions • Low, high and intermediate friction regimes • Diffusion controlled reactions Coming March 2006 Chapter 13 -15

Grenoble Sept 2005 (2) Electron transfer processes • Simple models • Marcus theory • The reorganization energy • Adiabatic and non-adiabatic limits • Solvent controlled reactions • Bridge assisted electron transfer Coming March 2006 • Coherent and incoherent transfer Chapter 16 • Electrode processes

Grenoble Sept 2005 (3) Molecular conduction Coming March 2006 Chapter 17 • Simple models for molecular conductions • Factors affecting electron transfer at interfaces • The Landauer formula • Molecular conduction by the Landauer formula • Relationship to electron-transfer rates. • Structure-function effects in molecular conduction • How does the potential drop on a molecule and why this is important • Probing molecules in STM junctions • Electron transfer by hopping

Donor gives an electron and goes from state “a” PROCESSES (reduced) ELECTRODE to state “b” (oxidized). Eb, a=Eb-Ea is the energy of the electron given to the metal Transition rate to a continuum (Golden Rule) D Rate of electron transfer to metal in vacuum Rate of electron transfer to metal in electrolyte solution Reorganization energy here – from donor only (~0. 5 of “regular” value) A EF M

Landauer formula For a single “channel”: (maximum=1) Maximum conductance per channel

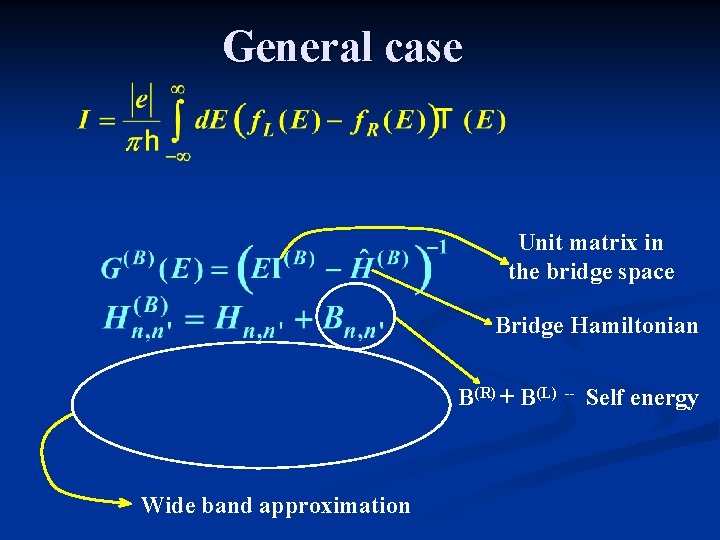
General case Unit matrix in the bridge space Bridge Hamiltonian B(R) + B(L) Wide band approximation -- Self energy

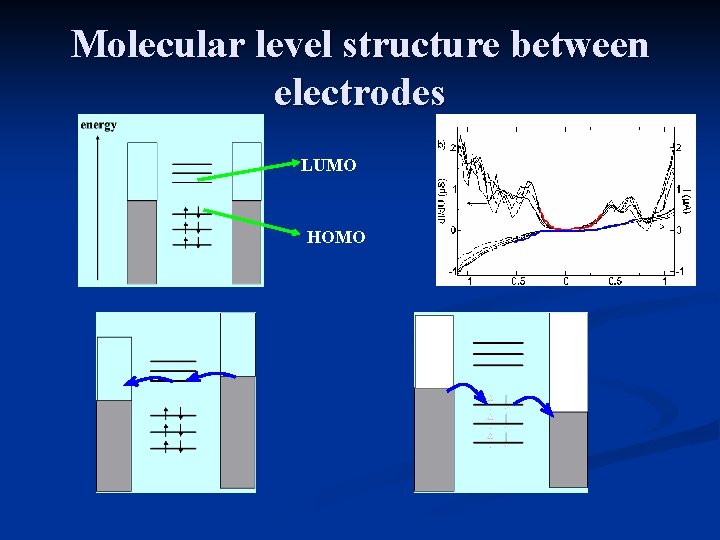
Molecular level structure between electrodes LUMO HOMO

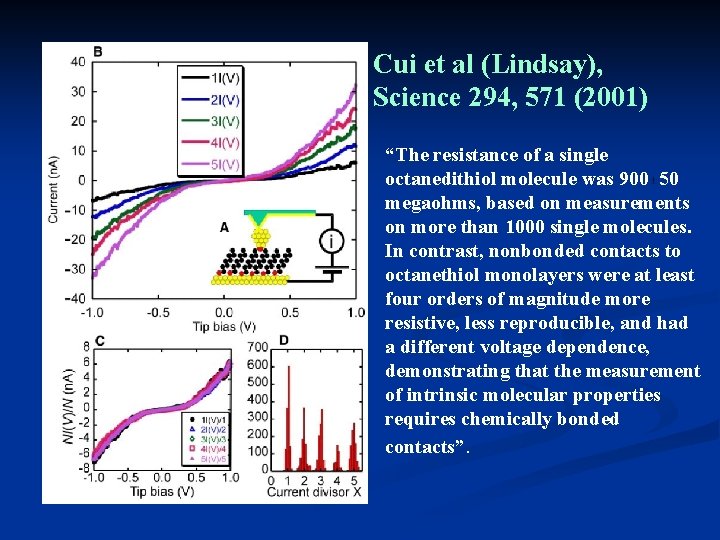
Cui et al (Lindsay), Science 294, 571 (2001) “The resistance of a single octanedithiol molecule was 900 50 megaohms, based on measurements on more than 1000 single molecules. In contrast, nonbonded contacts to octanethiol monolayers were at least four orders of magnitude more resistive, less reproducible, and had a different voltage dependence, demonstrating that the measurement of intrinsic molecular properties requires chemically bonded contacts”.

ET vs Conduction

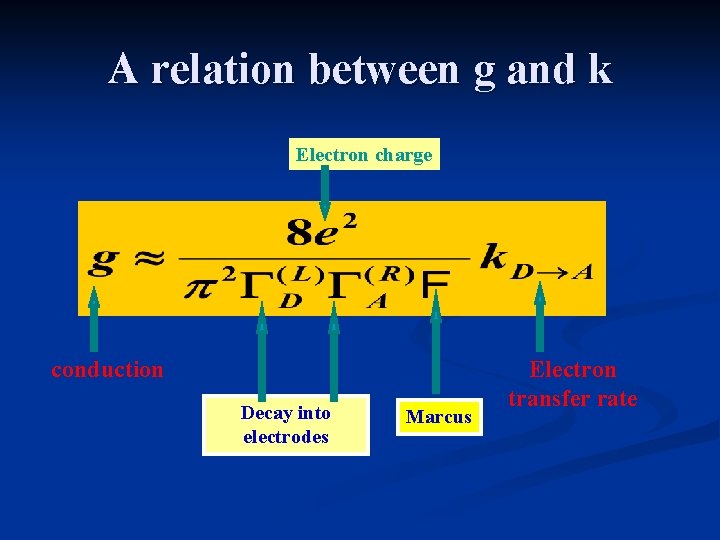
A relation between g and k Electron charge conduction Decay into electrodes Marcus Electron transfer rate

A relation between g and k l 0. 5 e. V

ET rate from steady state hopping

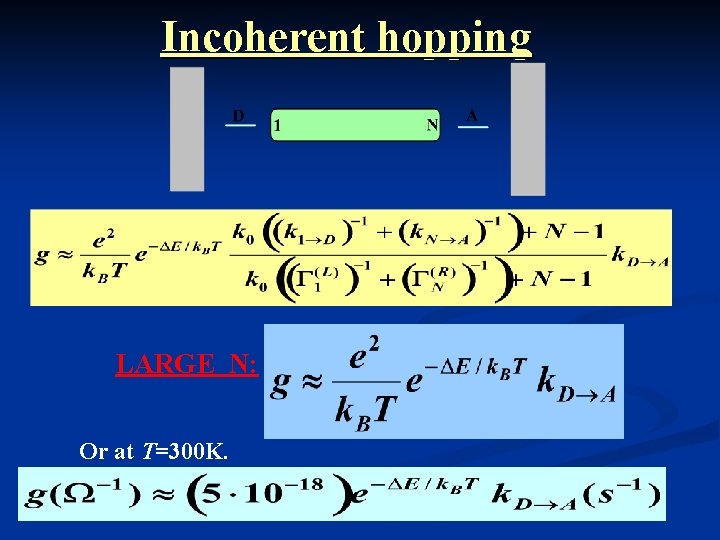
Incoherent hopping LARGE N: Or at T=300 K.

PART D Issues in molecular conductions

Grenoble Sept 2005 (3) Molecular conduction • Structure-function effects in molecular conduction • The role of contacts • How does the potential drop on a molecule and why this is important • Probing molecules in STM junctions • Electron transfer by hopping • Charging • Switching

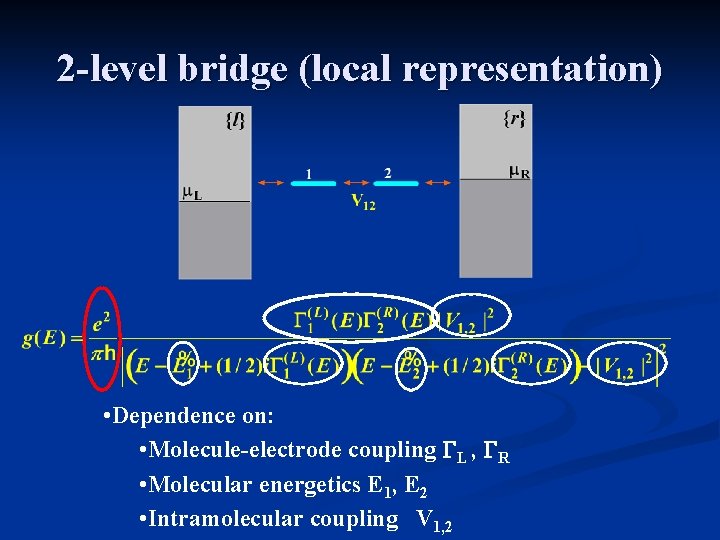
2 -level bridge (local representation) • Dependence on: • Molecule-electrode coupling GL , GR • Molecular energetics E 1, E 2 • Intramolecular coupling V 1, 2

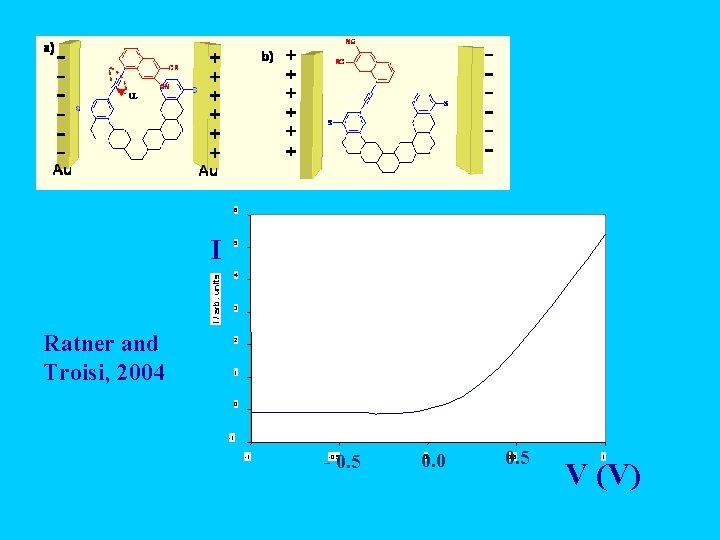
6 I / arb. units I Ratner and Troisi, 2004 5 4 3 2 1 0 -1 -1 - 0. 5 -0. 5 0. 0 0 0. 5 1 V (V)

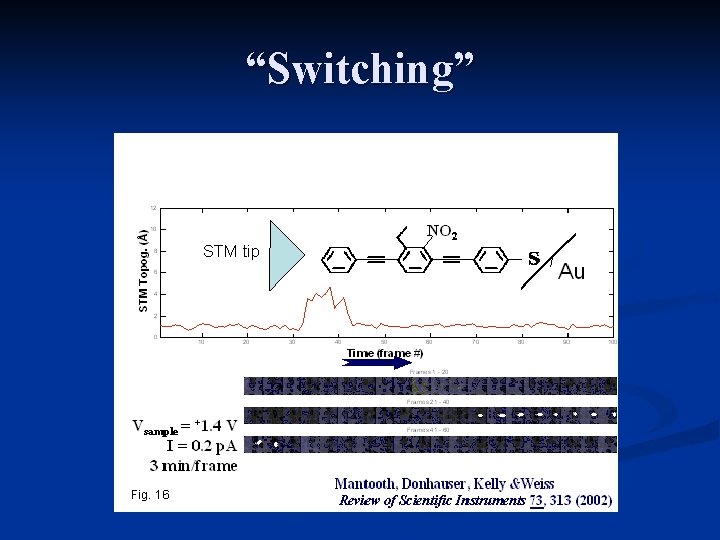
“Switching”

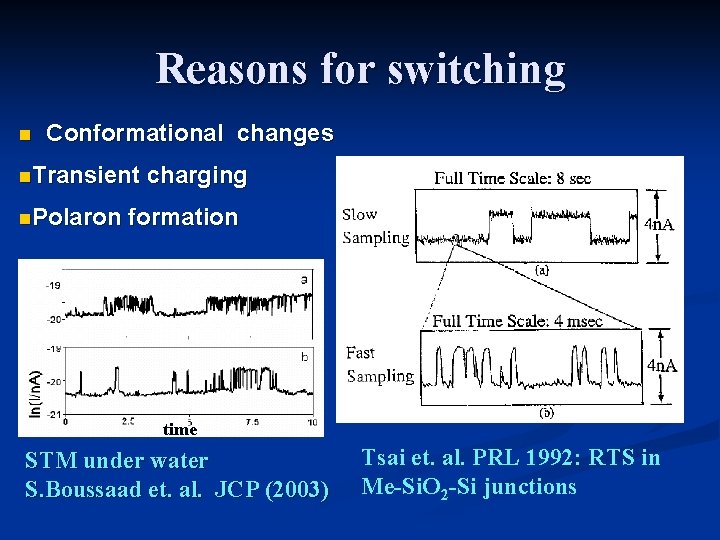
Reasons for switching n Conformational changes n. Transient n. Polaron charging formation time STM under water S. Boussaad et. al. JCP (2003) Tsai et. al. PRL 1992: RTS in Me-Si. O 2 -Si junctions
![I Inoue et al Journal of Physiology 541 3 pp 769 7782002 Ca21 x I. Inoue et al, Journal of Physiology 541. 3, pp. 769 -778(2002) [Ca+2]=1 x](https://slidetodoc.com/presentation_image_h/2c87698b30cbab11ff06f7ee63192acf/image-21.jpg)
I. Inoue et al, Journal of Physiology 541. 3, pp. 769 -778(2002) [Ca+2]=1 x 10 -6 M Single (K+) channel currents from Schwann cells isolated enzymatically from the giant axons of the squids Loligo forbesi, Loligo vulgaris and Loligo bleekeri. The channel conductance was 43. 6 p. S when both internal and external solutions contained 150 m. M K+. Activity was weakly dependent on membrane voltage but sensitive to the internal Ca 2+ concentration.

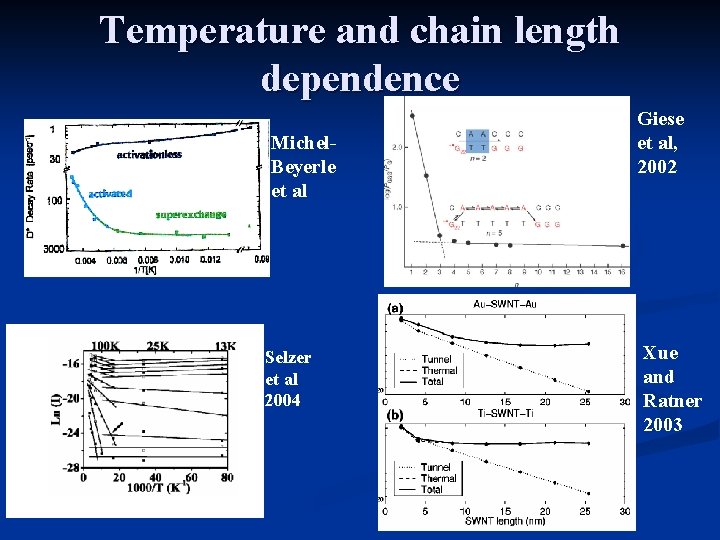
Temperature and chain length dependence Michel. Beyerle et al Selzer et al 2004 Giese et al, 2002 Xue and Ratner 2003

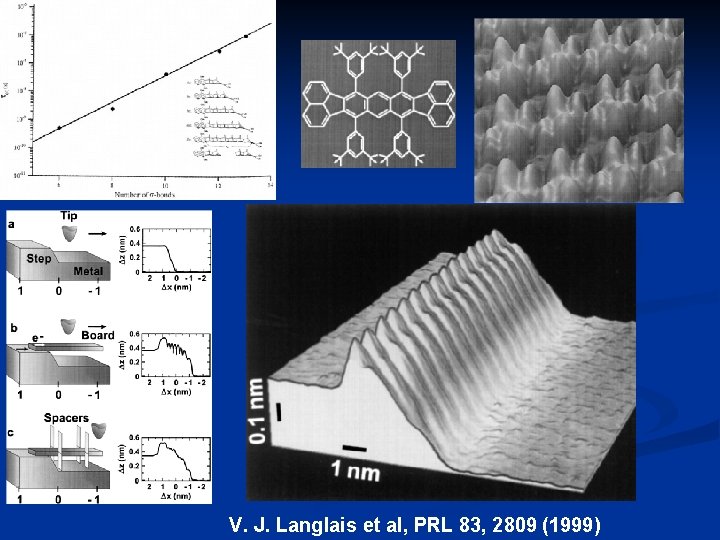
V. J. Langlais et al, PRL 83, 2809 (1999)

Electron transfer in DNA

DNA-news-1

DNA-news-4

DNS-news-3

DNA-news-2

“Prediction is very difficult, Especially of the future ” attributed to Niels Bohr

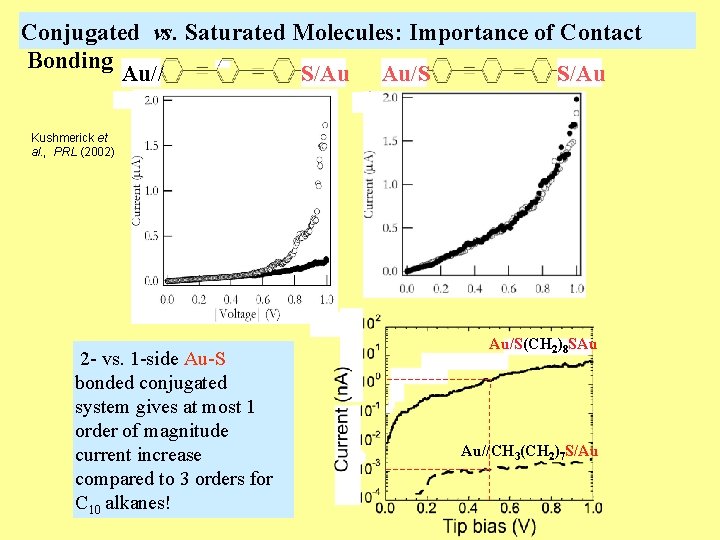
Conjugated vs. Saturated Molecules: Importance of Contact Bonding Au// S/Au Au/S S/Au Kushmerick et al. , PRL (2002) 2 - vs. 1 -side Au-S bonded conjugated system gives at most 1 order of magnitude current increase compared to 3 orders for C 10 alkanes! Au/S(CH 2)8 SAu Au//CH 3(CH 2)7 S/Au

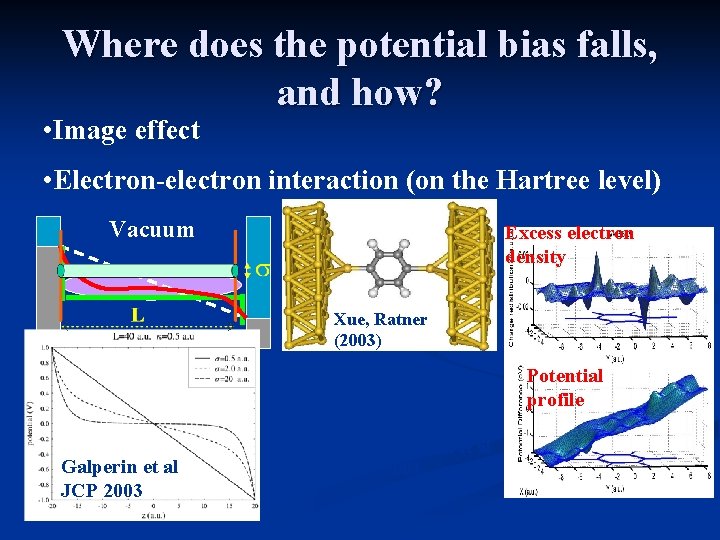
Where does the potential bias falls, and how? • Image effect • Electron-electron interaction (on the Hartree level) Vacuum Excess electron density Xue, Ratner (2003) Potential profile Galperin et al JCP 2003 Galperin et al 2003

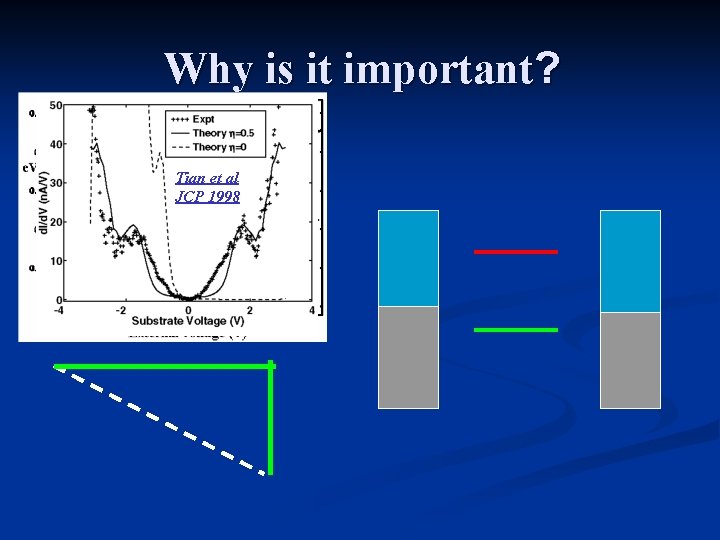
Why is it important? D. Segal, AN, JCP 2002 Heat Release on junction Tian et al JCP 1998

Experiment Theoretical Model

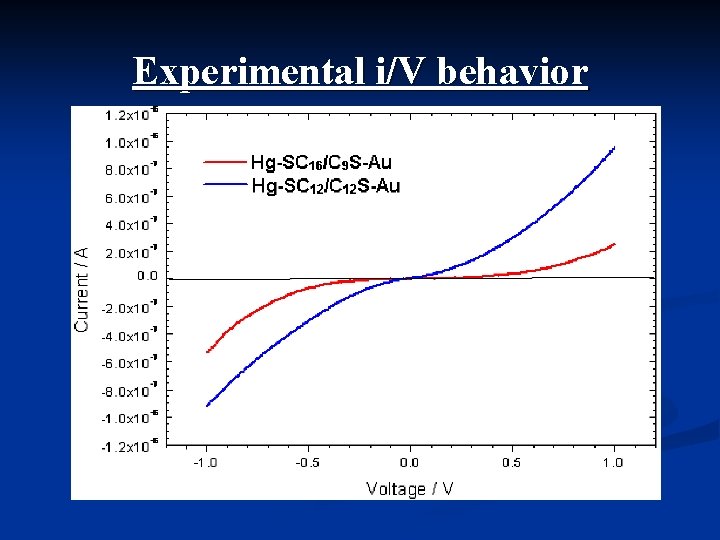
Experimental i/V behavior

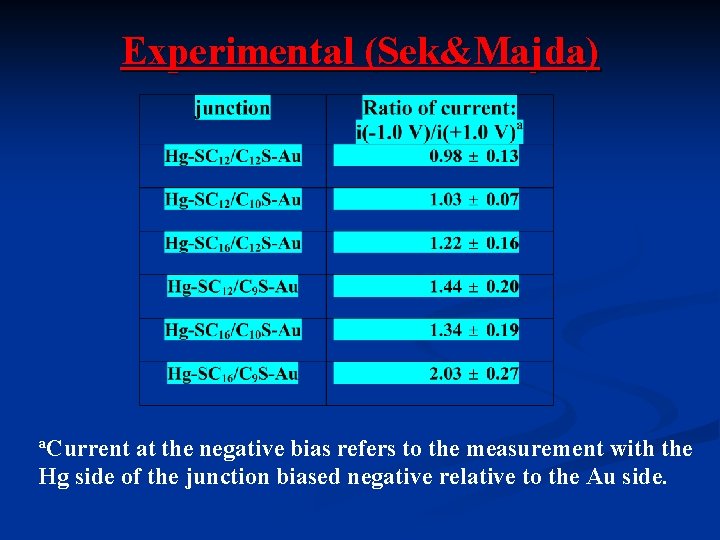
Experimental (Sek&Majda) a. Current at the negative bias refers to the measurement with the Hg side of the junction biased negative relative to the Au side.

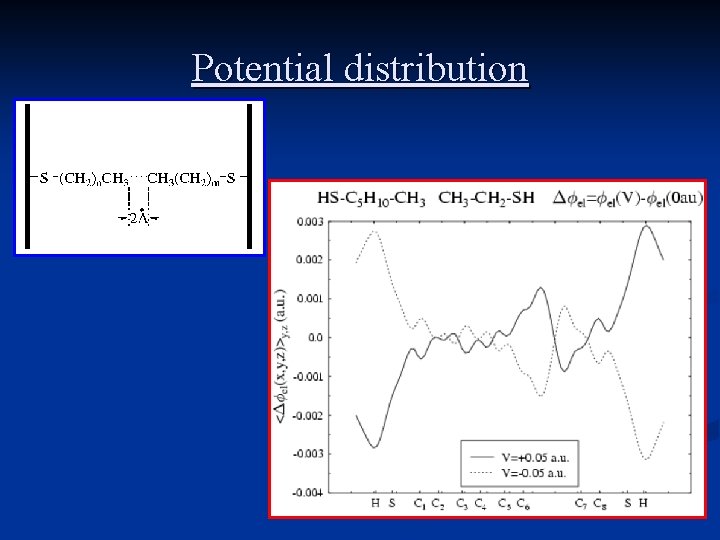
Potential distribution

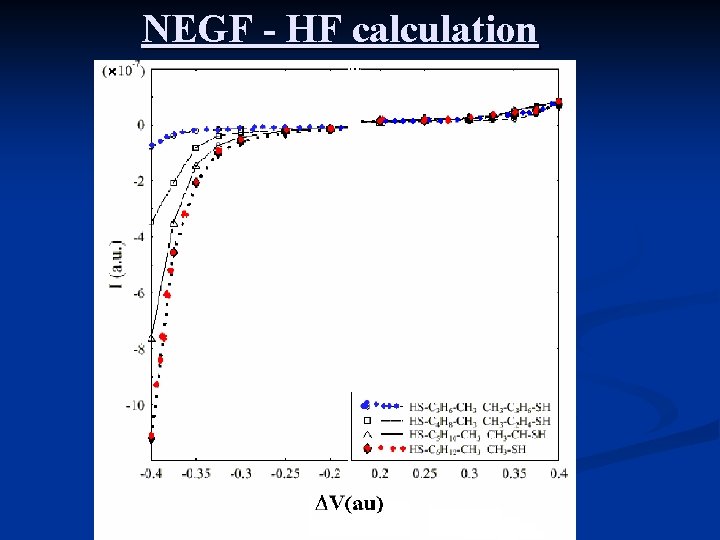
NEGF - HF calculation

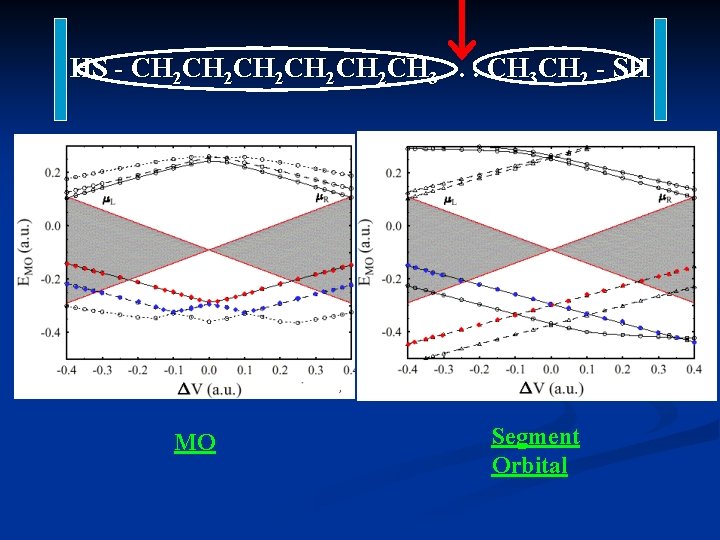
HS - CH 2 CH 2 CH 2 CH 3. . . CH 3 CH 2 - SH MO Segment Orbital

B A A B

TIMESCALE CONSIDERATIONS Does the tunneling electron interact with other degrees of freedom and what are the possible consequences of this interaction? The case of electron tunneling in water

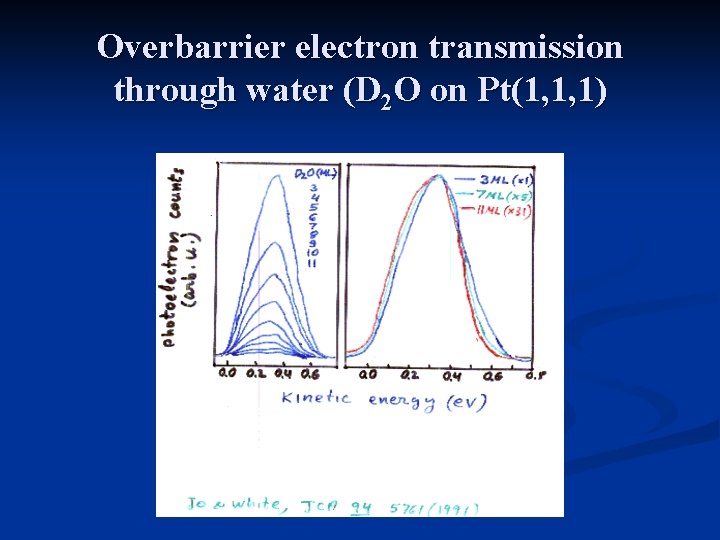
Overbarrier electron transmission through water (D 2 O on Pt(1, 1, 1)

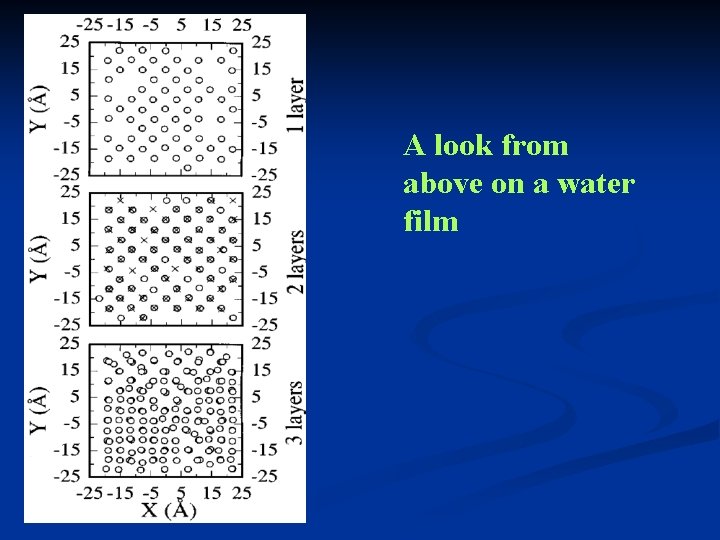
A look from above on a water film

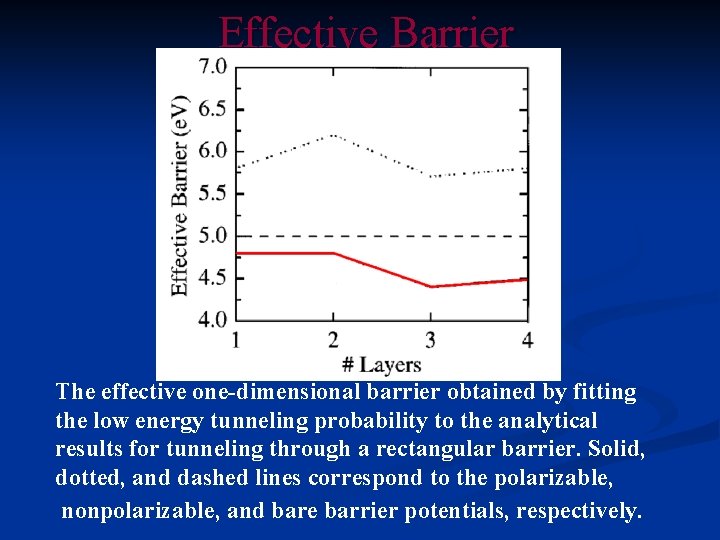
Effective Barrier The effective one-dimensional barrier obtained by fitting the low energy tunneling probability to the analytical results for tunneling through a rectangular barrier. Solid, dotted, and dashed lines correspond to the polarizable, nonpolarizable, and bare barrier potentials, respectively.

The numerical problem (1) Get a potential (2) Electrostatics (3) Generate Water configurations (4) Tunneling calculations (5) Integrate to get current

Potentials for electron transmission through water Water-Water. . . . . RWKM, SPC/E Electron-Water. . . Barnett et al +correction for many body polarizability Water-Wall. . . . Henziker et al (W-Pt), Hautman et al (W-Au) Electron-Wall. . . Square Barrier Earlier studies – Tunneling through static water configurations

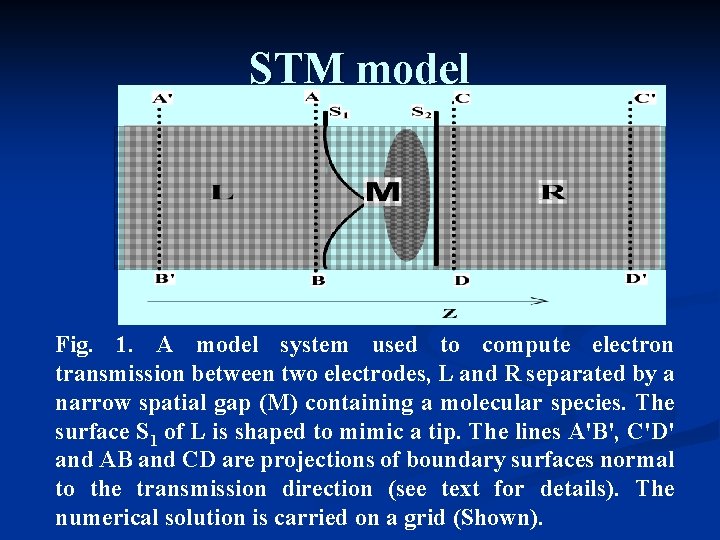
STM model Fig. 1. A model system used to compute electron transmission between two electrodes, L and R separated by a narrow spatial gap (M) containing a molecular species. The surface S 1 of L is shaped to mimic a tip. The lines A'B', C'D' and AB and CD are projections of boundary surfaces normal to the transmission direction (see text for details). The numerical solution is carried on a grid (Shown).

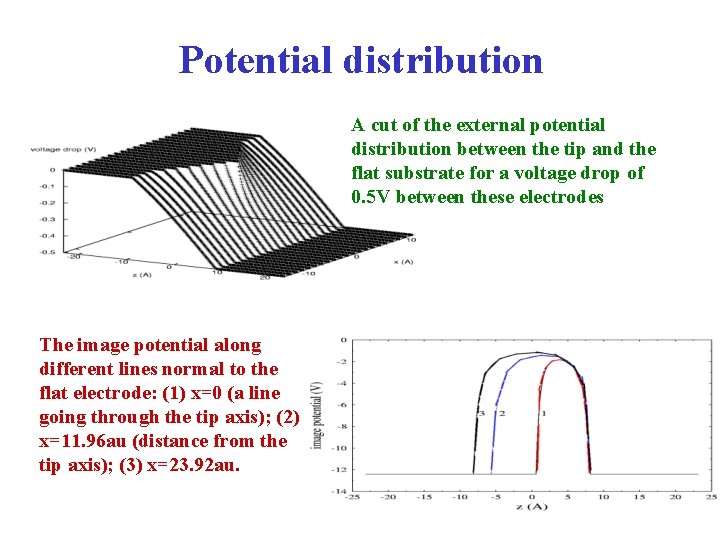
Potential distribution A cut of the external potential distribution between the tip and the flat substrate for a voltage drop of 0. 5 V between these electrodes The image potential along different lines normal to the flat electrode: (1) x=0 (a line going through the tip axis); (2) x=11. 96 au (distance from the tip axis); (3) x=23. 92 au.

MOLECULAR DYNAMICS TO GENERATE WATER CONFIGURATIONS Figure - Ohmine et al

CALCULATION OF TRANSMISSION FACTORS

CALCULATION OF TRANSMISSION FACTORS Absorbing boundary conditions Green's function method: Replace by i (r), smoothly rising towards edges of M system, provided LM and MR boundaries are set far enough

The self energy - 2 For nearest neighbor coupling:

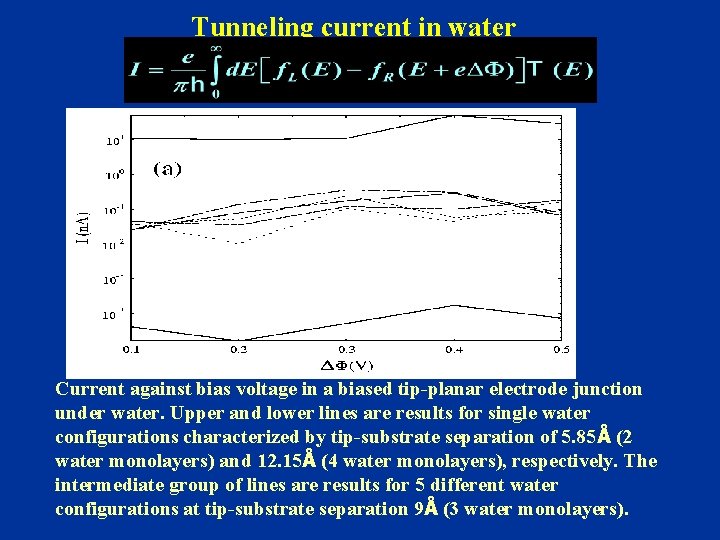
Tunneling current in water Current against bias voltage in a biased tip-planar electrode junction under water. Upper and lower lines are results for single water configurations characterized by tip-substrate separation of 5. 85Å (2 water monolayers) and 12. 15Å (4 water monolayers), respectively. The intermediate group of lines are results for 5 different water configurations at tip-substrate separation 9Å (3 water monolayers).

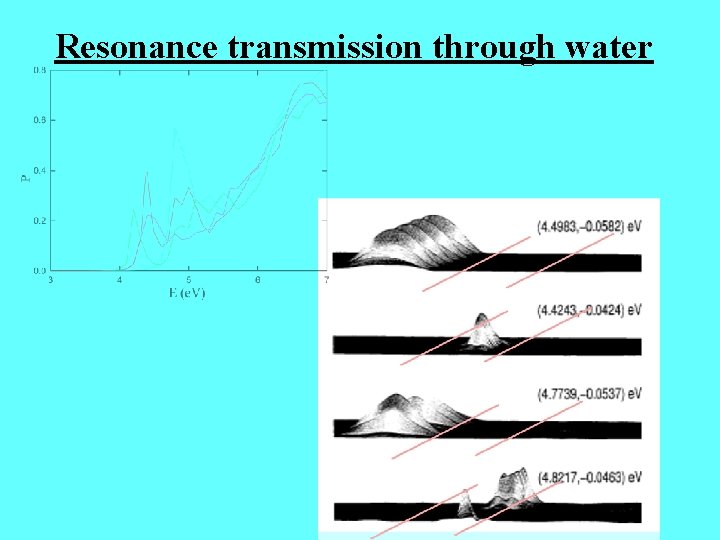
Resonance transmission through water

Tunneling supporting structures in water

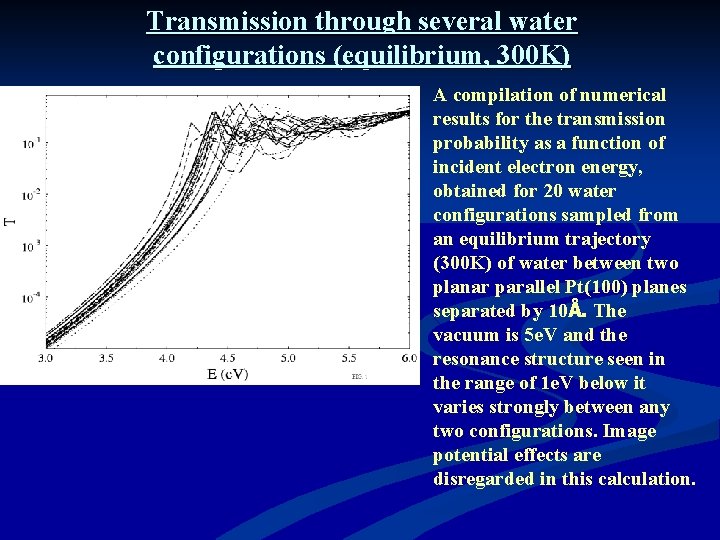
Transmission through several water configurations (equilibrium, 300 K) A compilation of numerical results for the transmission probability as a function of incident electron energy, obtained for 20 water configurations sampled from an equilibrium trajectory (300 K) of water between two planar parallel Pt(100) planes separated by 10Å. The vacuum is 5 e. V and the resonance structure seen in the range of 1 e. V below it varies strongly between any two configurations. Image potential effects are disregarded in this calculation.