A Nitzan Tel Aviv University The Abdus Salam

A. Nitzan, Tel Aviv University The Abdus Salam African College on Science at the Nanoscale Cape Town, November 2007 1. Relaxation, reactions and electron transfer in condensed molecular systems 2. Fundamentals of molecular conduction 3. Inelastic effects in electron transfer and molecular conduction

Molecular conduction

Molecular Rectifiers Arieh Aviram and Mark A. Ratner IBM Thomas J. Watson Research Center, Yorktown Heights, New York 10598, USA Department of Chemistry, New York University, New York 10003, USA Received 10 June 1974 Abstract The construction of a very simple electronic device, a rectifier, based on the use of a single organic molecule is discussed. The molecular rectifier consists of a donor pi system and an acceptor pi system, separated by a sigmabonded (methylene) tunnelling bridge. The response of such a molecule to an applied field is calculated, and rectifier properties indeed appear.

Xe on Ni(110)
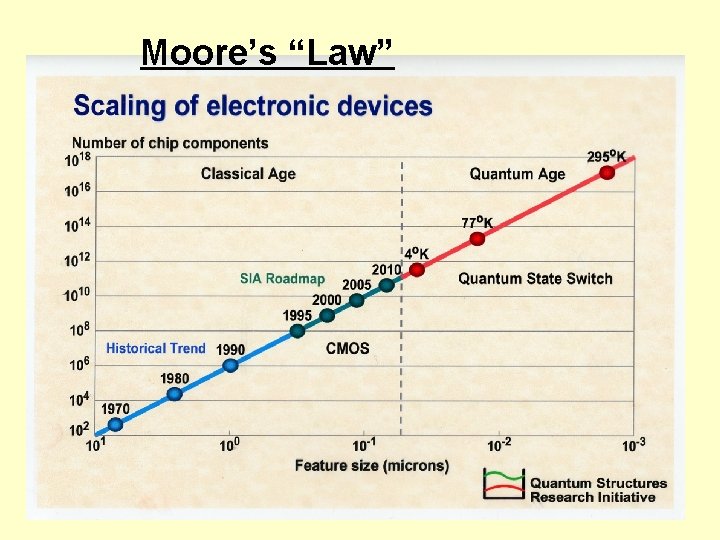
Moore’s “Law”

• Fabrication • Stability • Characterization • Funcionality • Control Cornell group

FABRICATION Adsorbed molecule addressed by STM tip Molecule between two electrodes: Break junction: Self-assembled monolayers Pt/Ir Tip ~1 -2 nm 1 nm SAM Au(111) Van Ruitenbeek, Wittenberg Lectures 2004 Dorogi et al. PRB 52 (95) @ Purdue Nanopore C 60 on gold STM tip Joachim et al. PRL 74 (95) Molecule lying on a surface Nanotube on Au Au Reed et al. APL 71 (97) Lieber et al. Nature 391 (98)
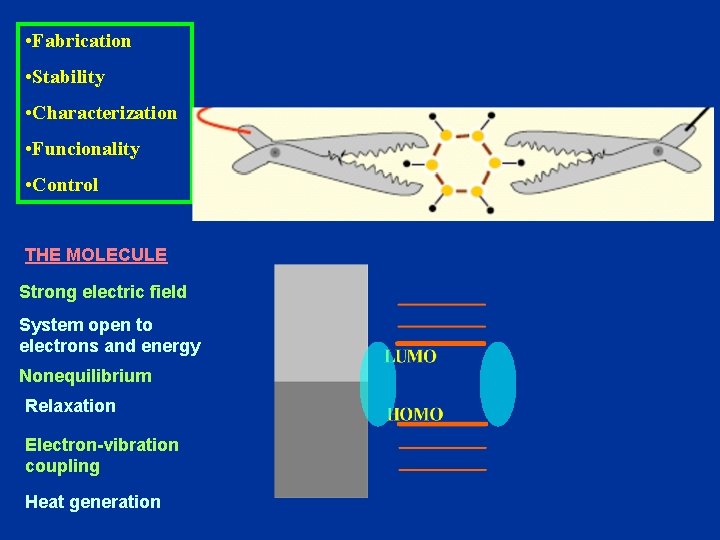
• Fabrication • Stability • Characterization • Funcionality • Control THE MOLECULE Strong electric field System open to electrons and energy Nonequilibrium Relaxation Electron-vibration coupling Heat generation

Cape Town, November 2006 (2) conduction (1 a) (3) Inelastic Relaxation andinreactions molecular in (1 b)Molecular Electroneffects transfer AN, Oxford University Press, 2006 Chapter 16 Chapter 13 -15 17 • Simple models for molecular conductions condensed conduction molecular systems processes • Factors affecting electron transfer at • • interfaces Timescales Dependence on nuclear Simple models • • The configurations Relaxation Landauer formula Marcus theory • • Molecular Solvation Electron-vibration coupling conduction by the Landauer The reorganization energy formula • • Timescales Activated processes Adiabatic rate and non-adiabatic • • Relationship to electron-transfer rates. Low, limits Coherent highand incoherent intermediate transport • Structure-function effects in molecular • • Heating friction regimes Solvent controlled reactions conduction • • • How Transition Current induced state theory nuclear changes Bridge electron doesassisted the potential drop transfer on a • • molecule Diffusion Heat conduction reactions andcontrolled whyincoherent this is important Coherent and • Probing Inelastic tunneling spectroscopy molecules in STM junctions transfer • • Electron transfer by hopping Electrode processes

1 A Relaxation and reactions in molecular systems

The importance of timescales

Molecular processes in condensed phases and interfaces Molecular timescales • Diffusion Electronic 10 -16 -10 -15 s • Relaxation Vibraional period 10 -14 s • Solvation Vibrational xxxxrelaxation 1 -10 -12 s • Nuclear rerrangement • Charge transfer (electron and xxxxxxxxproton) • Solvent: an active spectator – energy, friction, solvation Diffusion D~10 -5 cm 2/s 10 nm 10 -7 - 10 -8 s Chemical reactions xxxxx 1012 -10 -12 s Rotational period 10 -12 s Collision times 10 -12 s
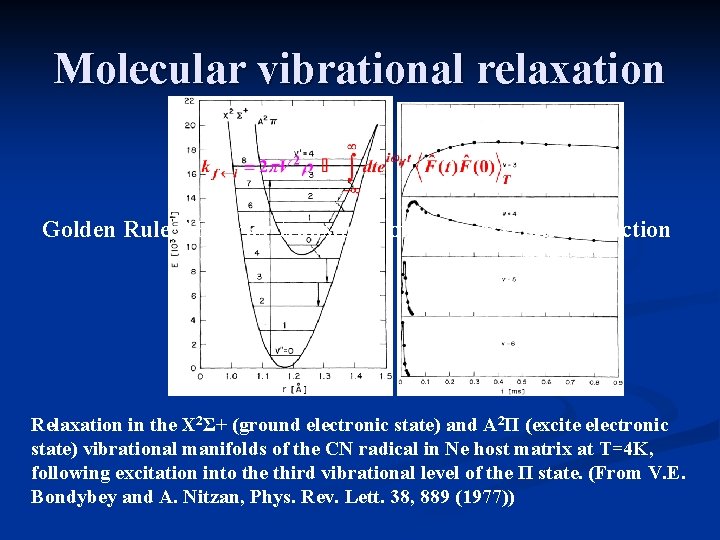
Molecular vibrational relaxation Golden Rule Fourier transform of bath correlation function Relaxation in the X 2Σ+ (ground electronic state) and A 2Π (excite electronic state) vibrational manifolds of the CN radical in Ne host matrix at T=4 K, following excitation into the third vibrational level of the Π state. (From V. E. Bondybey and A. Nitzan, Phys. Rev. Lett. 38, 889 (1977))
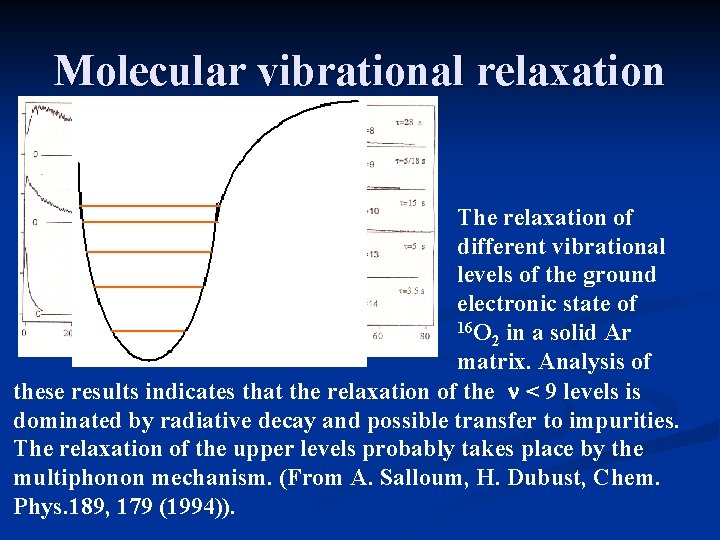
Molecular vibrational relaxation The relaxation of different vibrational levels of the ground electronic state of 16 O in a solid Ar 2 matrix. Analysis of these results indicates that the relaxation of the n < 9 levels is dominated by radiative decay and possible transfer to impurities. The relaxation of the upper levels probably takes place by the multiphonon mechanism. (From A. Salloum, H. Dubust, Chem. Phys. 189, 179 (1994)).

Frequency dependent friction MARKOVIAN LIMIT WIDE BAND APPROXIMATION

Dielectric solvation Born solvation energy Emission spectra of Coumarin 153 in formamide at different times. The times shown here are (in order of increasing peakwavelength) 0, 0. 05, 0. 1, 0. 2, 0. 5, 1, 2, 5, and 50 ps (Horng et al, J. Phys. Chem. 99, 17311 (1995))

Continuum dielectric theory of solvation How does solvent respond to a sudden change in the molecular charge distribution? Poisson equation Dielectric Electric Debye dielectric relaxation function displacement fieldmodel Electronic response polarization Dielectric susceptibility Total (static) response Debye relaxation time

Continuum dielectric theory of solvation WATER: t. D=10 ps t. L=125 fs

“real” solvation “Newton” dielectric The experimental solvation function for water using sodium salt of coumarin-343 as a probe. The line marked ‘expt’ is the experimental solvation function S(t) obtained from the shift in the fluorescence spectrum. The other lines are obtained from simulations [the line marked ‘Δq’ –simulation in water. The line marked S 0 –in a neutral atomic solute with Lennard Jones parameters of the oxygen atom]. (From R. Jimenez et al, Nature 369, 471 (1994)).

Electron solvation The first observation of hydration dynamics of electron. Absorption profiles of the electron during its hydration are shown at 0, 0. 08, 0. 2, 0. 4, 0. 7, 1 and 2 ps. The absorption changes its character in a way that suggests that two species are involved, the one that absorbs in the infrared is generated immediately and converted in time to the fully solvated electron. (From: A. Migus, Y. Gauduel, J. L. Martin and A. Antonetti, Phys. Rev Letters 58, 1559 (1987) Quantum solvation (1) Increase in the kinetic energy (localization) – seems NOT to affect dynamics (2) Non-adiabatic solvation (several electronic states involved)

Electron tunneling through water 1 2 3 Transient resonance through “structural defects” Polaronic state (solvated electron)
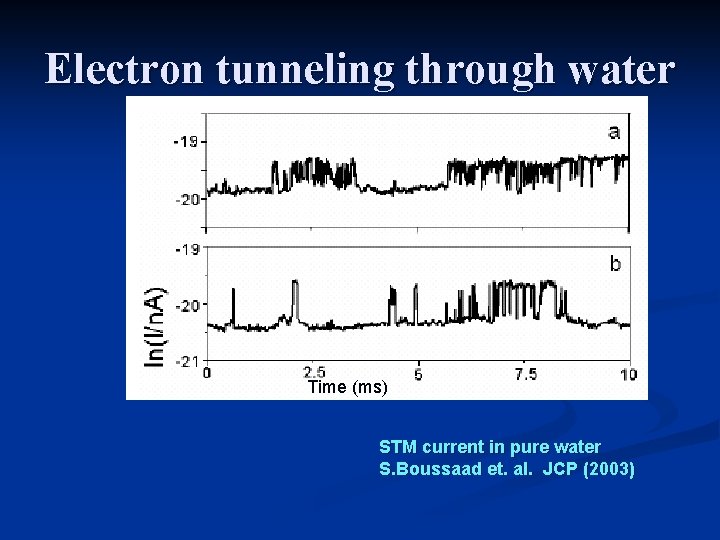
Electron tunneling through water Time (ms) STM current in pure water S. Boussaad et. al. JCP (2003)
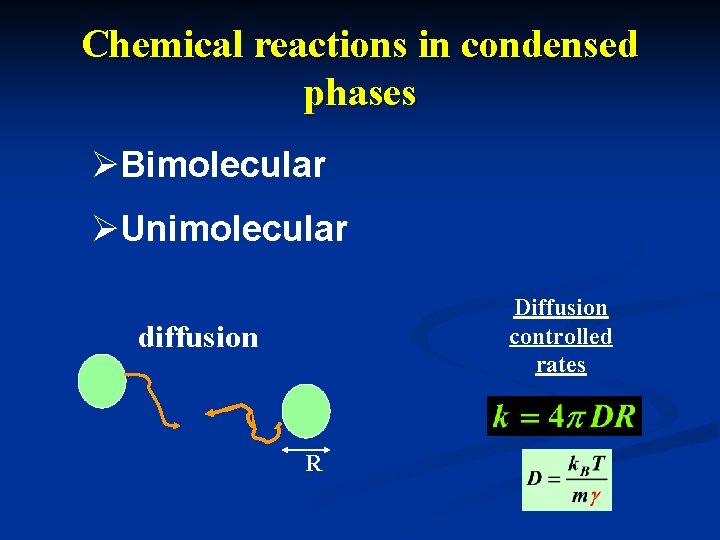
Chemical reactions in condensed phases ØBimolecular ØUnimolecular Diffusion controlled rates diffusion R
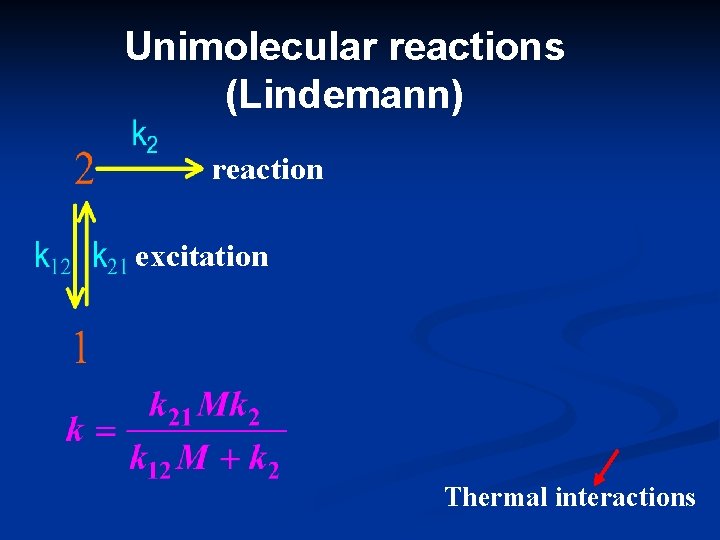
Unimolecular reactions (Lindemann) reaction excitation Thermal interactions
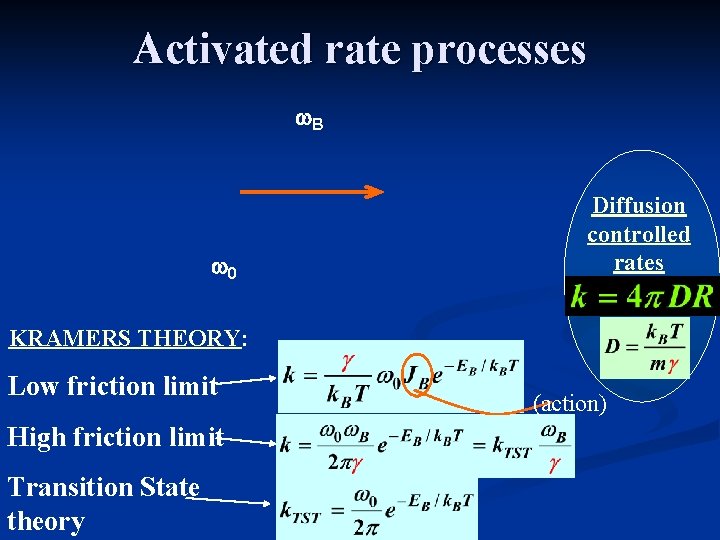
Activated rate processes w. B w 0 Diffusion controlled rates KRAMERS THEORY: Low friction limit High friction limit Transition State theory (action)

Effect of solvent friction TST A compilation of gas and liquid phase data showing the turnover of the photoisomerization rate of trans stilbene as a function of the “friction” expressed as the inverse self diffusion coefficient of the solvent (From G. R. Fleming and P. G. Wolynes, Physics Today, 1990). The solid line is a theoretical fit based on J. Schroeder and J. Troe, Ann. Rev. Phys. Chem. 38, 163 (1987)).

The physics of transition state rates Assume: (1) Equilibrium in the well (2) Every trajectory on the barrier that goes out makes it

The (classical) transition state rate is an upper bound • Assumed equilibrium in the well – in reality population will be depleted near the barrier • Assumed transmission coefficient unity above barrier top – in reality it may be less
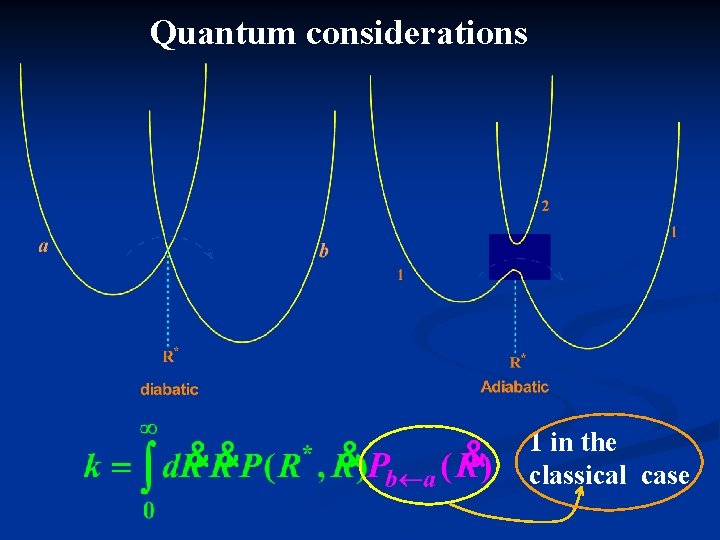
Quantum considerations 1 in the classical case

Cape Town, November 2006 (1 a) Relaxation and reactions in condensed molecular systems • Timescales • Relaxation • Solvation • Activated rate processes • Low, high and intermediate friction regimes • Transition state theory • Diffusion controlled reactions


A. Nitzan, Tel Aviv University The Abdus Salam African College on Science at the Nanoscale Cape Town, November 2007 1. Relaxation, reactions and electron transfer in condensed molecular systems 2. Fundamentals of molecular conduction 3. Inelastic effects in electron transfer and molecular conduction

Cape Town, November 2006 (1 a) Relaxation and reactions in condensed molecular systems • Timescales • Relaxation • Solvation • Activated rate processes • Low, high and intermediate friction regimes • Transition state theory • Diffusion controlled reactions

The importance of timescales
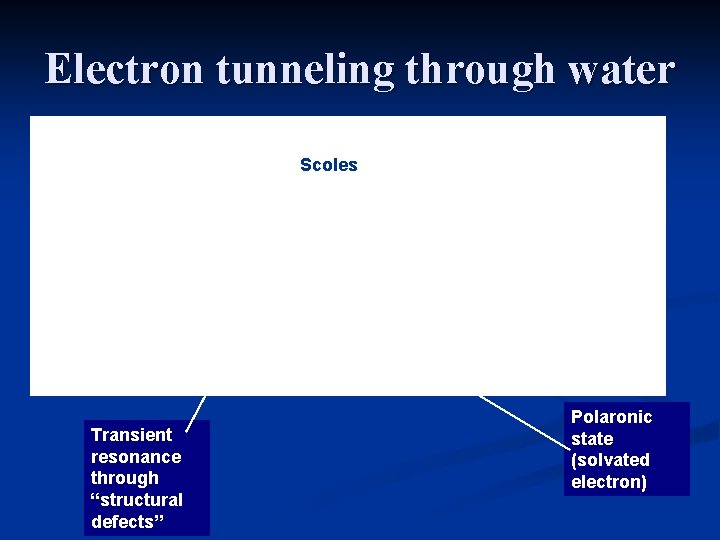
Electron tunneling through water 1 Scoles 2 3 Transient resonance through “structural defects” Polaronic state (solvated electron)
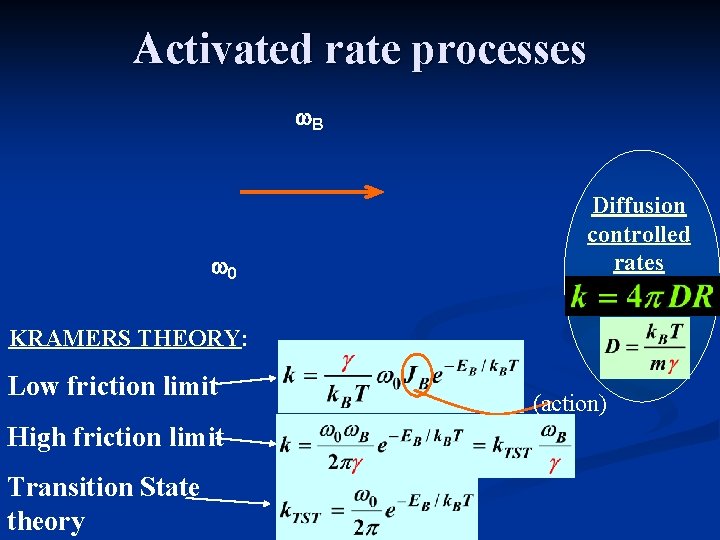
Activated rate processes w. B w 0 Diffusion controlled rates KRAMERS THEORY: Low friction limit High friction limit Transition State theory (action)

The physics of transition state rates Assume: (1) Equilibrium in the well (2) Every trajectory on the barrier that goes out makes it
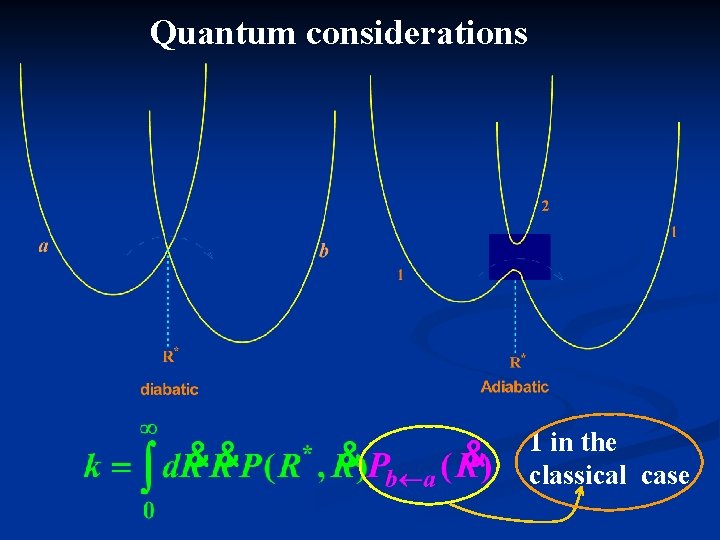
Quantum considerations 1 in the classical case

PART 1 B Electron transfer

Cape Town, November 2006 (1 b) Electron transfer processes Chapter 16 • Simple models • Marcus theory • The reorganization energy • Adiabatic and non-adiabatic limits • Solvent controlled reactions • Bridge assisted electron transfer • Coherent and incoherent transfer • Electrode processes

Theory of Electron Transfer n Rate – Transition state theory Transition rate n. Boltzmann n. Transition n. Activation probability energy

Electron transfer in polar media • Electrons are much faster than nuclei • Electronic transitions take place in fixed nuclear configurations • Electronic energy needs to be conserved during the change in electronic charge density Electronic transition Nuclear relaxation
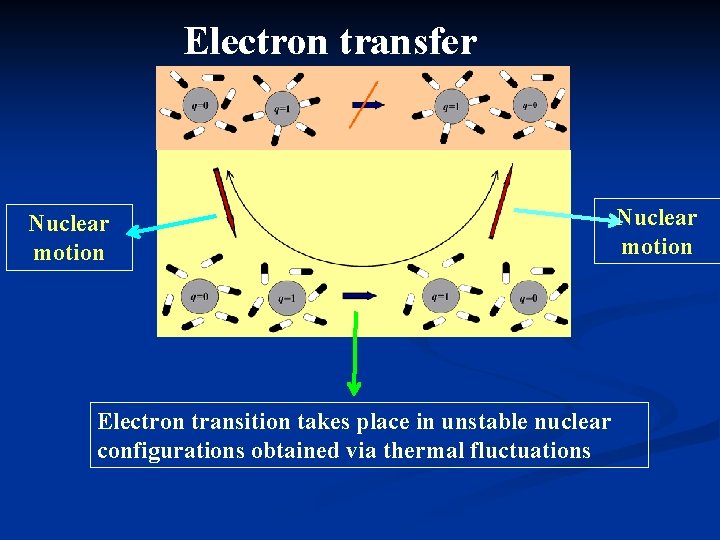
Electron transfer Nuclear motion Electron transition takes place in unstable nuclear configurations obtained via thermal fluctuations Nuclear motion
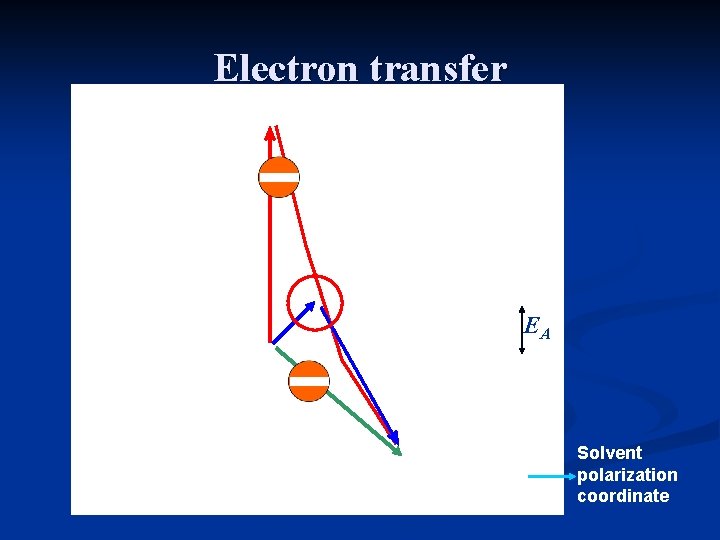
Electron transfer EA Solvent polarization coordinate

Transition state theory of electron transfer Adiabatic and non-adiabatic ET processes Landau-Zener problem (For harmonic diabatic surfaces (1/2)KR 2)
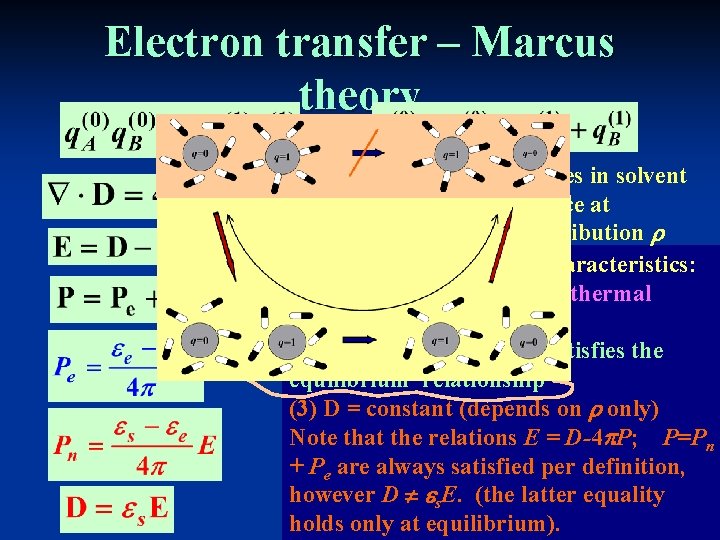
Electron transfer – Marcus theory We are interested in changes in solvent configuration that take place at constant solute charge distribution They have the following characteristics: (1) Pn fluctuates because of thermal motion of solvent nuclei. (2) Pe , as a fast variable, satisfies the equilibrium relationship (3) D = constant (depends on only) Note that the relations E = D-4 P; P=Pn + Pe are always satisfied per definition, however D s. E. (the latter equality holds only at equilibrium).
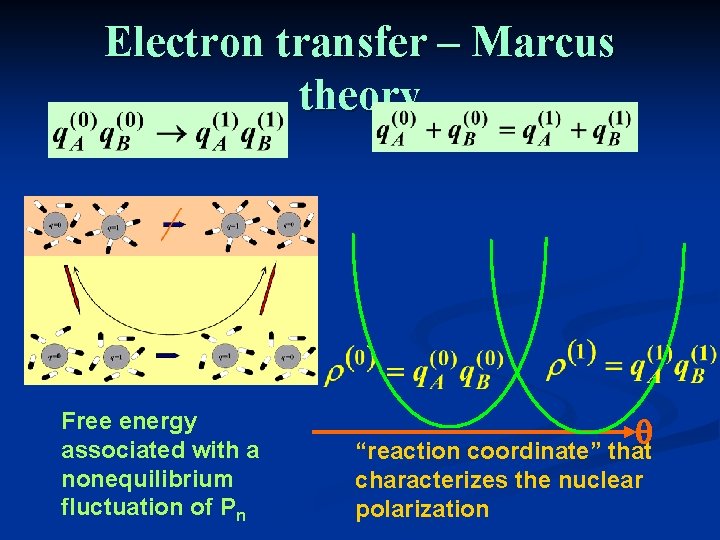
Electron transfer – Marcus theory Free energy associated with a nonequilibrium fluctuation of Pn q “reaction coordinate” that characterizes the nuclear polarization

The Marcus parabolas Use q as a reaction coordinate. It defines the state of the medium that will be in equilibrium with the charge distribution rq. Marcus calculated the free energy (as function of q) of the solvent when it reaches this state in the systems q =0 and q=1. q=0 q=1 q
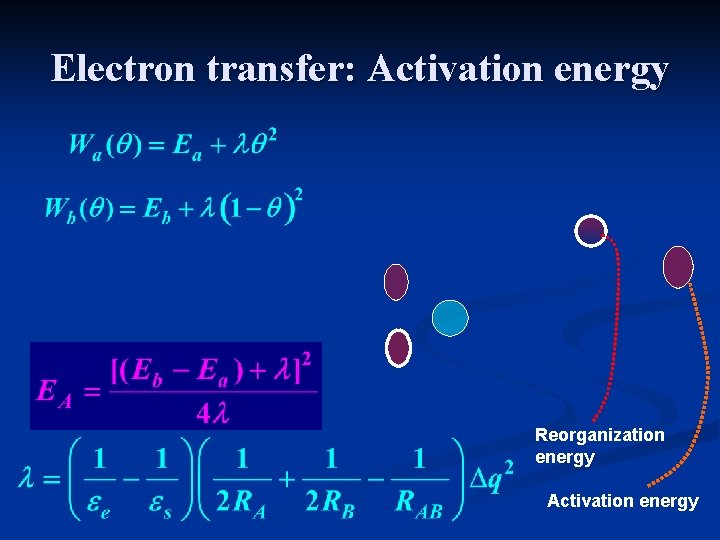
Electron transfer: Activation energy Reorganization energy Activation energy
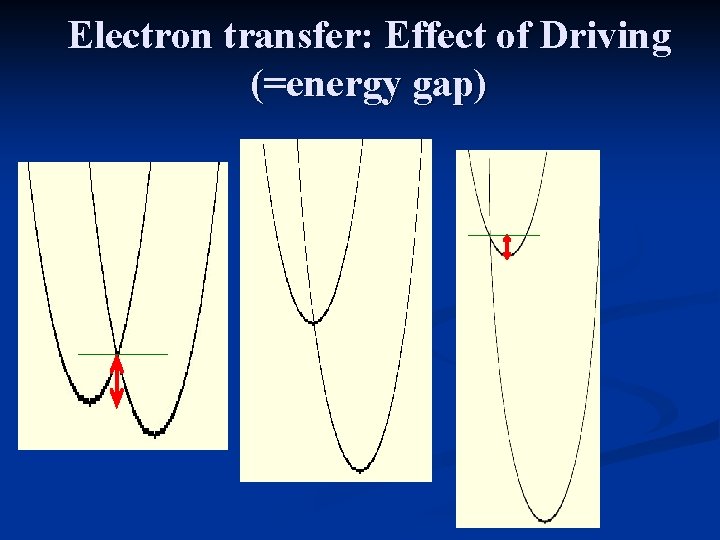
Electron transfer: Effect of Driving (=energy gap)

Experimental confirmation of the inverted regime Marcus papers 1955 -6 Miller et al, JACS(1984) Marcus Nobel Prize: 1992

Electron transfer – the coupling • From Quantum Chemical Calculations • The Mulliken-Hush formula • Bridge mediated electron transfer
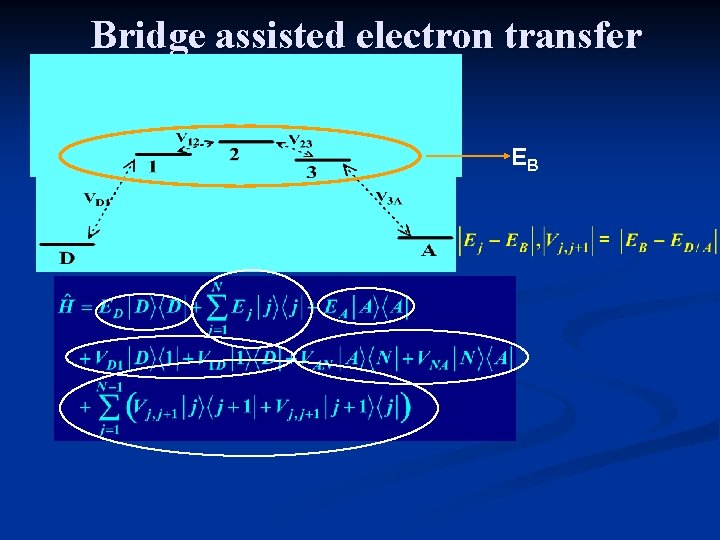
Bridge assisted electron transfer EB
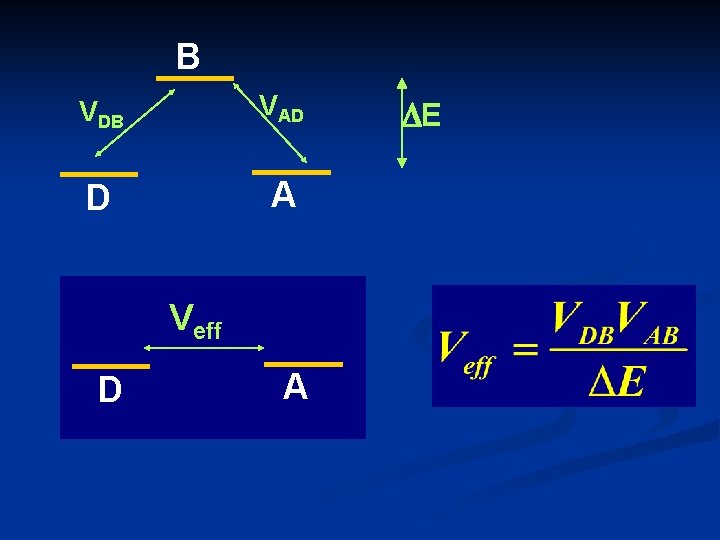
B VDB VAD D A Veff D A DE

B 1 DE VDB DD Veff B 2 … BN V 12 VAD A A Green’s Function

Marcus expresions for non-adiabatic ET rates Donor-to-Bridge/ Acceptor-to-bridge Bridge Green’s Function Franck-Condonweighted DOS Reorganization energy

Bridge mediated ET rate b’ (Å-1)= 0. 2 -0. 6 for highly conjugated chains 0. 9 -1. 2 for saturated hydrocarbons ~2 for vacuum
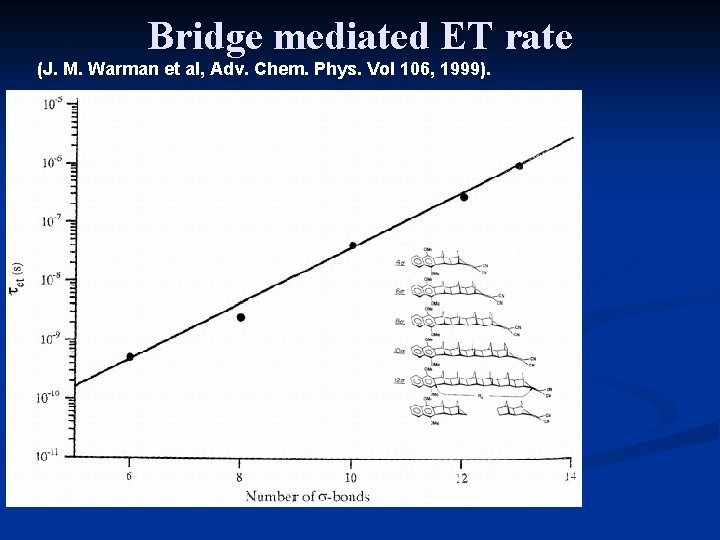
Bridge mediated ET rate (J. M. Warman et al, Adv. Chem. Phys. Vol 106, 1999).
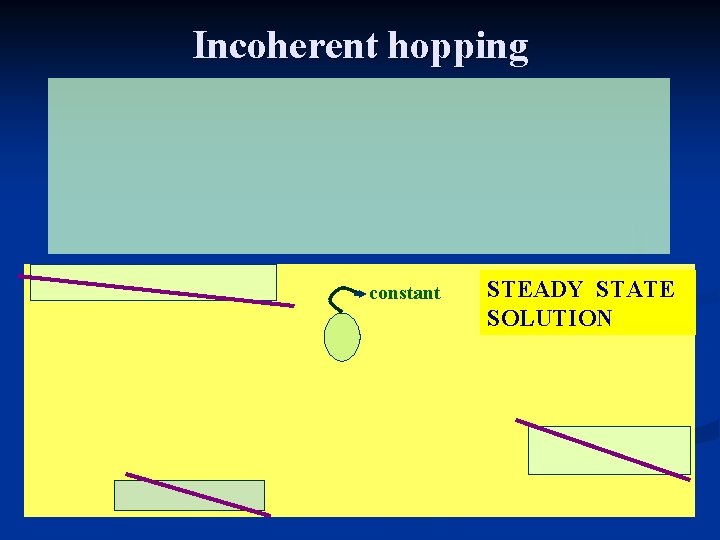
Incoherent hopping constant STEADY STATE SOLUTION

ET rate from steady state hopping

Dependence on temperature The integrated elastic (dotted line) and activated (dashed line) components of the transmission, and the total transmission probability (full line) displayed as function of inverse temperature. Parameters are as in Fig. 3.
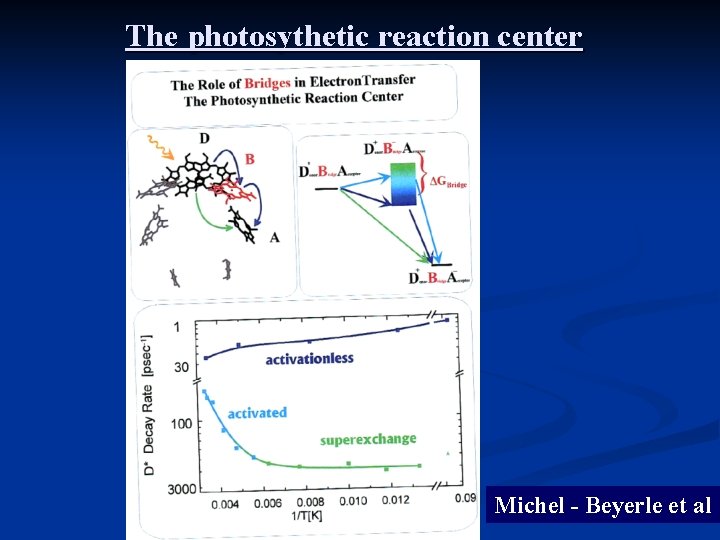
The photosythetic reaction center Michel - Beyerle et al

Dependence on bridge length
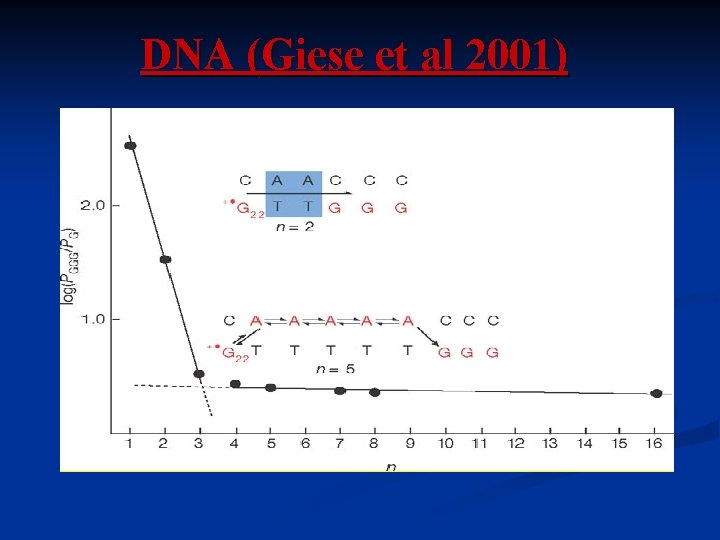
DNA (Giese et al 2001)

ELECTROCHEMISTRY

Donor gives an electron and goes from state a (reduced) to state b (oxidized). Eb, a=Eb - Ea is the energy of the electron given to the metal Transition rate to a continuum (Golden Rule) D Rate of electron transfer to metal in vacuum Rate of electron transfer to metal in electrolyte solution A EF M

Cape Town, November 2006 (1 b) Electron transfer processes Chapter 16 AN, Oxford University Press, 2006 • Simple models • Marcus theory • The reorganization energy • Adiabatic and non-adiabatic limits • Solvent controlled reactions • Bridge assisted electron transfer • Coherent and incoherent transfer • Electrode processes


A. Nitzan, Tel Aviv University The Abdus Salam African College on Science at the Nanoscale Cape Town, November 2007 1. Relaxation, reactions and electron transfer in condensed molecular systems 2. Fundamentals of molecular conduction 3. Inelastic effects in electron transfer and molecular conduction

Cape Town, November 2006 (1 a) Relaxation and reactions in condensed molecular systems • Timescales • Relaxation • Solvation • Activated rate processes • Low, high and intermediate friction regimes • Transition state theory • Diffusion controlled reactions

Cape Town, November 2006 (1 b) Electron transfer processes • Simple models • Marcus theory • The reorganization energy • Adiabatic and non-adiabatic limits • Solvent controlled reactions • Bridge assisted electron transfer • Coherent and incoherent transfer • Electrode processes
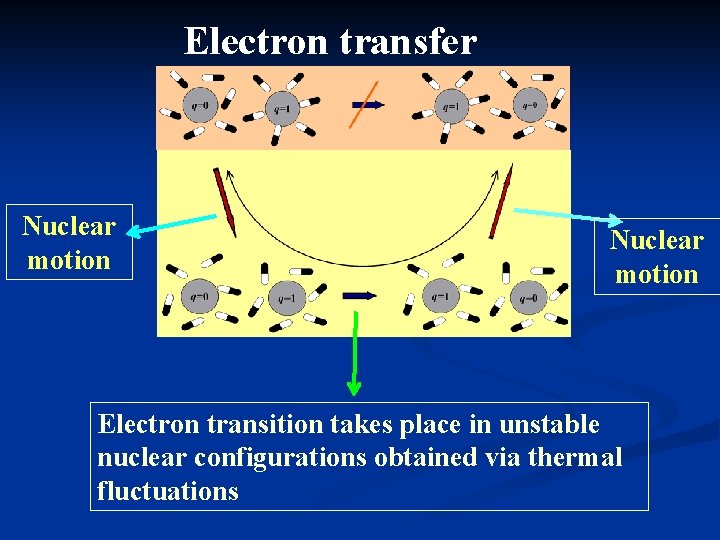
Electron transfer Nuclear motion Electron transition takes place in unstable nuclear configurations obtained via thermal fluctuations
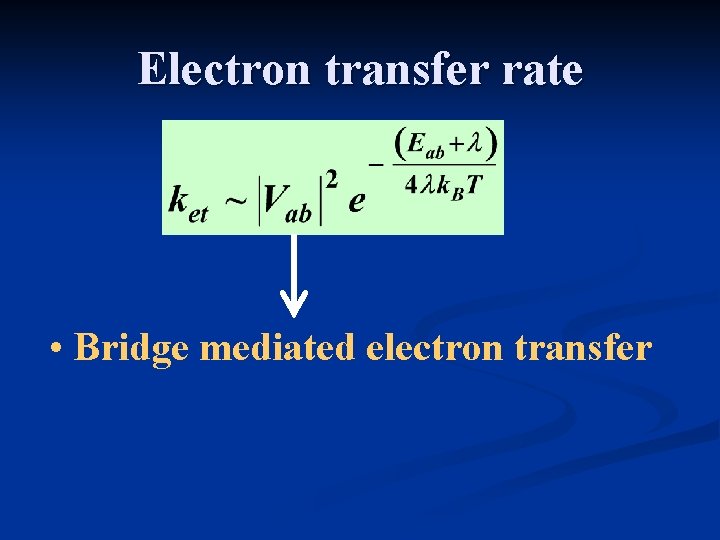
Electron transfer rate • Bridge mediated electron transfer

Marcus expresions for non-adiabatic ET rates Donor-to-Bridge/ Acceptor-to-bridge Bridge Green’s Function Franck-Condonweighted DOS Reorganization energy

Incoherent hopping

2 Molecular conduction

Cape Town, November 2006 (2) Molecular conduction Chapter 17 AN, Oxford University Press, 2006 • Simple models for molecular conductions • Factors affecting electron transfer at interfaces • The Landauer formula • Molecular conduction by the Landauer formula • Relationship to electron-transfer rates. • Structure-function effects in molecular conduction • Electron transfer by hopping • Inelastic tunneling spectroscopy

Steady state evaluation of rates Rate of water flow depends linearly on water height in the cylinder h Two ways to get the rate of water flowing out: (1) Measure h(t) and get the rate coefficient from k=(1/h)dh/dt (2) Keep h constant and measure the steady state outwards water flux J. Get the rate from k=J/h (3) = Steady state rate

Steady state quantum mechanics Starting from state 0 at t=0: P 0 = exp(-G 0 t) V 0 l G 0 = 2 |V 0 l|2 L (Golden Rule) Steady state derivation:

pumping damping
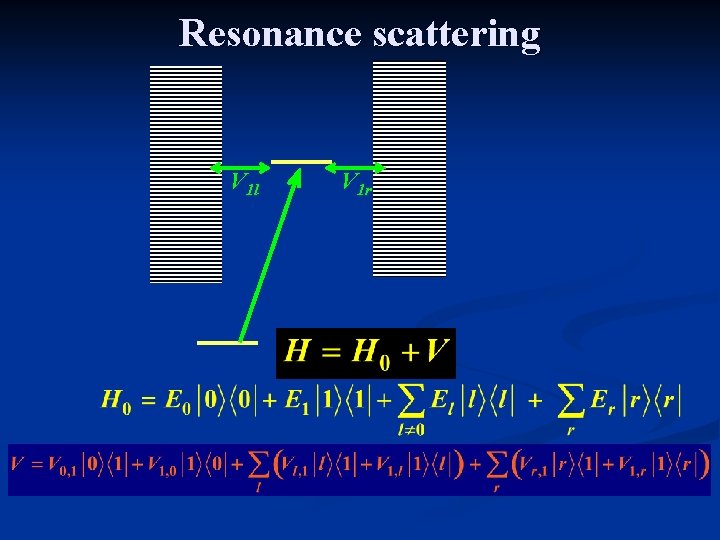
Resonance scattering V 1 l V 1 r

Resonance scattering For each r and l j = 0, 1, {l}, {r}
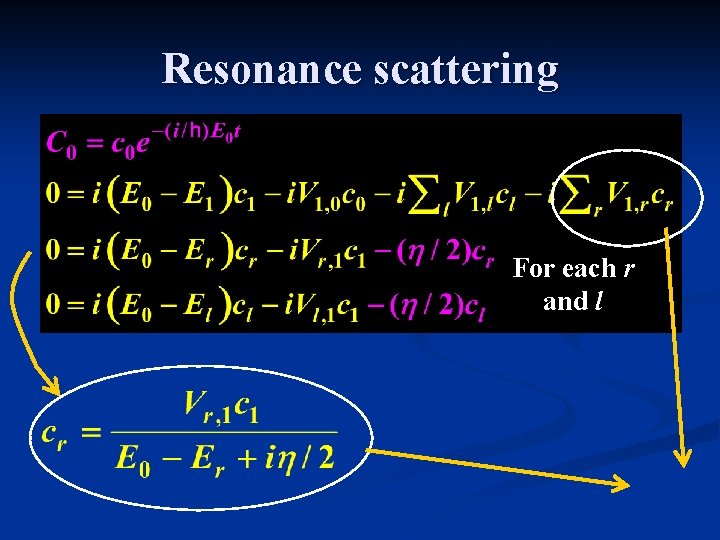
Resonance scattering For each r and l

SELF ENERGY

V 1 l V 1 r
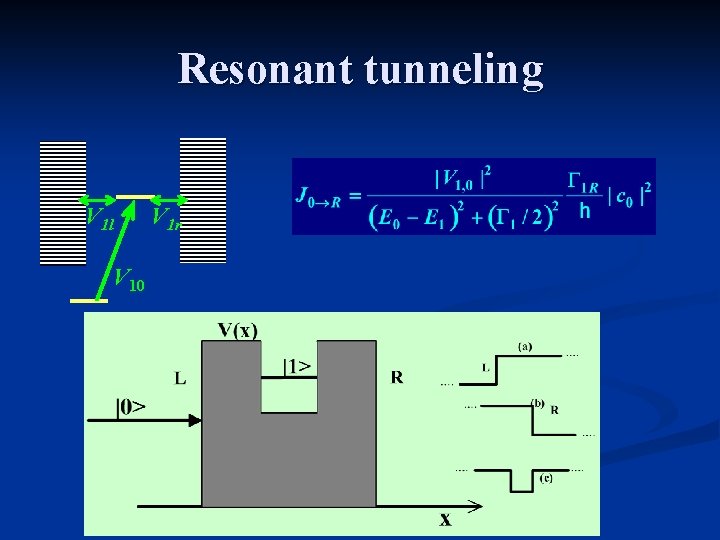
Resonant tunneling V 1 l V 10 V 1 r
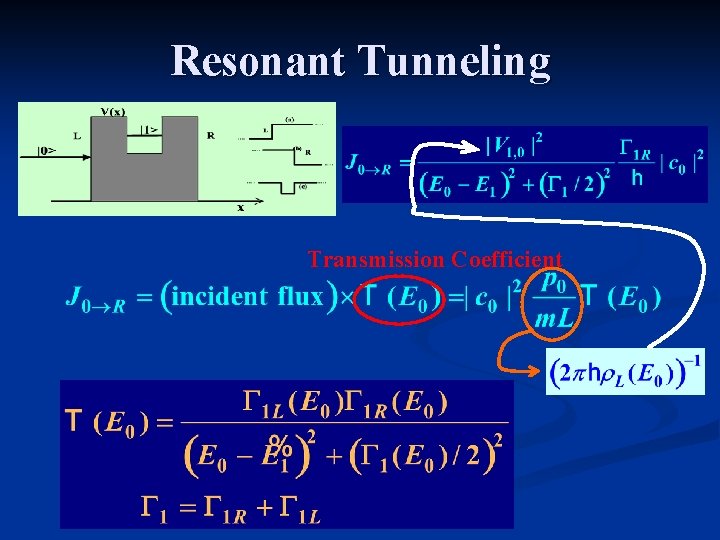
Resonant Tunneling Transmission Coefficient
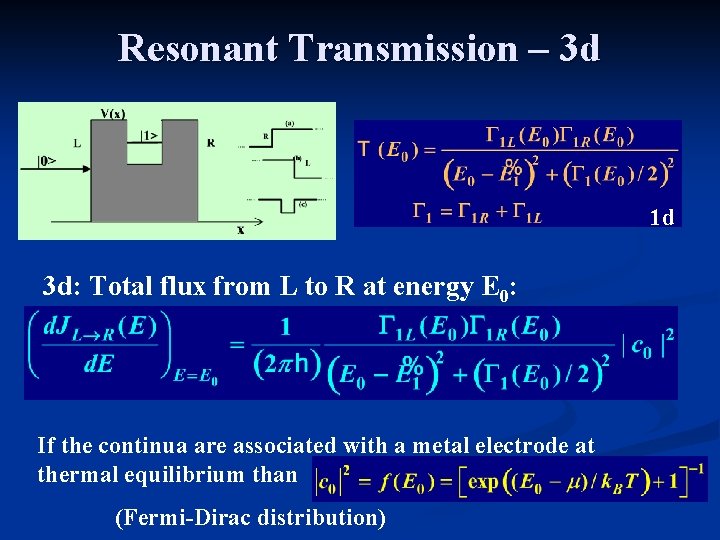
Resonant Transmission – 3 d 1 d 3 d: Total flux from L to R at energy E 0: If the continua are associated with a metal electrode at thermal equilibrium than (Fermi-Dirac distribution)

CONDUCTION L R m – |e|f m f(E 0) (Fermi function) 2 spin states Zero bias conduction

Landauer formula For a single “channel”: (maximum=1) Maximum conductance per channel
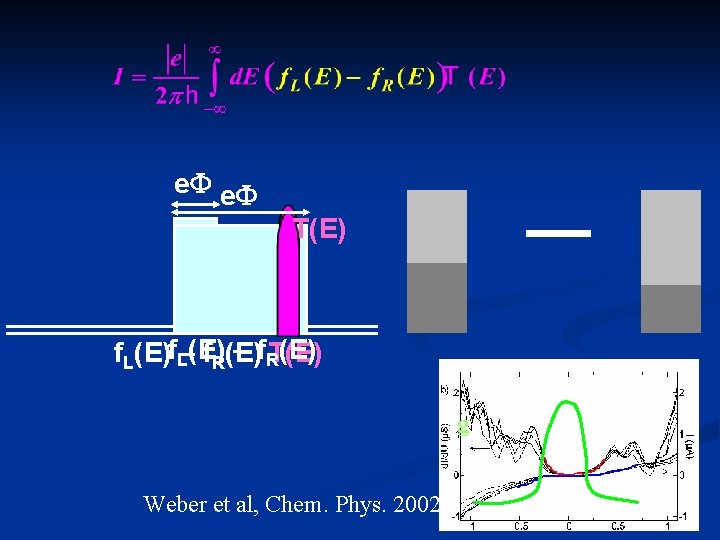
e. F T(E) – )f. RT(E) f. L(E)f. L–(Ef. R)(E I g Weber et al, Chem. Phys. 2002 F
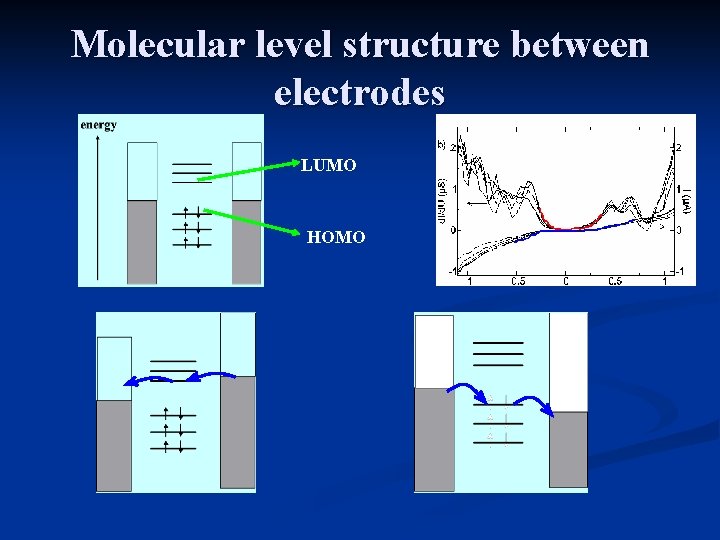
Molecular level structure between electrodes LUMO HOMO
GATING IONIC GATING
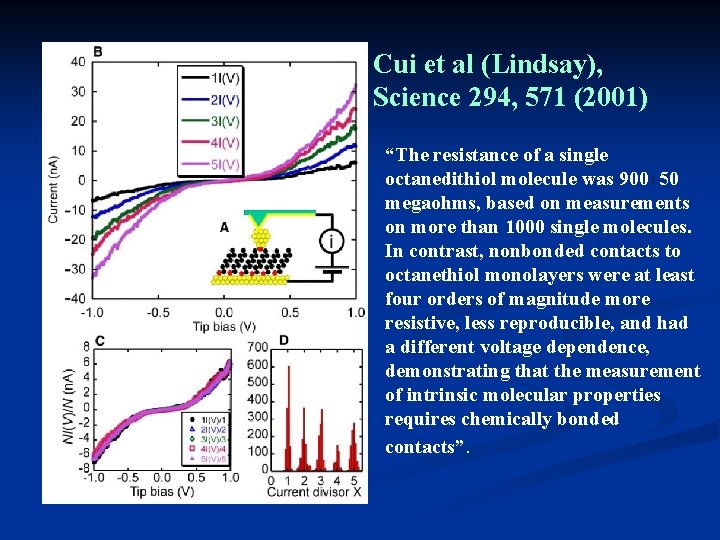
Cui et al (Lindsay), Science 294, 571 (2001) “The resistance of a single octanedithiol molecule was 900 50 megaohms, based on measurements on more than 1000 single molecules. In contrast, nonbonded contacts to octanethiol monolayers were at least four orders of magnitude more resistive, less reproducible, and had a different voltage dependence, demonstrating that the measurement of intrinsic molecular properties requires chemically bonded contacts”.
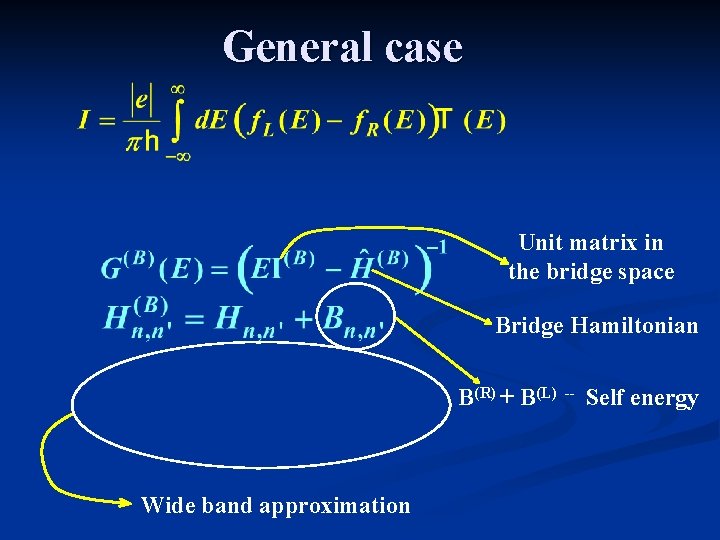
General case Unit matrix in the bridge space Bridge Hamiltonian B(R) + B(L) Wide band approximation -- Self energy
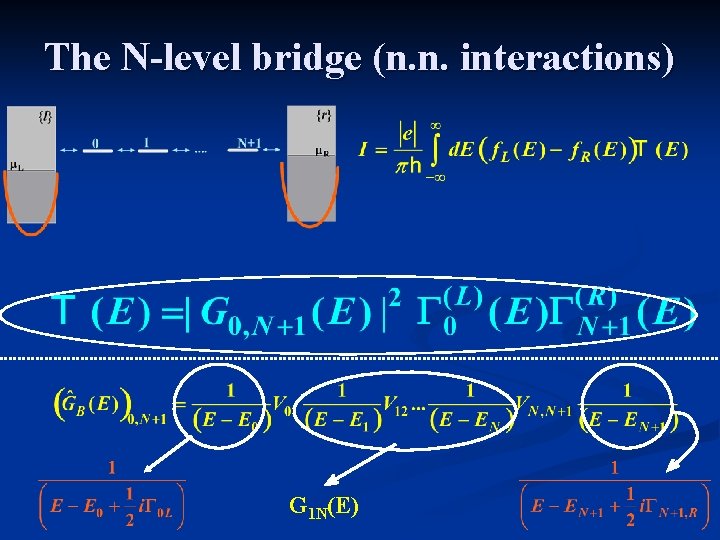
The N-level bridge (n. n. interactions) G 1 N(E)

ET vs Conduction
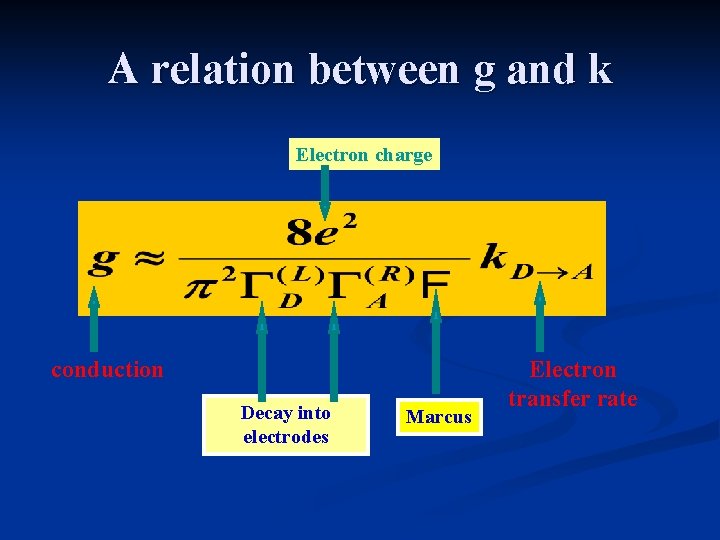
A relation between g and k Electron charge conduction Decay into electrodes Marcus Electron transfer rate

A relation between g and k l 0. 5 e. V

Comparing conduction to rates (M. Newton, 2003)
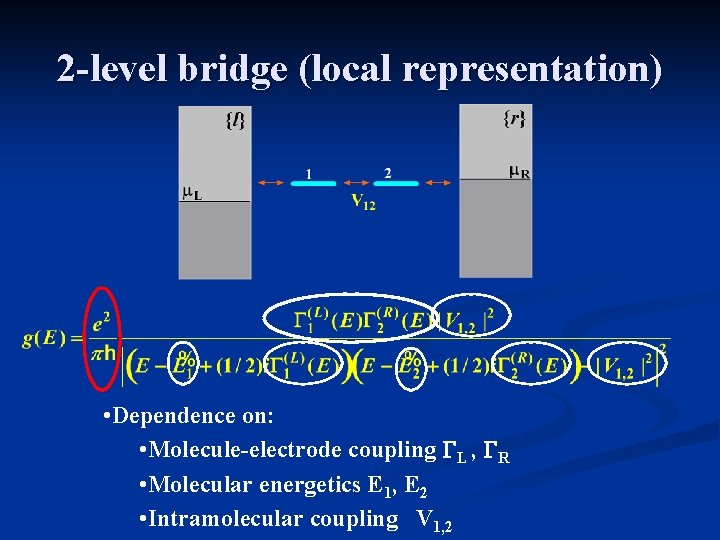
2 -level bridge (local representation) • Dependence on: • Molecule-electrode coupling GL , GR • Molecular energetics E 1, E 2 • Intramolecular coupling V 1, 2
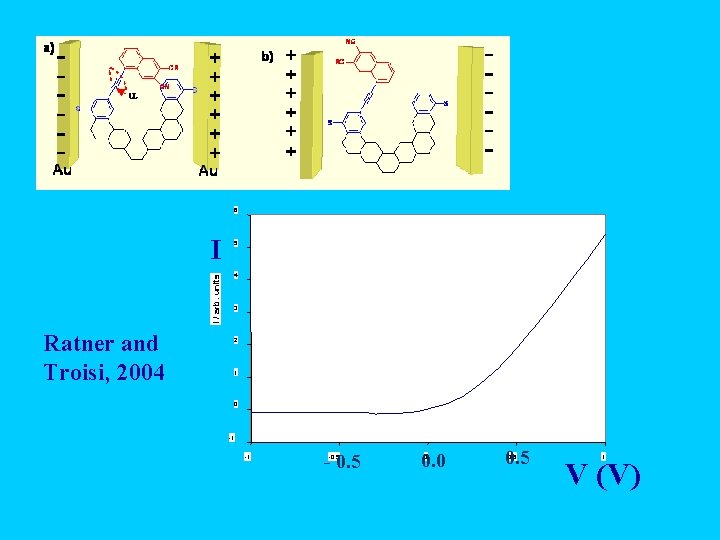
6 I / arb. units I Ratner and Troisi, 2004 5 4 3 2 1 0 -1 -1 - 0. 5 -0. 5 0. 0 0 0. 5 1 V (V)
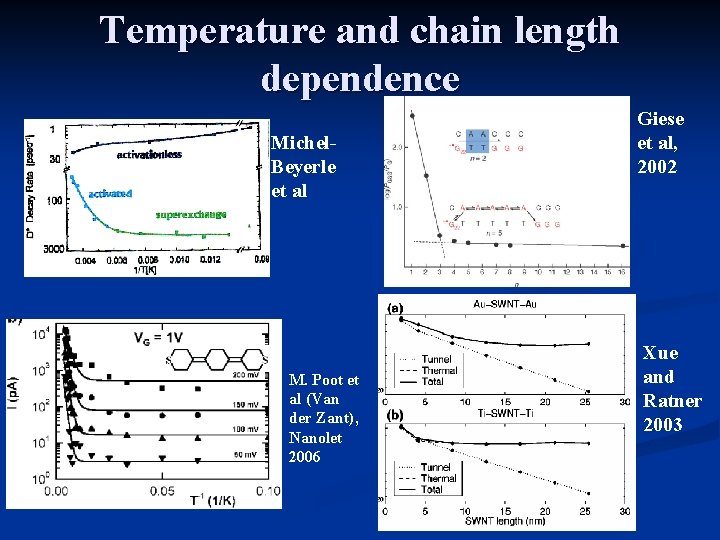
Temperature and chain length dependence Michel. Beyerle et al M. Poot et al (Van der Zant), Nanolet 2006 Giese et al, 2002 Xue and Ratner 2003

Barrier dynamics effects on electron transmission through molecular wires • RELEVANT TIMESCALES • INELASTIC CONTRIBUTIONS TO CURRENT • DEPHASING AND ACTIVATION • HEATING • HEAT CONDUCTION -- RECTIFICATION • INELASTIC TUNNELING SPECTROSCOPY • STRONG e-ph COUPLING: (a) resonance inelastic tunneling spectroscopy (b) multistability and hysteresis • NOISE
INELSTIC ELECTRON TUNNELING SPECTROSCOPY
Light Scattering incident scattered
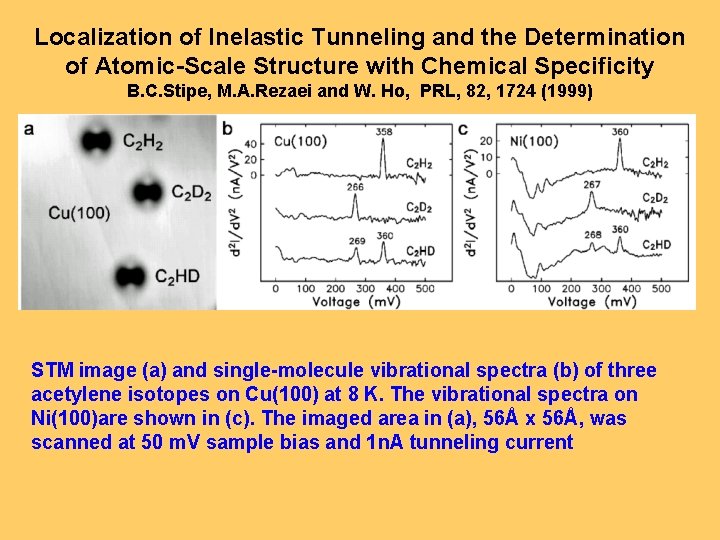
Localization of Inelastic Tunneling and the Determination of Atomic-Scale Structure with Chemical Specificity B. C. Stipe, M. A. Rezaei and W. Ho, PRL, 82, 1724 (1999) STM image (a) and single-molecule vibrational spectra (b) of three acetylene isotopes on Cu(100) at 8 K. The vibrational spectra on Ni(100)are shown in (c). The imaged area in (a), 56Å x 56Å, was scanned at 50 m. V sample bias and 1 n. A tunneling current
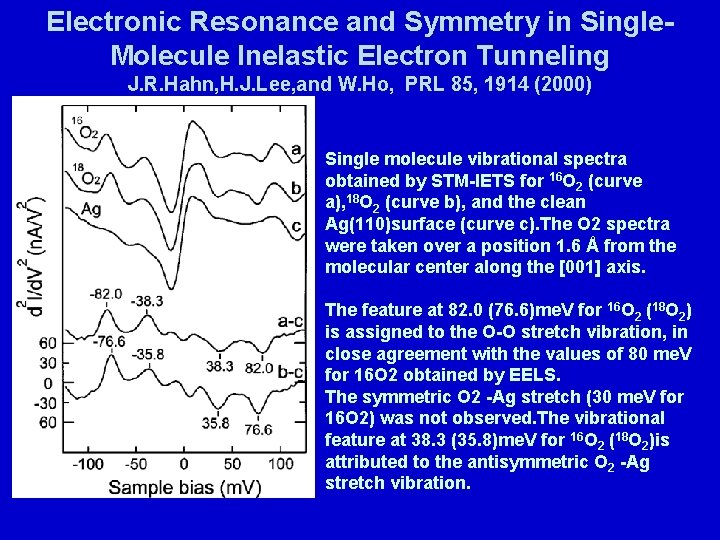
Electronic Resonance and Symmetry in Single. Molecule Inelastic Electron Tunneling J. R. Hahn, H. J. Lee, and W. Ho, PRL 85, 1914 (2000) Single molecule vibrational spectra obtained by STM-IETS for 16 O 2 (curve a), 18 O 2 (curve b), and the clean Ag(110)surface (curve c). The O 2 spectra were taken over a position 1. 6 Å from the molecular center along the [001] axis. The feature at 82. 0 (76. 6)me. V for 16 O 2 (18 O 2) is assigned to the O-O stretch vibration, in close agreement with the values of 80 me. V for 16 O 2 obtained by EELS. The symmetric O 2 -Ag stretch (30 me. V for 16 O 2) was not observed. The vibrational feature at 38. 3 (35. 8)me. V for 16 O 2 (18 O 2)is attributed to the antisymmetric O 2 -Ag stretch vibration.
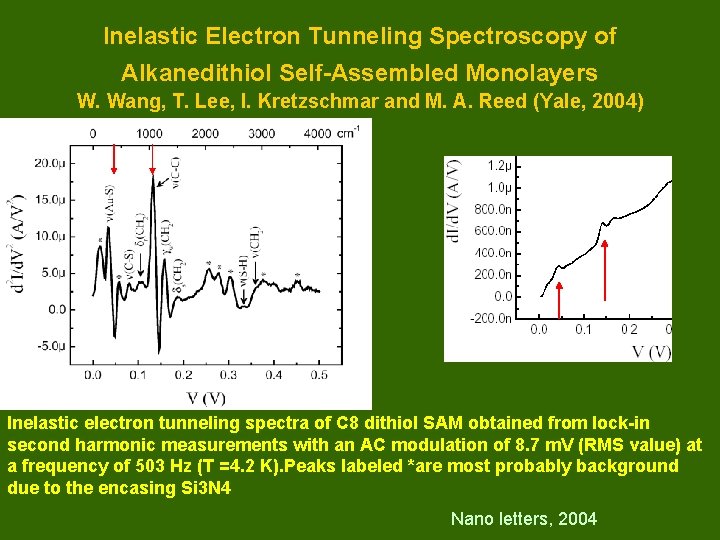
Inelastic Electron Tunneling Spectroscopy of Alkanedithiol Self-Assembled Monolayers W. Wang, T. Lee, I. Kretzschmar and M. A. Reed (Yale, 2004) Inelastic electron tunneling spectra of C 8 dithiol SAM obtained from lock-in second harmonic measurements with an AC modulation of 8. 7 m. V (RMS value) at a frequency of 503 Hz (T =4. 2 K). Peaks labeled *are most probably background due to the encasing Si 3 N 4 Nano letters, 2004
INELSTIC ELECTRON TUNNELING SPECTROSCOPY
INELASTIC ELECTRON TUNNELING SPECTROSCOPY
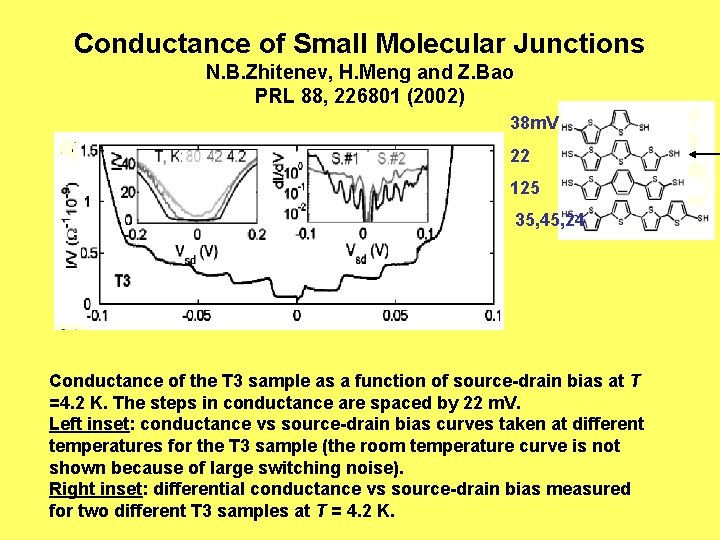
Conductance of Small Molecular Junctions N. B. Zhitenev, H. Meng and Z. Bao PRL 88, 226801 (2002) 38 m. V 22 125 35, 45, 24 Conductance of the T 3 sample as a function of source-drain bias at T =4. 2 K. The steps in conductance are spaced by 22 m. V. Left inset: conductance vs source-drain bias curves taken at different temperatures for the T 3 sample (the room temperature curve is not shown because of large switching noise). Right inset: differential conductance vs source-drain bias measured for two different T 3 samples at T = 4. 2 K.

SUMMARY THANK YOU (2) Molecular conduction (1 b) Electron transfer A. Nitzan, Tel Aviv University • (1 a) Simple. Relaxation models for molecular conductions and reactions in processes • Factors affecting electron transfer at • Simple models condensed interfaces INTRODUCTION TOmolecular ELECTRONsystems • Kinetic Marcus theory models • • The Landauer formula TRANSFER AND MOLRCULAR • Transition The reorganization • • Molecular conduction byenergy the Landauer CONDUCTION state theory formula Adiabatic and non-adiabatic • • Kramers theory and its extensions Download: • • Relationship to electron-transfer rates. limits Low, high and intermediate http: //atto. tau. ac. il/~nitzan/Cape Town 07. ppt • Structure-function effects in molecular • Solvent controlled reactions friction regimes conduction • Diffusion Bridge assisted electron transfer controlled • • Electronic conduction byreactions hopping AN, Oxford University • Coherent and incoherent • Inelastic tunneling spectroscopy Press, 2006 Chapter 16 Chapter 13 -15 17 transfer • Electrode processes

- Slides: 114