Why As youve already seen Chemical reactions occur
























- Slides: 28

Why? ? ? As you’ve already seen… Chemical reactions occur around us everyday--- so many in fact that looking at them all individually would be REALLY overwhelming! So instead we name them based what happens!

Classification of Chemical Reactions �Chemical reactions can be classified in one of two ways: 1. Based on atoms moving (rearrangement) 2. Based on how energy/ heat is transferred VS.

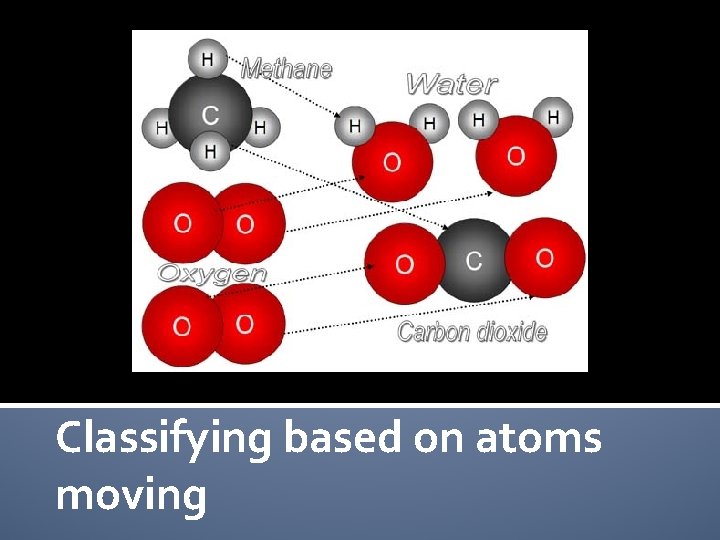
Classifying based on atoms moving

Boy + Girl = Couple 1) Synthesis Reaction This is a reaction in which two or more elements or compounds combine to form a single product. This type of reaction follows the general equation A + B AB owhere A and B may be either elements or compounds. +

Example: Synthesis Burning Magnesium � 2 Mg + O 2 2 Mg. O

The couple breaks up! Decomposition Reaction In this type of reaction a single reactant, a compound, breaks into two or more parts. Often these are the most difficult to predict. Here is the general equation: AB A + B owhere A and B may be either elements or compounds. +

Example: Decomposition Hydrogen Peroxide � 2 H 2 O 2 2 H 2 O +O 2

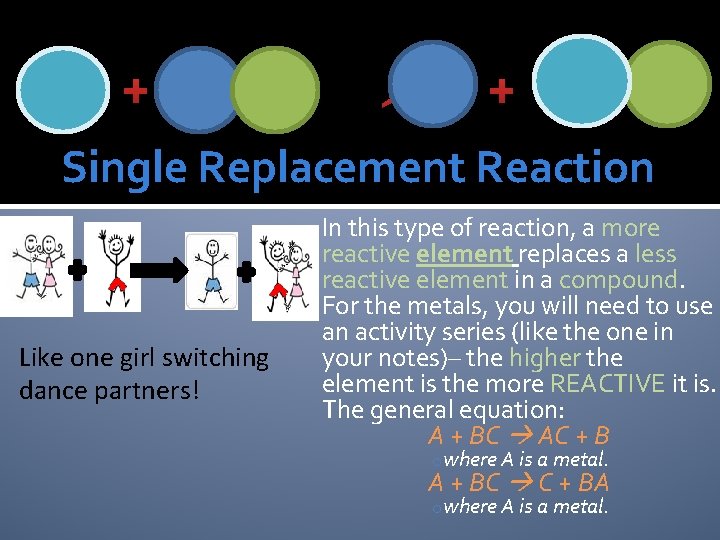
+ + Single Replacement Reaction Like one girl switching dance partners! In this type of reaction, a more reactive element replaces a less reactive element in a compound. For the metals, you will need to use an activity series (like the one in your notes)– the higher the element is the more REACTIVE it is. The general equation: A + BC AC + B owhere A is a metal. A + BC C + BA owhere A is a metal.

Example: Single Replacement Magnesium and Carbon Dioxide � 2 Mg + CO 2 2 Mg. O + C

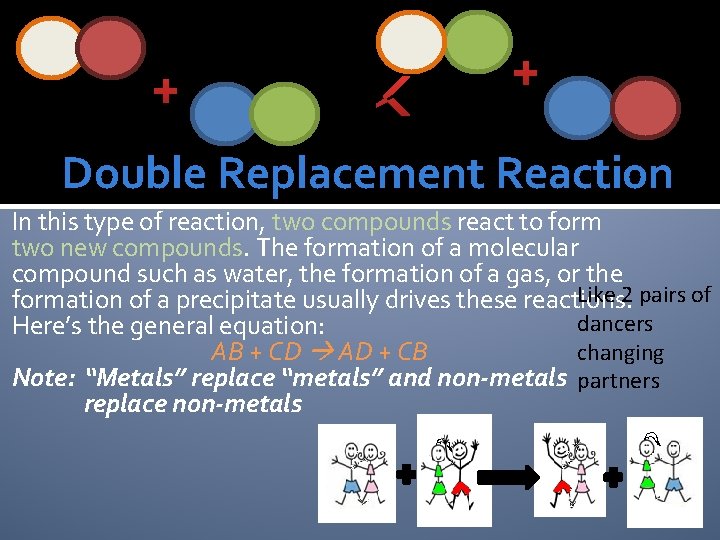
+ + Double Replacement Reaction In this type of reaction, two compounds react to form two new compounds. The formation of a molecular compound such as water, the formation of a gas, or the Like 2 pairs of formation of a precipitate usually drives these reactions. dancers Here’s the general equation: changing AB + CD AD + CB Note: “Metals” replace “metals” and non-metals partners replace non-metals

Example: Double Replacement Potassium Iodide and Lead (II) Nitrate 2 KI(aq) + Pb(NO 3)2(aq) 2 KNO 3(aq) + Pb. I 2(s)

Combustion Reaction In this type of reaction, a hydrocarbon is burned in the presence of oxygen gas to form carbon dioxide and water. Here is the general equation in the presence of plenty of oxygen: Cx. Hy + O 2(g) CO 2(g) + H 2 O(l) or (g) Note: If combustion is inefficient (insufficient oxygen) then carbon monoxide is formed!

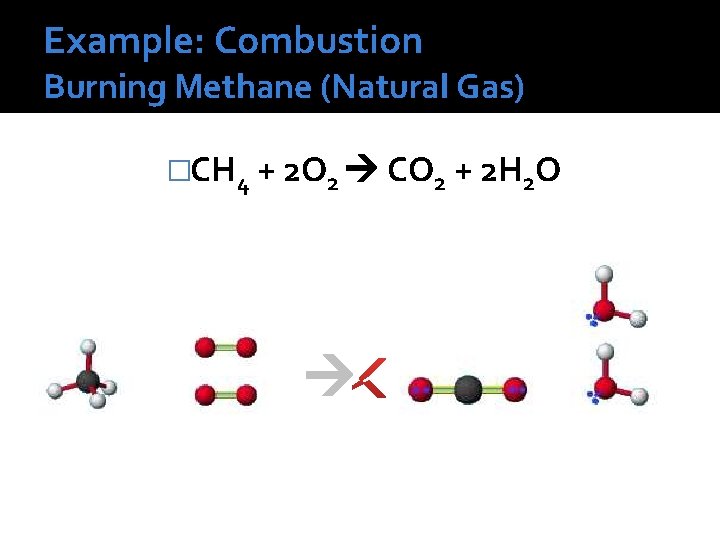
Example: Combustion Burning Methane (Natural Gas) �CH 4 + 2 O 2 CO 2 + 2 H 2 O

Neutralization Reactions Unfortunately we cannot forget about neutralization reactions (a. k. a. acidbase reactions)! In this type of reaction, an acid and a base react to form a salt and water. The general equation for this type of reaction is: HX + MOH MX +H 2 O Where M is a metal ion and X is a non-metal ion

Classifying based on energy transfer

Endothermic vs. Exothermic Reactions are classified by looking at whether energy is required or released during a chemical reaction: 1. EXOTHERMIC (exo= outside)- energy is released during a chemical reaction (i. e. is a product), given off as heat 2. ENDOTHERMIC (endo= inside)- energy is required during a chemical reaction (i. e. is a reactant), and heat is absorbed

Example of an Exothermic Reaction: Decomposition of H 2 O 2

Example of an Endothermic Reaction: The “Green” Cold pack— Ammonium Nitrate in Water

Six Questions to find the type of Chemical Reaction Follow this series of questions. When you can answer "yes" to a question, then stop! 1) Does your reaction have oxygen as one of it's reactants and carbon dioxide and water as products? If yes, then it's a combustion reaction 2) Does your reaction have two (or more) chemicals combining to form one chemical? If yes, then it's a synthesis reaction 3) Does your reaction have one large molecule falling apart to make several small ones? If yes, then it's a decomposition reaction

Six Questions to find the type of Chemical Reaction cont. 4) Does your reaction have any molecules that contain only one element? If yes, then it's a single displacement reaction 5) Does your reaction have water as one of the products? If yes, then it's an acid-base reaction 6) If you haven't answered "yes" to any of the questions above, then you've got a double displacement reaction

Practice Problems 1. Fe 2 O 3 5. C 18 H 38 +O 2 2. Al + Ni. Br 2 6. Li + N 2 3. Cl 2 + Ni. Br 2 7. Ag. NO 3 + Na 2 Cr. O 4 4. HCl + Mg(OH)2

Fe 2 O 3 Skeleton Equation: Fe 2 O 3 Fe + O Balanced Equation: Fe 2 O 3 4 Fe + 3 O 2

Al + Ni. Br 2 Skeleton Equation: Al + Ni. Br 2 Ni + Al. Br 3 Balanced Equation: 2 Al + 3 Ni. Br 2 3 Ni + 2 Al. Br 3

Cl 2 + Ni. Br 2 Skeleton Equation: Cl 2 + Ni. Br 2 + Ni. Cl 2 Balanced Equation: Cl 2 + Ni. Br 2 + Ni. Cl 2

HCl + Mg(OH)2 Skeleton Equation: HCl + Mg(OH)2 Mg. Cl 2 + H 2 O Balanced Equation: 2 HCl + Mg(OH)2 Mg. Cl 2 + 2 H 2 O

C 18 H 38 +O 2 Skeleton Equation: C 18 H 38 +O 2 CO 2 + H 2 O Balanced Equation: 2 C 18 H 38 + 55 O 2 36 CO 2 + 38 H 2 O

Li + N 2 Skeleton Equation: Li + N 2 Li 3 N Balanced Equation: 6 Li + N 2 2 Li 3 N

Ag. NO 3 + Na 2 Cr. O 4 Skeleton Equation: Ag. NO 3 + Na 2 Cr. O 4 Na. NO 3 + Ag 2 Cr. O 4 Balanced Equation: 2 Ag. NO 3 + Na 2 Cr. O 4 2 Na. NO 3 + Ag 2 Cr. O 4