Hybridization Localized Electron Model Why Hybridization Occurs n

Hybridization: Localized Electron Model

Why Hybridization Occurs n n n Example: Methane (CH 4) 4 hydrogens bonded to the central carbon atom The 4 electrons involved in bonding on the C atom are: 2 s 2 ¨ 2 p 2 ¨ n n Which of these electrons have higher energy levels? But all 4 bonds have the SAME energy level.

Why Hybridization Occurs (continued) n To accomplish this “same energy level bonds” arrangement, the 4 electron orbitals “hybridize” to a combined s and p orbital, with energy somewhere between the two original s and p orbitals.
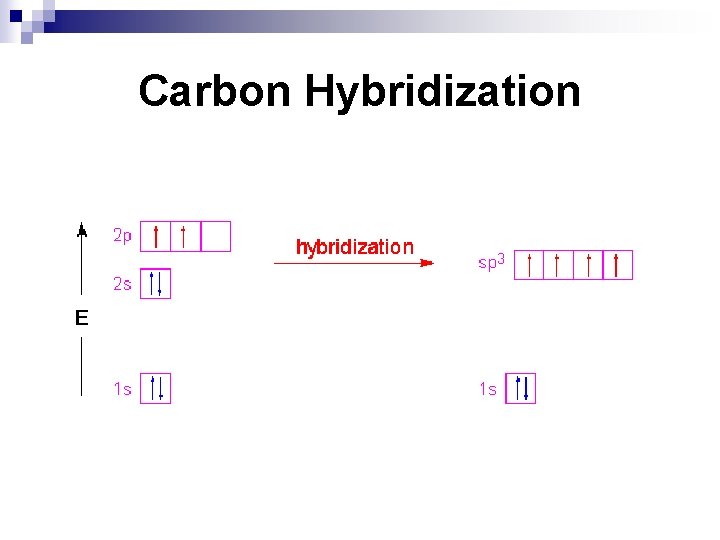
Carbon Hybridization

Hybridization Generalizations n Hybridizations refer to effective pairs! A double bond is how many effective pairs? ¨ A triple bond is how many effective pairs? ¨ 4 effective pairs = sp 3 orbitals n 3 effective pairs = sp 2 orbitals n 2 effective pairs = ? ? ? n 5 effective pairs = ? ? ? n

sp 2 Hybridization n n n Example: H 2 CO Central atom? How many effective pairs around the central atom? What electrons on the C atom are involved in bonding? This time, the 2 s 2 and 2 p 2 electrons only need to hybridizes to form 3 orbitals. You will have 3 sp 2 hybrid orbitals and a lone original p orbital.
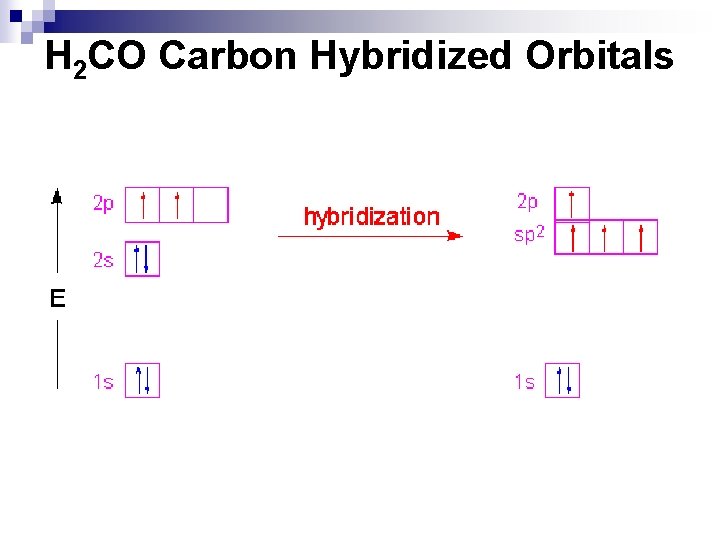
H 2 CO Carbon Hybridized Orbitals

H 2 CO Oxygen Orbitals n n n How many effective pairs exist around the oxygen atom in H 2 CO? What type of hybridization is this? In the molecule, how may of each of the following exist? ¨ ¨ ¨ Lone pairs Single bonds Double bonds Hybrid orbitals on the C atom Hybrid orbitals on the O atom Regular (unhybridized) orbitals on C/O atoms

sp 2 Hybridization and Double Bonds σ (“sigma”) bonds join the atoms together in single OR multiple bonds. n In a multiple bond, π bonds are composed of overlapping regular p orbitals parallel to the main σ bond. n
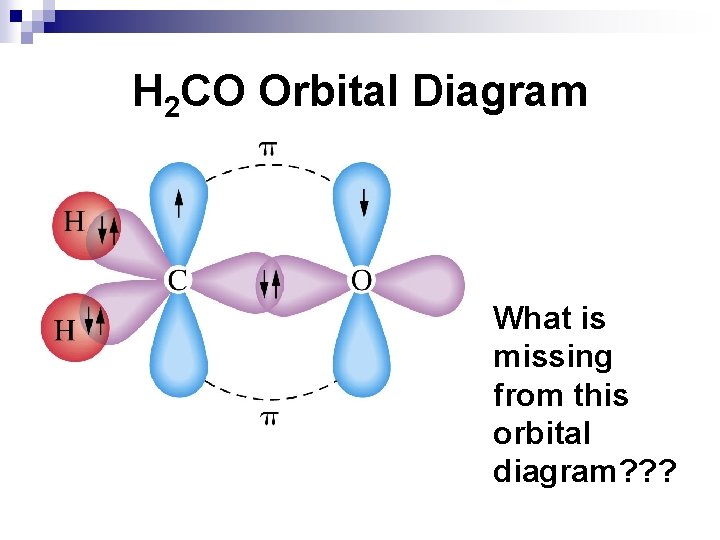
H 2 CO Orbital Diagram What is missing from this orbital diagram? ? ?

sp Hybridization How many effective pairs would be on the atom? n If we are still examining C, which electrons (and orbitals) are involved in bonding? n So what would the new orbitals be AFTER hybridization? n Example: acetylene (ethyne) C 2 H 2 n

dsp 3 Hybridization How many effective pairs on the atom? n Why can’t we use C as an example? n Give me an element we CAN use. n Example 1: (using element provided) n Example 2: I 3 -1 n Determine the BEST Lewis structure based on formal charges. ¨ Examine the bond types on each I atom. ¨

6 Effective Pairs What would the hybridization be? n Give an example of a molecule you have “seen” that has 6 effective pairs. n
- Slides: 13