TUAT Growth of chalcopyrite type magnetic semiconductors K
















![EDX Mn flux [Torr] Sample#1 Sample#2 2. 0× 10 Ge flux [Torr] -8 -8 EDX Mn flux [Torr] Sample#1 Sample#2 2. 0× 10 Ge flux [Torr] -8 -8](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-38.jpg)
![Sample#3 Mn flux [Torr] Ge flux [Torr] TBP flow rate [sccm] Growth Temp. [℃] Sample#3 Mn flux [Torr] Ge flux [Torr] TBP flow rate [sccm] Growth Temp. [℃]](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-39.jpg)


![Intensity [cps] XRD Patterns 10 6 10 5 10 4 10 3 10 2 Intensity [cps] XRD Patterns 10 6 10 5 10 4 10 3 10 2](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-42.jpg)

- Slides: 46

TUAT Growth of chalcopyrite type magnetic semiconductors K. Sato, T. Ishibashi, V. Smirnov, H. Yuasa, J. Jogo, T. Nagatsuka, Y. Kangawa and A. Koukitu

Scope of this talk • Brief summary of previous studies of chalcopyrite type magnetic semiconductors • Results of in-situ photoelectron spectroscopy • Suggested existence of chalcopyrite Mn. Ge. P 2 • Thermodynamic analysis • MOMBE growth of Mn. Ge. P 2 • Characterization

Brief summary of previous studies of chalcopyrite type magnetic semiconductors • We have been working with Mn-substituted chalcopyrite type semiconductors Cd. Ge. P 2 and Zn. Ge. P 2, in which we have confirmed ferromagnetic behavior up to 423 K and 350 K, respectively. Magneto-optical effect was also observed. • These samples were obtained by deposition and subsequent diffusion of Mn to bulk single crystals of ternary compounds. • Ab-initio calculation suggests that Cd. Ge. P 2 system with vacancies or non-stoichiometric composition will lead to ferromagnetism although ferromagnetism is not favored in stoichiometric (Cd, Mn)Ge. P 2.

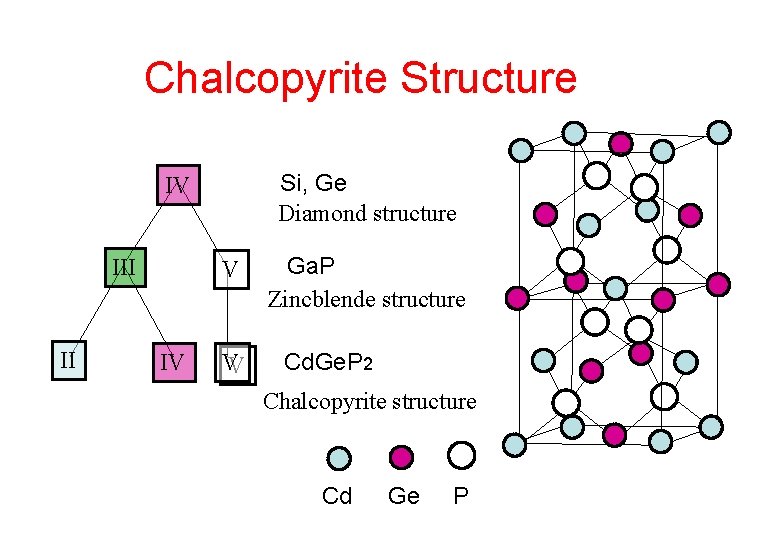
Chalcopyrite Structure Si, Ge Diamond structure IV III II V IV VV Ga. P Zincblende structure Cd. Ge. P 2 Chalcopyrite structure Cd Ge P

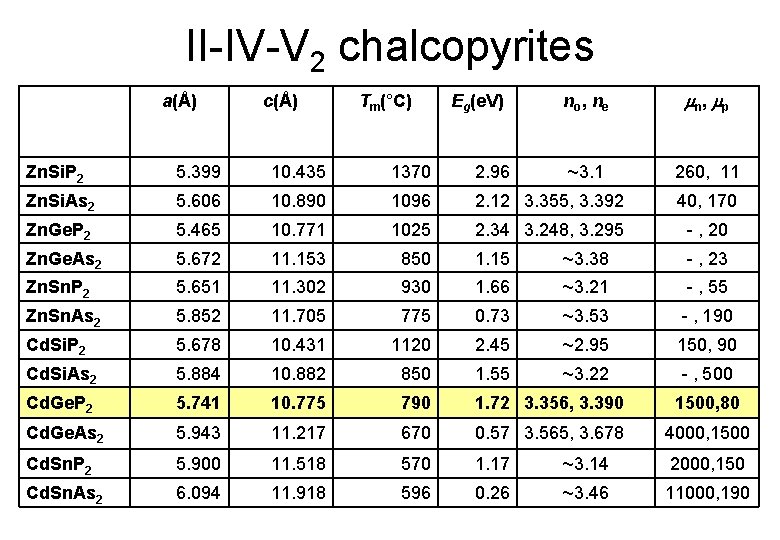
II-IV-V 2 chalcopyrites a(Å) c(Å) Tm(°C) Eg(e. V) n o, n e n, p ~3. 1 260, 11 Zn. Si. P 2 5. 399 10. 435 1370 2. 96 Zn. Si. As 2 5. 606 10. 890 1096 2. 12 3. 355, 3. 392 40, 170 Zn. Ge. P 2 5. 465 10. 771 1025 2. 34 3. 248, 3. 295 - , 20 Zn. Ge. As 2 5. 672 11. 153 850 1. 15 ~3. 38 - , 23 Zn. Sn. P 2 5. 651 11. 302 930 1. 66 ~3. 21 - , 55 Zn. Sn. As 2 5. 852 11. 705 775 0. 73 ~3. 53 - , 190 Cd. Si. P 2 5. 678 10. 431 1120 2. 45 ~2. 95 150, 90 Cd. Si. As 2 5. 884 10. 882 850 1. 55 ~3. 22 - , 500 Cd. Ge. P 2 5. 741 10. 775 790 1. 72 3. 356, 3. 390 1500, 80 Cd. Ge. As 2 5. 943 11. 217 670 0. 57 3. 565, 3. 678 4000, 1500 Cd. Sn. P 2 5. 900 11. 518 570 1. 17 ~3. 14 2000, 150 Cd. Sn. As 2 6. 094 11. 918 596 0. 26 ~3. 46 11000, 190

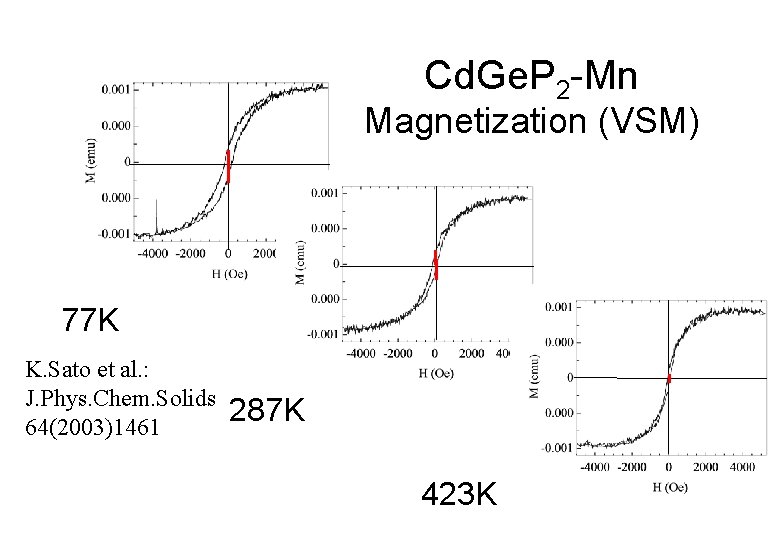
Cd. Ge. P 2 -Mn Magnetization (VSM) 77 K K. Sato et al. : J. Phys. Chem. Solids 64(2003)1461 287 K 423 K

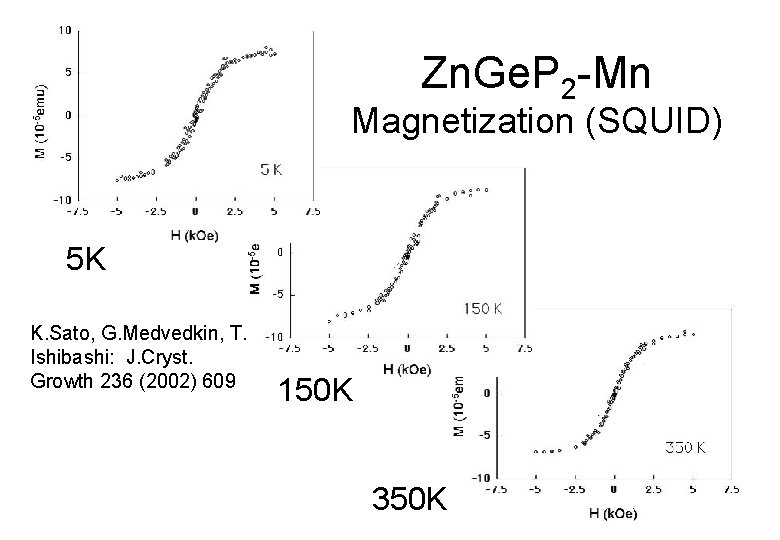
Zn. Ge. P 2 -Mn Magnetization (SQUID) 5 K K. Sato, G. Medvedkin, T. Ishibashi: J. Cryst. Growth 236 (2002) 609 150 K 350 K

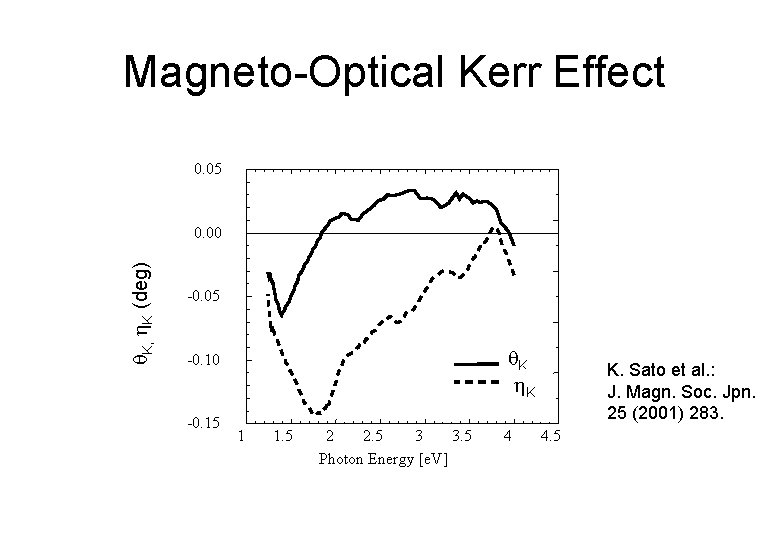
Magneto-Optical Kerr Effect 0. 05 K, K (deg) 0. 00 -0. 05 K K K -0. 10 -0. 15 1 1. 5 2 2. 5 3 3. 5 Photon Energy [e. V] 4 K. Sato et al. : J. Magn. Soc. Jpn. 25 (2001) 283. 4. 5

Previous preparation method for chalcopyritetype magnetic semiconductors • Mn was deposited on single crystals of Cd. Ge. P 2 and Zn. Ge. P 2 at Tsub 400 C, by which Mn was diffused into the bulk to substitute group II and IV cations. • During growth RHEED pattern of chalcopyrite structure seems to remain. • Mn-diffused crystals show ferromagnetism above room temperature.

II-IV-V 2 single crystals • Cd. Ge. P 2{112} • Directional freezing of the stoichiometric melt in a quartz ampoule or graphite crusible • Rate: 4 deg/h for 48 h • Highly compensated ntype • Prepared at Ioffe Inst. • Zn. Ge. P 2(001) • Vertical bridgeman technique • Bulk ingot of 28 mm and 150 mm in length • Highly compensated ptype • Prepared at Siberian Physico-Technical Inst.

Preparation of Mn-doped chalcopyrites II-IV-V 2 single crystal Host crystal: Cd. Ge. P 2, Zn. Ge. P 2 Mn II-IV-V 2 single crystal Mn-diffused layer II-IV-V 2 single crystal Mn deposition Tsub=RT to 380 -400°C Mn diffusion @T=300 -500°C

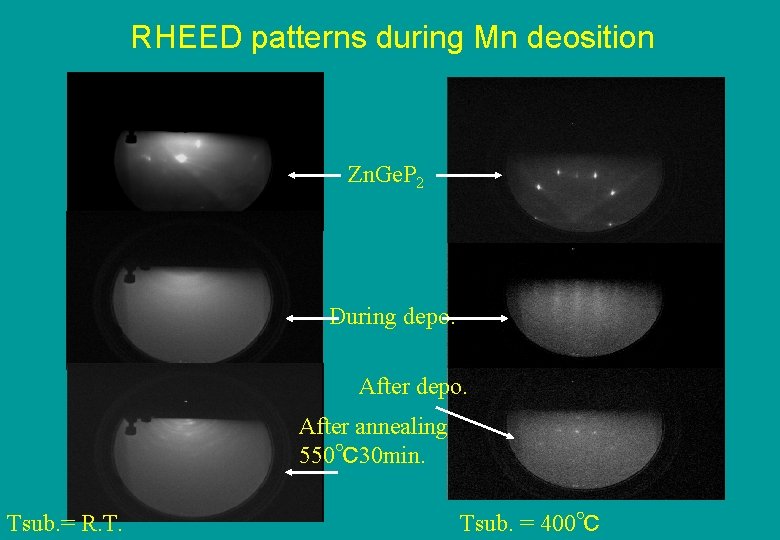
RHEED patterns during Mn deosition Zn. Ge. P 2 During depo. After annealing 550℃30 min. Tsub. = R. T. Tsub. = 400℃

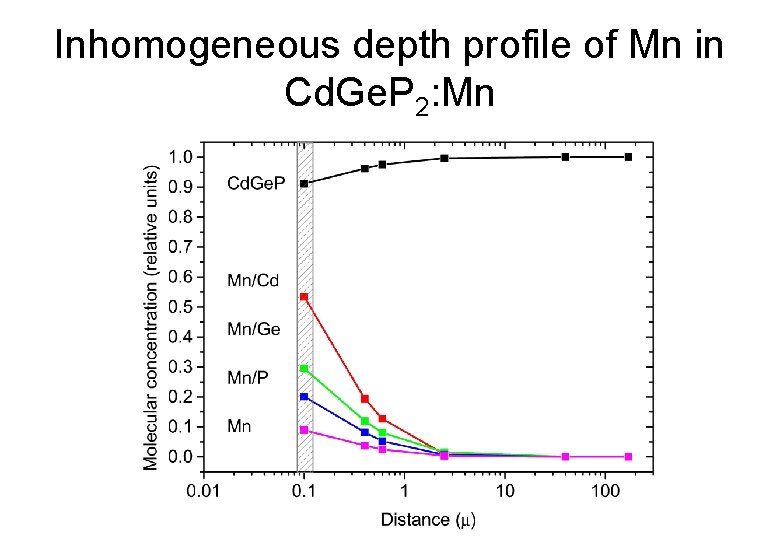
Problems • Inhomogeneous depth profile of Mn obtained by the deposition-diffusion technique. • Electrical properties of the surface shows a metallic behavior. • Preparation of homogeneously Mn-doped layer is necessary. Effort to obtain Cd. Ge. P 2: Mn thin films by MBE is proceeding

Inhomogeneous depth profile of Mn in Cd. Ge. P 2: Mn

Careful preparation necessary • Synthesis of bulk or powder Cd. Ge. P 2: Mn from constituent elements was tried. However, It was difficult to prevent formation of second phase compounds. • In bulk Zn. Ge. P 2: Mn prepared at elevated temperature, room-temperature ferromagnetism is suspected as due to Mn. P precipitated in the material. • Careful preparation of films with homogeneous distribution of Mn is strongly required.

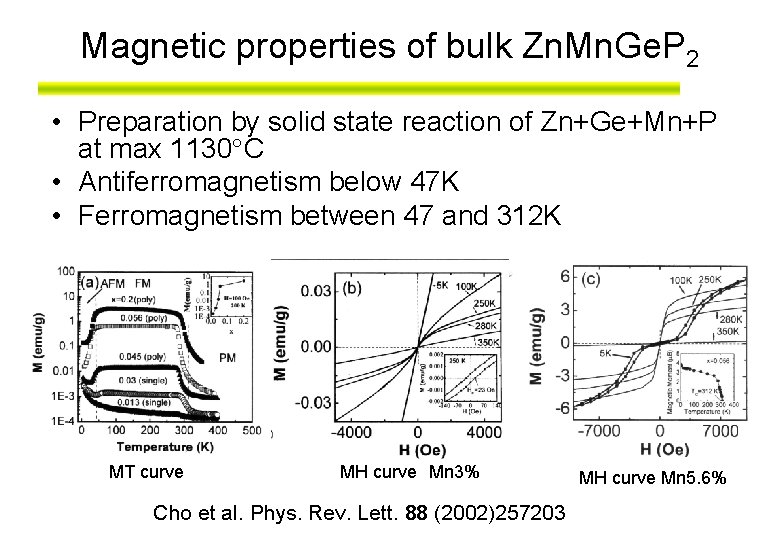
Magnetic properties of bulk Zn. Mn. Ge. P 2 • Preparation by solid state reaction of Zn+Ge+Mn+P at max 1130 C • Antiferromagnetism below 47 K • Ferromagnetism between 47 and 312 K MT curve MH curve Mn 3% Cho et al. Phys. Rev. Lett. 88 (2002)257203 MH curve Mn 5. 6%

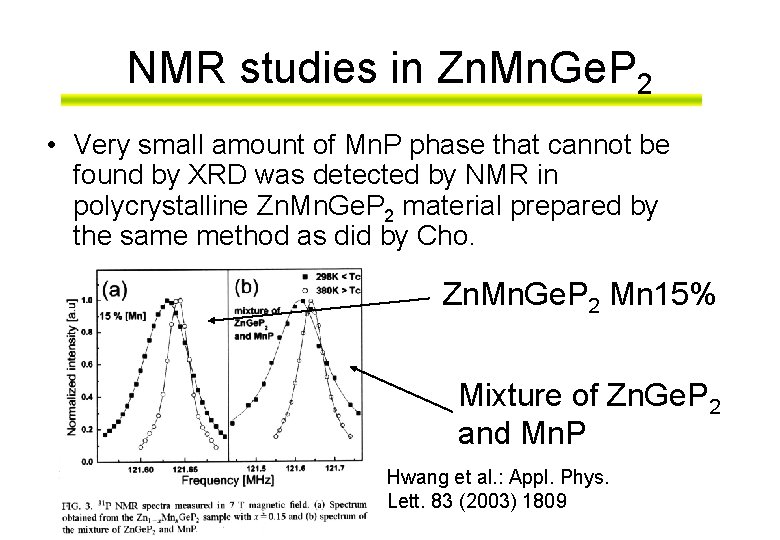
NMR studies in Zn. Mn. Ge. P 2 • Very small amount of Mn. P phase that cannot be found by XRD was detected by NMR in polycrystalline Zn. Mn. Ge. P 2 material prepared by the same method as did by Cho. Zn. Mn. Ge. P 2 Mn 15% Mixture of Zn. Ge. P 2 and Mn. P Hwang et al. : Appl. Phys. Lett. 83 (2003) 1809

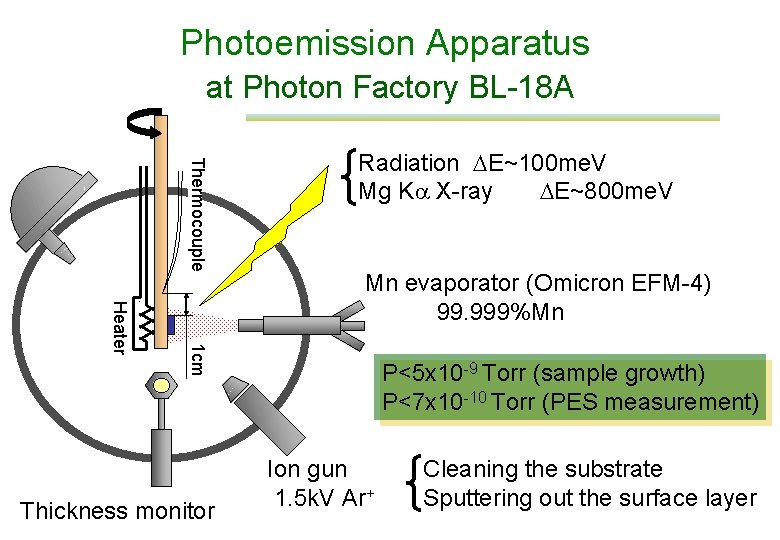
In-situ photoelectron spectroscopy • Photoelectron spectrometer with MBE system With Ar-ion etching device • Synchrotron radiation: Photon Factory BL-18 A • Specimen: Zn. Ge. P 2 single crystal, polished and etched • Deposit Mn and interrupt to measure PES • After deposition of 50 nm Mn, sputter-etched by Ar-ion and at each stage PES was measured

Photoemission Apparatus at Photon Factory BL-18 A Thermocouple Radiation E~100 me. V Mg Ka X-ray E~800 me. V 1 cm Heater Mn evaporator (Omicron EFM-4) 99. 999%Mn Thickness monitor P<5 x 10 -9 Torr (sample growth) P<7 x 10 -10 Torr (PES measurement) Ion gun 1. 5 k. V Ar+ Cleaning the substrate Sputtering out the surface layer

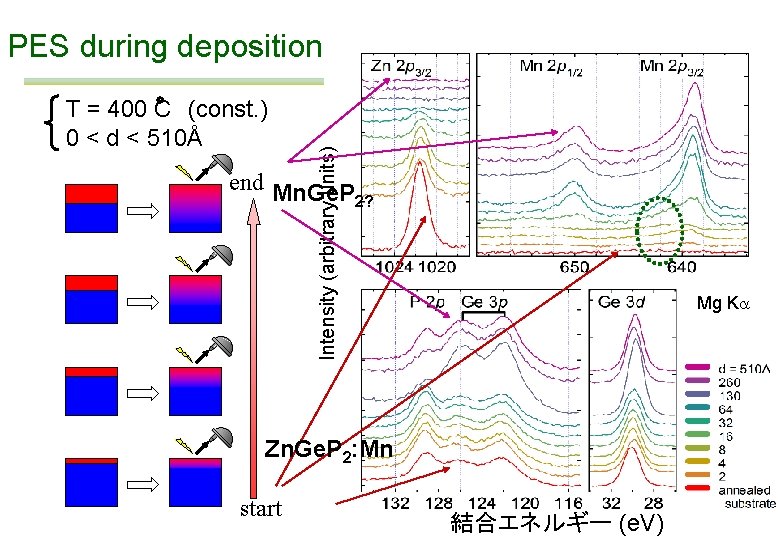
T = 400 C (const. ) 0 < d < 510Å Intensity (arbitrary units) PES during deposition end Mn. Ge. P 2? Mg Ka Zn. Ge. P 2: Mn start 結合エネルギー (e. V)

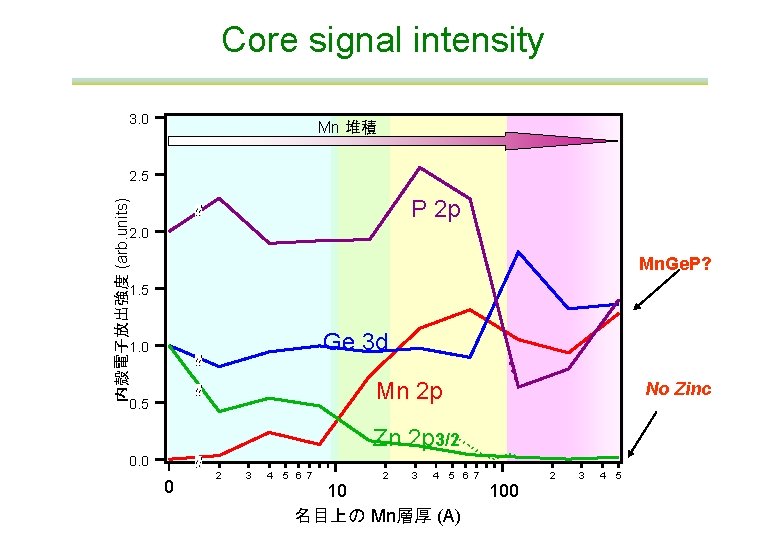
Core signal intensity 3. 0 Mn 堆積 2. 5 内殻電子放出強度 (arb. units) P 2 p 2. 0 Mn. Ge. P? 1. 5 Ge 3 d 1. 0 Mn 2 p 0. 5 No Zinc Zn 2 p 3/2 0. 0 1 0 2 3 4 5 6 7 10 名目上の Mn層厚 (A) 100 2 3 4 5

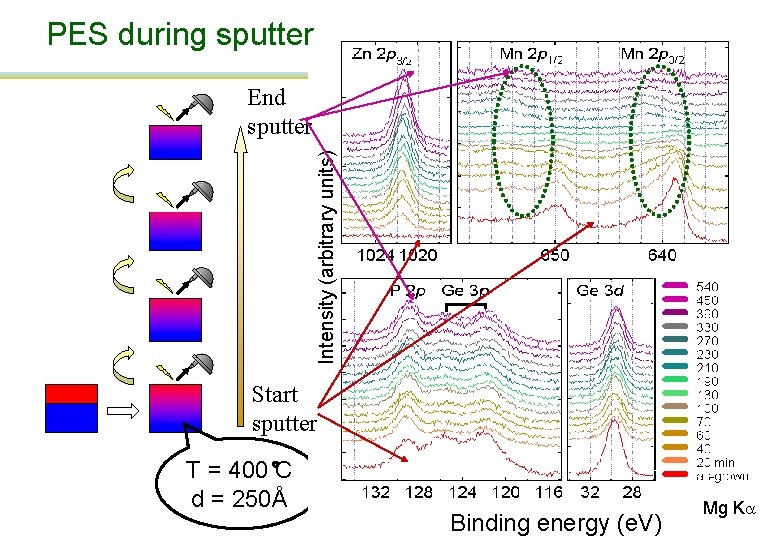
PES during sputter Intensity (arbitrary units) End sputter Start sputter T = 400 C d = 250Å Binding energy (e. V) Mg Ka

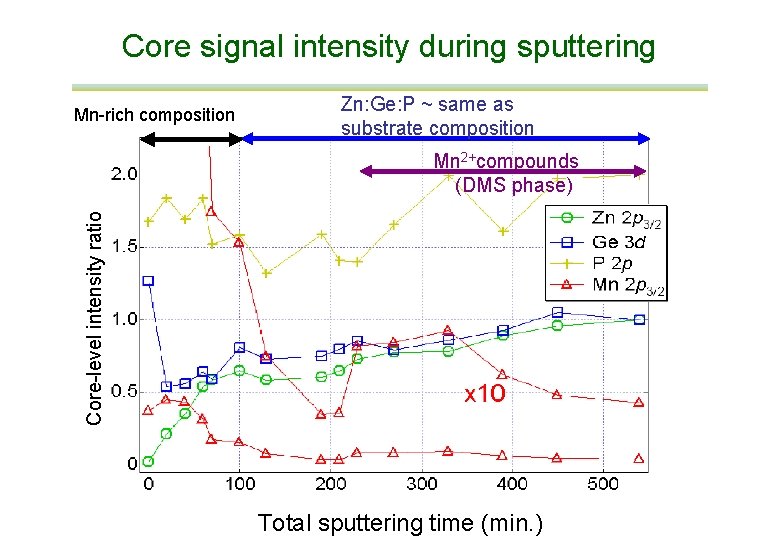
Core signal intensity during sputtering Mn-rich composition Zn: Ge: P ~ same as substrate composition Core-level intensity ratio Mn 2+compounds (DMS phase) Total sputtering time (min. )

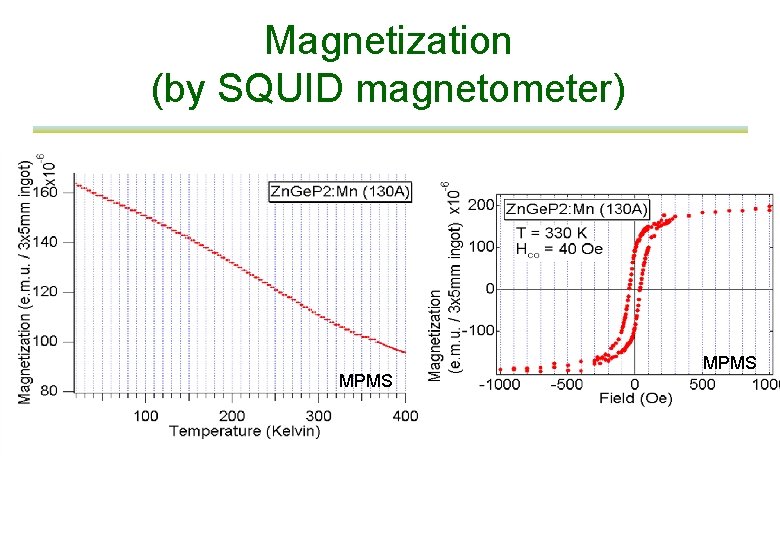
Magnetization (by SQUID magnetometer) MPMS

Suggested existence of chalcopyrite Mn. Ge. P 2 • Photoemission The surface composition is Mn. Ge. P 2 • RHEED pattern of initial chalcopyrite structure remained during growth Is chalcopyrite-type Mn. Ge. P 2 really exist?

MOMBE growth of Mn. Ge. P 2 • We applied MOMBE technique to obtain Mn. Ge. P 2 films on Ga. As substrate. • Mn and Ge are supplied from solid state source using K-cells • As P source, TBP (tertiary butyl phosphine) MO source is employed. • TBP is cracked to form P 2 and P 4 using cracking cell at 813 C

Thermodynamic analysis for MOMBE growth of Mn. Ge. P 2 • To know whether Mn. Ge. P 2 can be obtained as a stable compound using the MOMBE technique, thermodynamic analysis is performed.

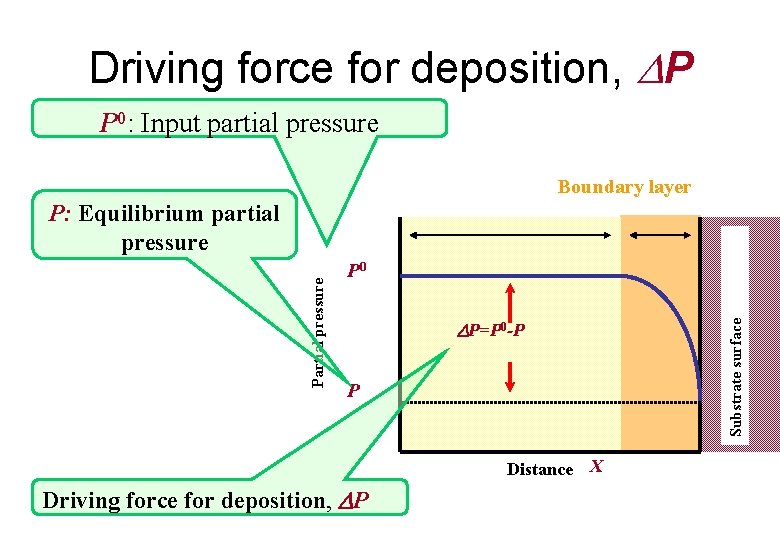
Driving force for deposition • In thermodynamic analysis, we used parameters of driving force for deposition P, Input partial pressure, P 0, and equilibrium partial pressure at vapor-solid interface, P. • Here, driving force for deposition P is the difference between input partial pressure and equilibrium partial pressure : P=P 0 -P; where P 0 is input partial pressure, and P equilibrium partial pressure • Using these parameters, we can obtain Input mole ratio, RMn, and solid composition, x, as follows:

Driving force for deposition, P P 0: Input partial pressure Boundary layer P 0 P=P 0 -P P Distance X Driving force for deposition, P Substrate surface Partial pressure P: Equilibrium partial pressure

MOMBE • Here, we assume P 2 • Mn(g)+1/2 P 2(g) = Mn. P(s) molecule as a group. V source, because • Ge(g)+1/2 P 2(g) = Ge. P(s) more than 80% of Conservation constraints TBP is cracked and 0 0 • PMn+Ge -PMn+Ge= 2(PP 2 -PP 2) changed to P 2 rather than P 4 at 813 C. • Pi = PMn+PGe+PP 2 Equilibrium equation for reaction using these equations equilibrium partial pressure, which is unknoun parameter, is calculated.

Ab-initio calculation of enthalpy of mixing • Enthalpy of mixing Hm Hm=EMn. Ge. P-{x. EMn. P+(1 -x)EGe. P} • Interaction parameter = Hm/x(1 -x) • Solid composition x x= PMn/( PMn+ PGe) vs Input molar ratio of Mn RMn=P 0 Mn/(P 0 Mn+P 0 Ge)

enthalpy of mixing of (Mn, Ge)P as a function of solid composition • The function, Hm, is estimated from the ab initio total energy calculations for structure models.

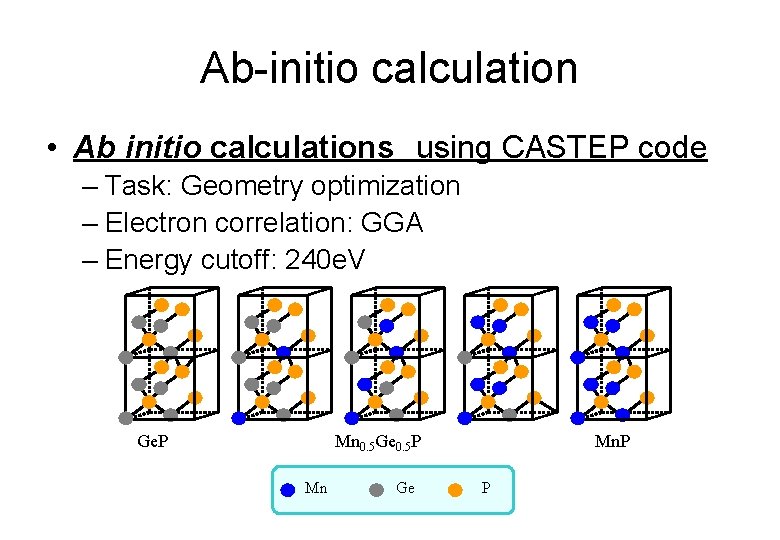
Ab-initio calculation • Ab initio calculations using CASTEP code – Task: Geometry optimization – Electron correlation: GGA – Energy cutoff: 240 e. V Ge. P Mn 0. 5 Ge 0. 5 P Mn Ge P

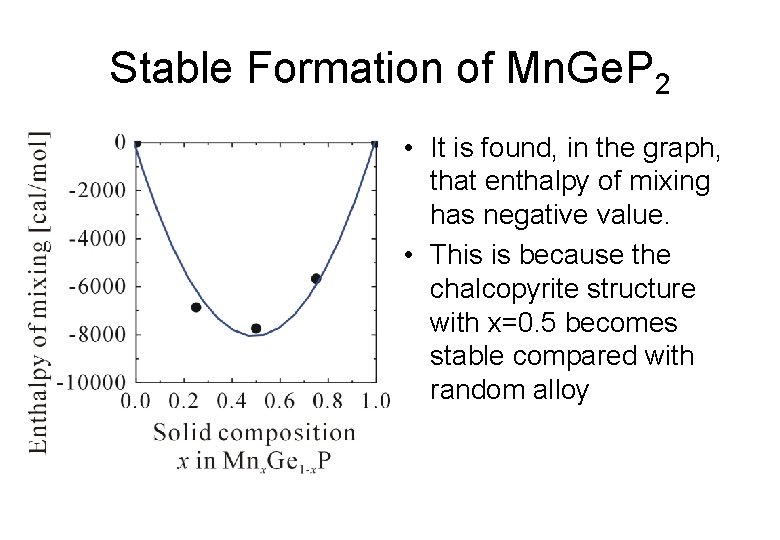
Stable Formation of Mn. Ge. P 2 • It is found, in the graph, that enthalpy of mixing has negative value. • This is because the chalcopyrite structure with x=0. 5 becomes stable compared with random alloy

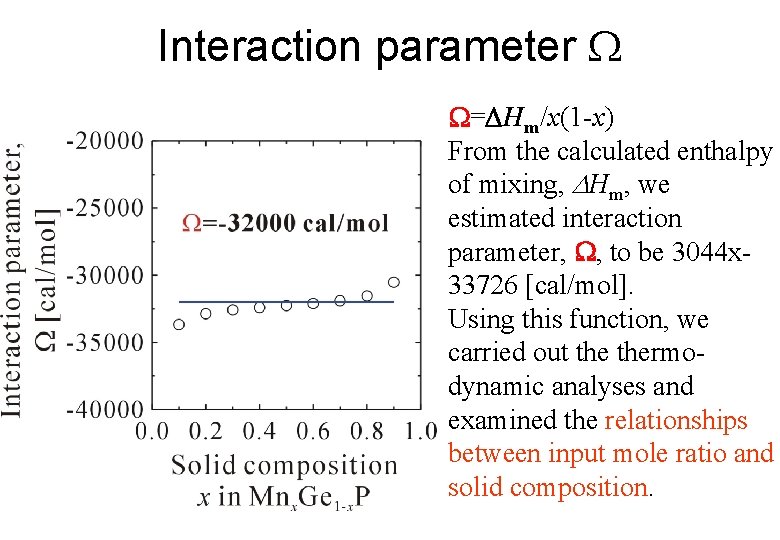
Interaction parameter W=DHm/x(1 -x) From the calculated enthalpy of mixing, Hm, we estimated interaction parameter, W, to be 3044 x 33726 [cal/mol]. Using this function, we carried out thermodynamic analyses and examined the relationships between input mole ratio and solid composition.

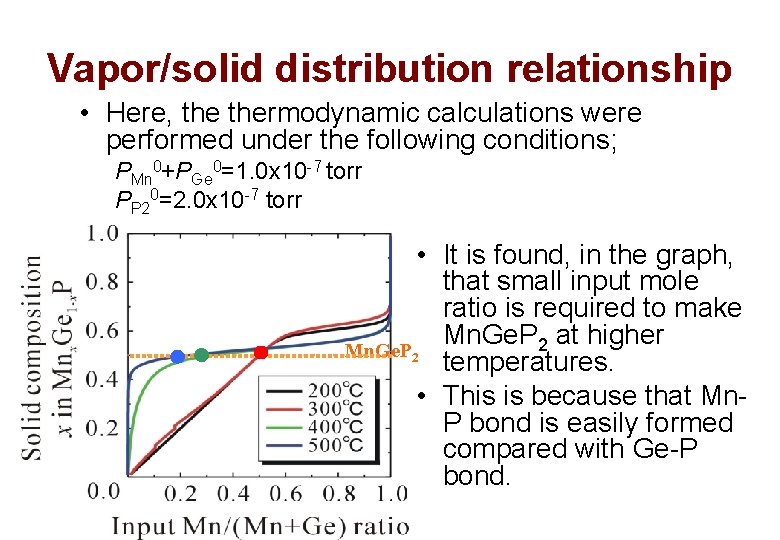
Vapor/solid distribution relationship • Here, thermodynamic calculations were performed under the following conditions; PMn 0+PGe 0=1. 0 x 10 -7 torr PP 20=2. 0 x 10 -7 torr • It is found, in the graph, that small input mole ratio is required to make Mn. Ge. P 2 at higher Mn. Ge. P 2 temperatures. • This is because that Mn. P bond is easily formed compared with Ge-P bond.

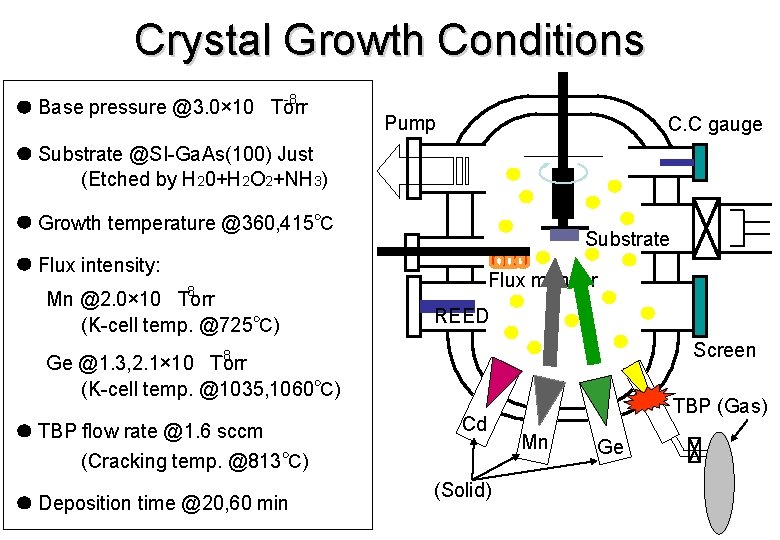
Crystal Growth Conditions -8 Base pressure @3. 0× 10 Torr Pump C. C gauge Substrate @SI-Ga. As(100) Just (Etched by H 20+H 2 O 2+NH 3) Growth temperature @360, 415℃ Substrate Flux intensity: Flux monitor -8 Mn @2. 0× 10 Torr (K-cell temp. @725℃) REED Screen -8 Ge @1. 3, 2. 1× 10 Torr (K-cell temp. @1035, 1060℃) TBP flow rate @1. 6 sccm (Cracking temp. @813℃) Deposition time @20, 60 min Cd (Solid) TBP (Gas) Mn Ge
![EDX Mn flux Torr Sample1 Sample2 2 0 10 Ge flux Torr 8 8 EDX Mn flux [Torr] Sample#1 Sample#2 2. 0× 10 Ge flux [Torr] -8 -8](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-38.jpg)
EDX Mn flux [Torr] Sample#1 Sample#2 2. 0× 10 Ge flux [Torr] -8 -8 2. 1× 10 1. 3× 10 -8 -8 TBP flow rate [sccm] Growth Temp. [℃] Depo. time [min] 1. 6 360 20 #1 Mn Ge P 0. 99 1. 00 2. 67 Mn Ge P 2. 09 1. 00 5. 20 #2
![Sample3 Mn flux Torr Ge flux Torr TBP flow rate sccm Growth Temp Sample#3 Mn flux [Torr] Ge flux [Torr] TBP flow rate [sccm] Growth Temp. [℃]](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-39.jpg)
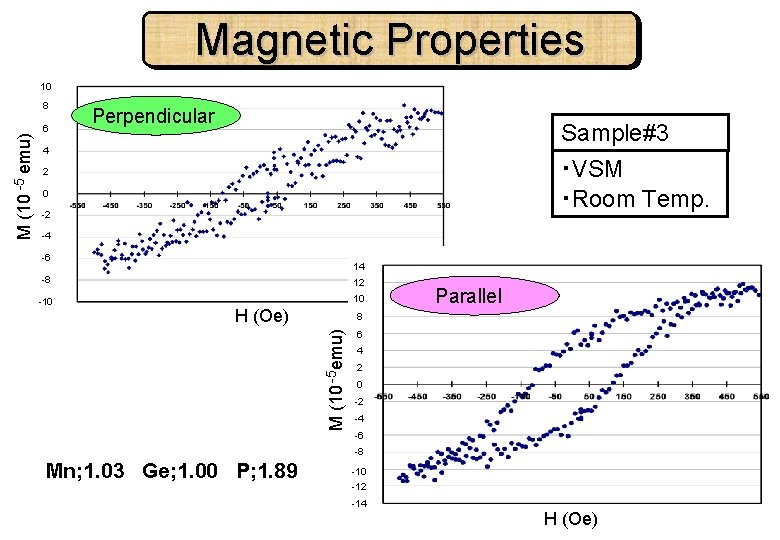
Sample#3 Mn flux [Torr] Ge flux [Torr] TBP flow rate [sccm] Growth Temp. [℃] Depo. time [min] 2. 0× 10 -8 1. 6 415 60 #3 Mn Ge P 1. 03 1. 00 1. 89

SEM Observation Sample#1 400 nm Sample#2 300 nm Sample#3 400 nm

Sample#1 800 nm Sample#2 800 nm Sample#3 800 nm
![Intensity cps XRD Patterns 10 6 10 5 10 4 10 3 10 2 Intensity [cps] XRD Patterns 10 6 10 5 10 4 10 3 10 2](https://slidetodoc.com/presentation_image_h/31533ec58617813eef9bcb47b4e86a6a/image-42.jpg)
Intensity [cps] XRD Patterns 10 6 10 5 10 4 10 3 10 2 Sample#3 Sample#1 Sample#2 65. 6 65. 8 66 66. 2 2θ [deg] 66. 4

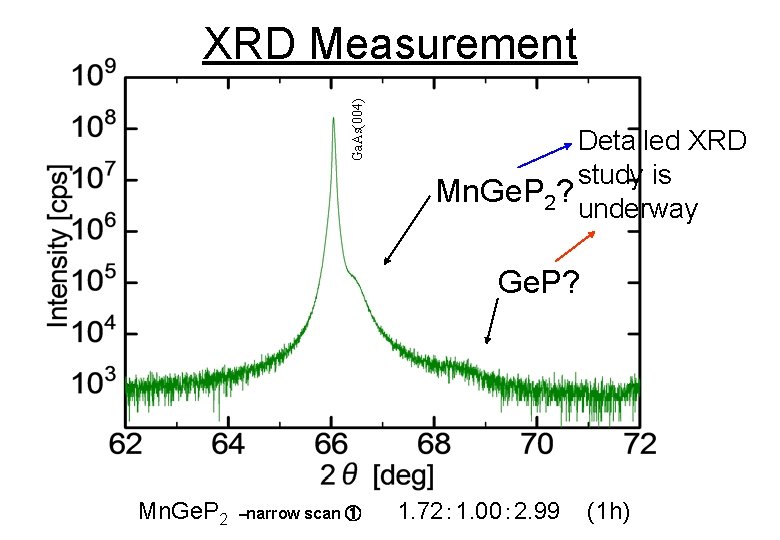
Ga. As(004) XRD Measurement Detailed XRD study is Mn. Ge. P 2? underway Ge. P? Mn. Ge. P 2 –narrow scan ① 1. 72: 1. 00: 2. 99 (1 h)

Magnetic Properties 10 6 Perpendicular Sample#3 4 ・VSM ・Room Temp. 2 0 -2 -4 -6 14 -8 12 -10 10 H (Oe) Parallel 8 M (10 -5 emu) 8 6 4 2 0 -2 -4 -6 -8 Mn; 1. 03 Ge; 1. 00 P; 1. 89 -10 -12 -14 H (Oe)

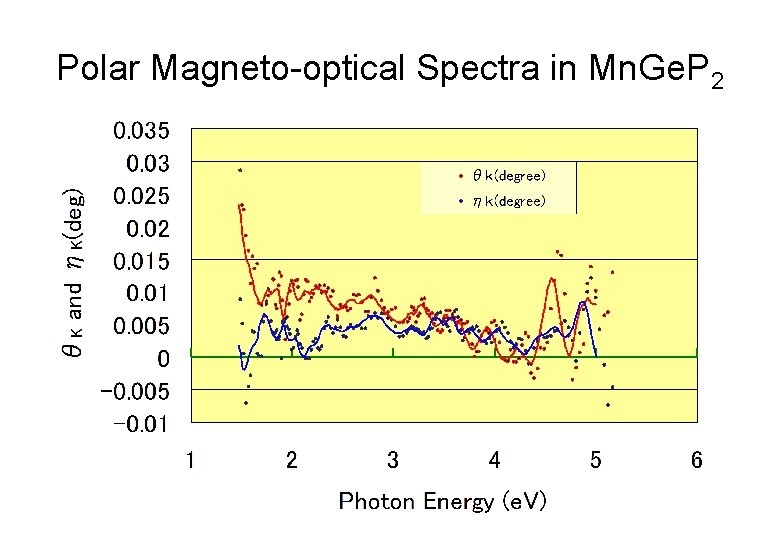
Polar Magneto-optical Spectra in Mn. Ge. P 2

Summary • As one of approaches to elucidate the origin of room temperature ferromagnetism in magnetic chalcopyrites, growth of chalcopyrite type Mn. Ge. P 2, which is not existing in nature is investigated. • Thermodynamic study including ab-initio evaluation of Hm confirms stable formation of Mn. Ge. P 2 by MBE technique. • MOMBE growth of Mn. Ge. P 2 films are studied. Nearly stoichiometric compounds are obtained. They show ferromagnetism and weak magneto-optical effect. • Further careful investigation is necessary to discriminate the effect of possible second phase material.