Total Synthesis of Rapamycin Isolation and Structure Determination











































- Slides: 45

Total Synthesis of Rapamycin Isolation and Structure Determination: Vézina, C. ; Kudelski, A. ; Sehgal, S. N. J. Antibiotics 1975, 28, 721. Swindells, D. C. N. ; White, P. S. ; Findlay, J. A. Can. J. Chem. 1978, 56, 2491. Findlay, J. A. ; Radics, L. Can. J. Chem. 1981, 59, 49. Mc. Alpine, J. B. ; Swanson, S. J. ; Jackson, M. ; Whittern, D. N. J. Antibiotics 1991, 44, C-3. Total Syntheses: Nicolaou, K. C. ; Chakraborty, T. K. ; Piscopio, A. D. ; Minowa, N. ; Bertinato, P. J. Am. Chem. Soc. 1993, 115, 4419. Hayward, C. M. ; Yohannes, D. ; Danishefsky, S. J. J. Am. Chem. Soc. 1993, 115, 9345. Romo, D. ; Meyer, S. D. ; Johnson, D. D. ; Schreiber, S. L. J. Am. Chem. Soc. 1993, 115, 7906. Smith, A. B. , III; Condon, S. M. ; Mc. Cauley, J. A. ; Leazer, J. L. , Jr. ; Leahy, J. W. ; Maleczka, R. E. , Jr. J. Am. Chem. Soc. 1995, 117, 5407 -5408.

Immunomodulators rapamycin FK-506 cyclosporin A

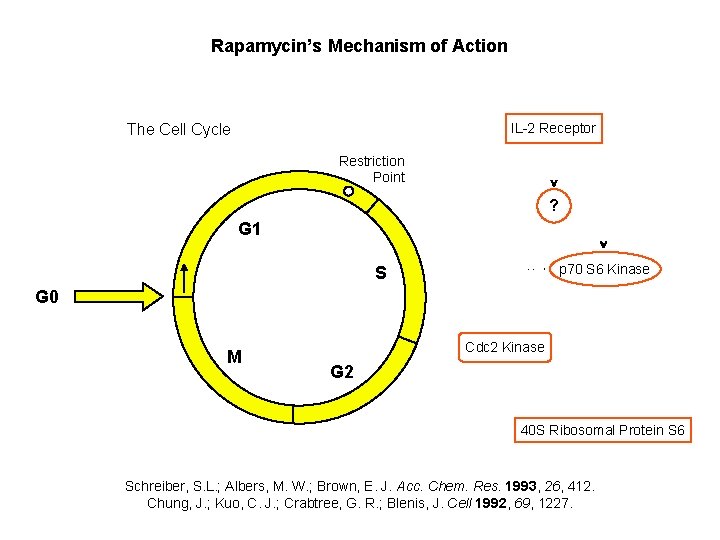
Rapamycin’s Mechanism of Action IL-2 Receptor The Cell Cycle Restriction Point ? G 1 p 70 S 6 Kinase S G 0 M Cdc 2 Kinase G 2 40 S Ribosomal Protein S 6 Schreiber, S. L. ; Albers, M. W. ; Brown, E. J. Acc. Chem. Res. 1993, 26, 412. Chung, J. ; Kuo, C. J. ; Crabtree, G. R. ; Blenis, J. Cell 1992, 69, 1227.

KCN's Retrosynthetic Analysis of Rapamycin rapamycin

Synthesis of Oxazolidone A

Synthesis of Oxazolidone A (continued)

KCN's Retrosynthetic Analysis of Rapamycin rapamycin

Synthesis of Subunit B Z-enolate

Synthesis of Subunit B (continued)

KCN's Retrosynthetic Analysis of Rapamycin rapamycin

Synthesis of Vinyliodide D

Synthesis of Vinyliodide D (continued)

KCN's Retrosynthetic Analysis of Rapamycin rapamycin

The Union of A + B + E

Elaboration of EAB

The Introduction of D rapamycin EABD

The End Game – Tricarbonyl Formation Note: the first HF step removes the TES groups and the second HF step removes the TIPS groups

The End Game – The “Stitching” Stille Reaction rapamycin

Summary • Completed the first total synthesis of (-)-rapamycin. – The longest linear sequence from an article of commerce consists of thirty-seven steps. – The longest linear sequence from our five sub-targets is sixteen steps. – Total steps: 102 • Instructional applications of the Stille reaction, oxidation chemistry, chiral auxiliaries, organosilicons, protective groups, etc.

Smith’s Retrosynthetic Analysis of Rapamycin and Demethoxyrapamycin

Synthesis of Iodide A

Synthesis of Dithiane B

Synthesis of Dithiane C

Retrosynthetic Analysis of Rapamycin

Synthesis of the Ortho Ester Exploitation of Alternate Ortho Ester Diastereomer Employed in Smith’s Latrunculin Synthetic Venture

Synthesis of the E and Z Eneynes

Mechanism of Olefin Isomerization

Stereochemistry of Eneyne Addition to Aldehyde

Synthesis of Dienylstannane D

Retrosynthetic Analysis of Rapamycin

Construction of a C 27 -C 42 Aldehyde

Construction of the C 22 -C 42 Subunit

Synthesis of Demethoxyrapamycin: Construction of Advanced ABC Intermediate

Retrosynthetic Analysis of Rapamycin and Demethoxyrapamycin: Introduction of the Tricarbonyl Segment

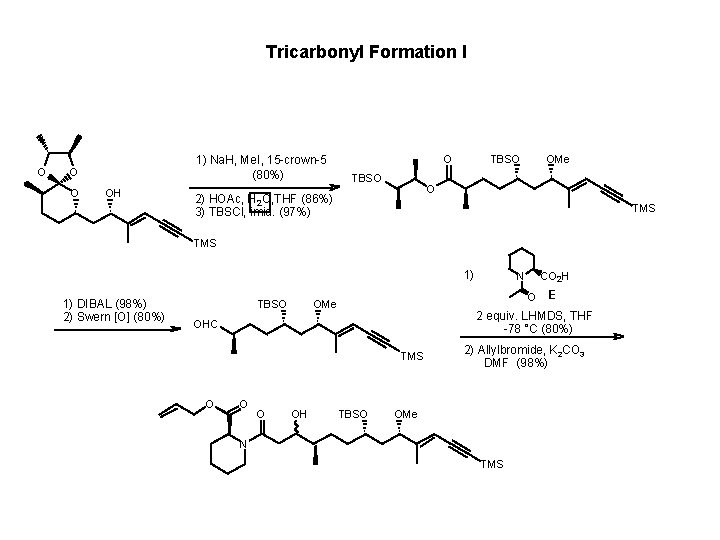
Tricarbonyl Formation I O 1) Na. H, Me. I, 15 -crown-5 (80%) O O OH O TBSO OMe O 2) HOAc, H 2 O, THF (86%) 3) TBSCl, imid. (97%) TMS 1) 1) DIBAL (98%) 2) Swern [O] (80%) TBSO O OH E 2 equiv. LHMDS, THF -78 °C (80%) TMS O CO 2 H O OMe OHC O N TBSO 2) Allylbromide, K 2 CO 3 DMF (98%) OMe N TMS

Tricarbonyl Formation II

Pipecolinyl Acylation

Proposed Endgame: Bis-Hydrostannylation

Attempted Macrocyclizations

Preparation of ABC vinylstannane & DE vinyl iodide

Proposed Endgame Strategy for the Total Synthesis of Rapamycin and Demethoxyrapamycin

Macrocyclization

Demethoxyrapamycin

Rapamycin

Summary • Developed a highly convergent and efficient total synthesis of (-)-rapamycin. – The longest linear sequence from an article of commerce consists of thirty-three steps. – The longest linear sequence from our five sub-targets is fourteen steps. – After the coupling of the C(1)-C(20) fragment to the C(22)C(42) fragment only three steps are required to complete the synthesis. • Completed the first total synthesis of demethoxyrapamycin. – The synthesis serves as a structure proof. – The synthesis establishes our unified synthetic approach as being amenable to the preparation of analogs.