Total Acid Number Determination Total acid number of

Total Acid Number Determination Ø Total acid number of biodiesel Ø Distilled biodiesel experiment Ø Ultra low sulfur diesel (ULSD) experiment 1

Total Acid Number l Definition The mass of potassium hydroxide (KOH) in milligrams that is required to neutralize the acids in one gram of oil. l Formula Veq : Titrant consumed by sample+spiking solution beq : Tiltrant consumed by spiking solution N: Molarity of titrant 2

Factors Contributing to the TAN l FFA l Esters l Catalyst oxides l Salts of heavy metals l Additives such as inhibitors and detergents …… 3

Potentiometric Titration ü The sample is dissolved in toluene and propanol with a little water ü Titrated with alcoholic potassium hydroxide ü A glass electrode and reference electrode is connected to a voltmeter/potentiometer. ü The meter reading (in millivolts) is plotted against the volume of titrant. ü The end point is taken at the distinct inflection of the resulting curve corresponding to the basic buffer solution. 4
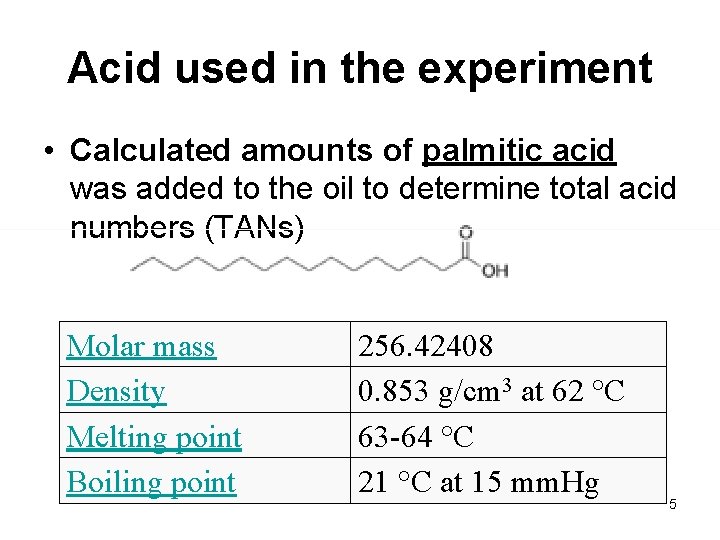
Acid used in the experiment • Calculated amounts of palmitic acid was added to the oil to determine total acid numbers (TANs) Molar mass Density Melting point Boiling point 256. 42408 0. 853 g/cm 3 at 62 °C 63 -64 °C 21 °C at 15 mm. Hg 5
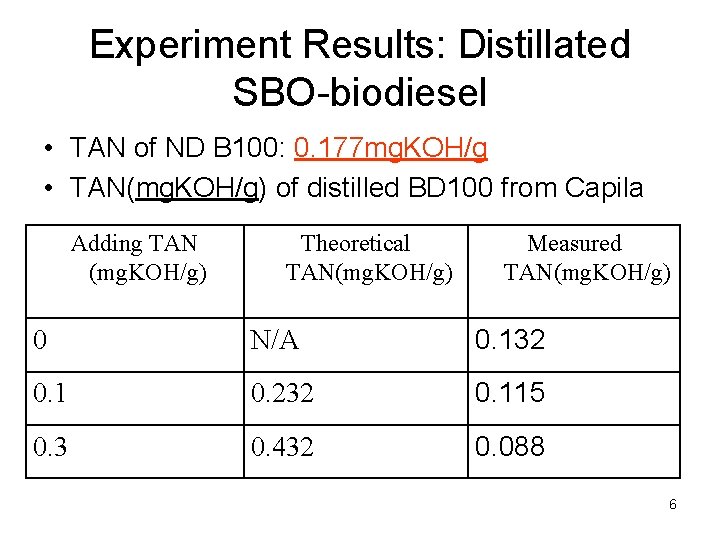
Experiment Results: Distillated SBO-biodiesel • TAN of ND B 100: 0. 177 mg. KOH/g • TAN(mg. KOH/g) of distilled BD 100 from Capila Adding TAN (mg. KOH/g) Theoretical TAN(mg. KOH/g) Measured TAN(mg. KOH/g) 0 N/A 0. 132 0. 1 0. 232 0. 115 0. 3 0. 432 0. 088 6
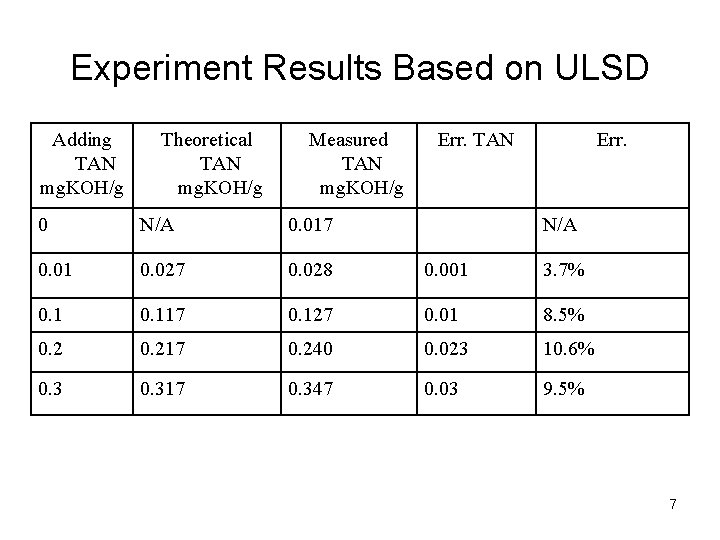
Experiment Results Based on ULSD Adding TAN mg. KOH/g Theoretical TAN mg. KOH/g Measured TAN mg. KOH/g Err. TAN Err. 0 N/A 0. 017 N/A 0. 01 0. 027 0. 028 0. 001 3. 7% 0. 117 0. 127 0. 01 8. 5% 0. 217 0. 240 0. 023 10. 6% 0. 317 0. 347 0. 03 9. 5% 7

Current Work • Synthetic antioxidants by GC-MS • Acid number • Writing proposal • Catalyst activities 8
- Slides: 8