The Use of Non Profit Industrial Consortia to





![Why Industrial Biomedical Consortia [IBCs]? • Fill a research gap, not being effectively filled Why Industrial Biomedical Consortia [IBCs]? • Fill a research gap, not being effectively filled](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-8.jpg)
![Common Characteristics (TSC/SAEC) • Defined project finite timeframe • Non-Profit [501 c 3] > Common Characteristics (TSC/SAEC) • Defined project finite timeframe • Non-Profit [501 c 3] >](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-10.jpg)




![i. SAEC Organization and Committees BOD Genotyping Core [Expression Analysis, Inc] Scientific/Clinical SMC SAE i. SAEC Organization and Committees BOD Genotyping Core [Expression Analysis, Inc] Scientific/Clinical SMC SAE](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-16.jpg)






![Learning #3 Develop a high-quality plan [before recruiting membership] • Well organized “formation phase” Learning #3 Develop a high-quality plan [before recruiting membership] • Well organized “formation phase”](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-24.jpg)








![Acknowledgments SMC n Ann Daly & DILIGEN [Newcastle] n Mariam Molokhia & EUDRAGENE [London] Acknowledgments SMC n Ann Daly & DILIGEN [Newcastle] n Mariam Molokhia & EUDRAGENE [London]](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-33.jpg)


- Slides: 35

The Use of Non Profit, Industrial Consortia to Improve Pharmaceutical Industry R & D “Lessons Learned” Wellcome Trust’s “Pre-Competitive Boundaries and Open Innovation in Drug Discovery and Development” Meeting Arthur Holden Founder, Chairman, and CEO

Introduction • Executive not scientist • 30 years of leadership experience – commercial & non-profit consortia; start-ups to mid size to large corporations – – Baxter International (1984 -1994) Celsis International (1994 -1998) First Genetic Trust (2000 -2006) Illumina (2006 -2007) • Formed and managed numerous large-scale consortia – The SNP Consortium, Ltd. (1999 -2002) – The International SAE Consortium, Ltd. (2007 - 2012) – Supported many others (MSC, Hap. Map, etc. ) 2

My Comments • Consortia Context – Model – TSC – SAEC • Some Lessons Learned 3

Context

“Pharmaceuticals” Context § Declining Productivity & novel therapeutics § Increasing Costs & Competition at “Market Entry” § “Block Buster” mentality softening § Scale “Innovation” ? ? ? § > Healthcare pricing pressures in all core markets “pay for performance” paradigm of the future § End of “biotech risk subsidy” by the public markets etc, etc. Increasing R & D productivity is critical. 5

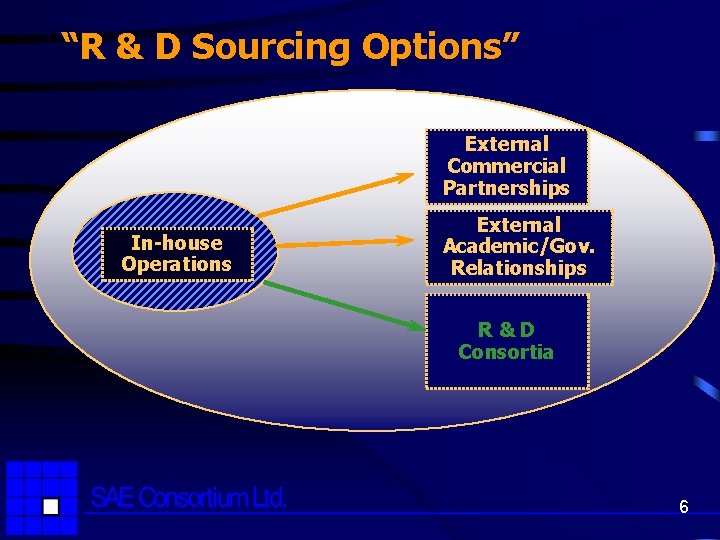
“R & D Sourcing Options” External Commercial Partnerships In-house Operations External Academic/Gov. Relationships R&D Consortia 6

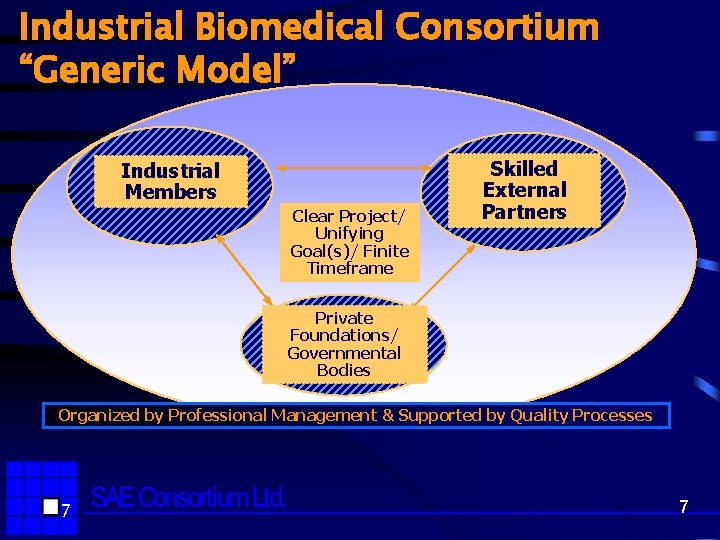
Industrial Biomedical Consortium “Generic Model” Industrial Members Clear Project/ Unifying Goal(s)/ Finite Timeframe Skilled External Partners Private Foundations/ Governmental Bodies Organized by Professional Management & Supported by Quality Processes 7 7
![Why Industrial Biomedical Consortia IBCs Fill a research gap not being effectively filled Why Industrial Biomedical Consortia [IBCs]? • Fill a research gap, not being effectively filled](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-8.jpg)
Why Industrial Biomedical Consortia [IBCs]? • Fill a research gap, not being effectively filled by existing players [government or private] • Establish common/often risky “research platforms”, which help all researchers, while meeting specific industrial scientific requirements [pre-competitive/pro-competitive] • Establish industry standards • Reduce legal/IP barriers • Gain greater efficiency/effectiveness (scale) via pooling of talents and resources across industry 8

Two Consortia The SNP Consortium 1999 -2002 The SAE Consortium 2007 -2012 9
![Common Characteristics TSCSAEC Defined project finite timeframe NonProfit 501 c 3 Common Characteristics (TSC/SAEC) • Defined project finite timeframe • Non-Profit [501 c 3] >](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-10.jpg)
Common Characteristics (TSC/SAEC) • Defined project finite timeframe • Non-Profit [501 c 3] > “public benefit orientation” – Open data access – No IP constraints • “Fixed, uniform” membership contributions • Designed for “leverage” and “time to result” • Collaboration with government (if advantageous to results) • Active (Committee) Participation and Investment by Members • “Governance by majority” 1 member/1 vote 10

SNP Consortium -- Mission & Results “The SNP Consortium worked to advance the field of medicine and the development of genetic based diagnostics and therapeutics, through the creation of a high quality, dense genome wide single nucleotide polymorphism (SNP) map, which is available to all parties at no cost. ” The Results: § Created a high quality HG SNP map for GWAS § Identify 300, 000 SNPs [ 2. 7 M Common SNPs] § Map 150, 000 SNPs [ 2. 5 M Common SNPs] § Broad, evenly spaced well annotated map within two years § Maximize public accessibility [~ 2. 4 M “unencumbered” SNPs] § Provided order and orientation data to assembled base WG project sequence and a significant “backbone” to the “Hap. Map” § $42 M project, which exceeded expectations with $9 M in budget favorability 11

Drivers of the SNP Consortium ü Industry standard SNP map strong industry support ü Universal access to base genomic/SNP data public domain (IP considerations) ü Facilitate pharmaco-genomics / genetics association, linkage & haplotyping studies ü Economics: § Favorable economy of scale>>cost & risk sharing § Favorable cost to access high quality SNP datasets 12

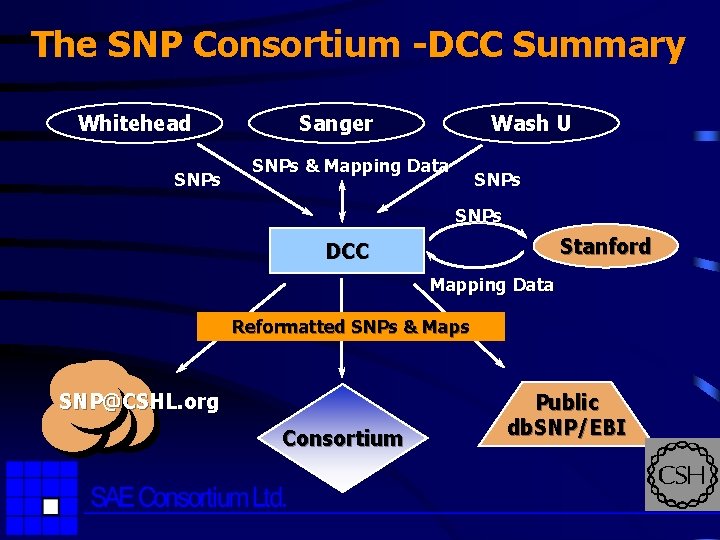
The SNP Consortium -DCC Summary Whitehead SNPs Sanger Wash U SNPs & Mapping Data SNPs Stanford DCC Mapping Data Reformatted SNPs & Maps SNP@CSHL. org Consortium Public db. SNP/EBI

i. SAEC -- Mission & Results “The i. SAEC will identify and validate DNA-variants useful in predicting the risk of drug induced serious adverse events. ” The Results (to date): § Initial GWAS for Drug Induced Serious Skin Rash identifying marker(s) of potential significance § Assembled 2 nd largest DILI cohort, merged with DILIN, completed initial GWAS identifying numerous significant marker(s) within/across drugs § Initial GWAS for Drug Induced PQT/Td. P, merged with Le. Dueq Network, and identified marker(s) of potential significance § Completed four public data releases (clinical and GWAS data) with FDA § > 200 genetic markers associated with DILI and SSR in the public domain. § Favorable to budget in phase by $2 M § Organized Phase 2 focused on “drug induced immunologic SAEs” associated with specific drugs and ethnicities, and forming novel SAE research channels vital to drug-specific SAE research. 14

Drivers of the i. SAEC ü Need to improve new product (safety) productivity ü Scale required to execute safety pharmacogenetics (top R & D management priority) common markers in public domain ü Need to standardize “SAE phenotypes” ü Need to develop new and innovative methods to source cases and controls ü Pooled technology assessment & risk (e. g. GWAS, WG sequencing) ü FDA’s Industrial Advisory Board recommendation ü $ resources required (poor public funding, given the health impacts) 15
![i SAEC Organization and Committees BOD Genotyping Core Expression Analysis Inc ScientificClinical SMC SAE i. SAEC Organization and Committees BOD Genotyping Core [Expression Analysis, Inc] Scientific/Clinical SMC SAE](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-16.jpg)
i. SAEC Organization and Committees BOD Genotyping Core [Expression Analysis, Inc] Scientific/Clinical SMC SAE [s] Bioinformatics Core [Columbia University] CEO Legal/IP PR/ Comm. Data Anal. Research Collaborators [Academic & Commercial] Outsourced Service Suppliers [Legal & PR] 16

i. SAEC Web Site http: //www. saeconsortium. org 17

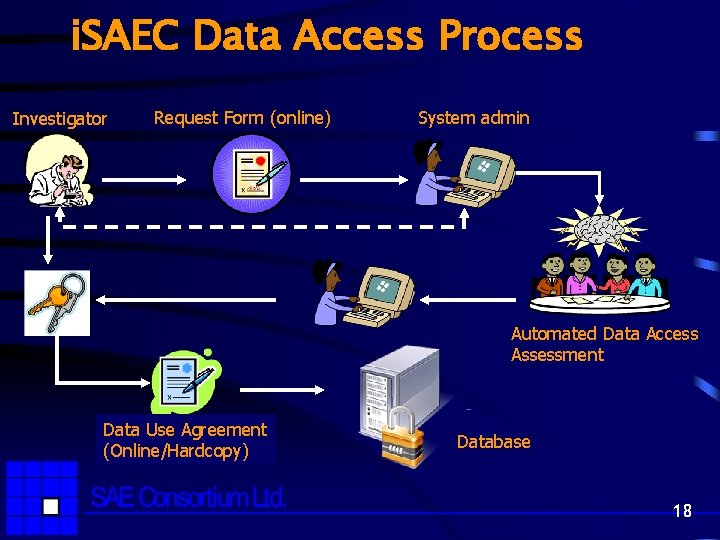
i. SAEC Data Access Process Investigator Request Form (online) System admin Automated Data Access Assessment Data Use Agreement (Online/Hardcopy) Database 18

Lessons Learned

10 Lessons Learned (Success Factors) 1. A clear unifying objective is a must. 2. Ensure the effort is both “pre-competitive” and “pro-competitive. ” 3. Have a quality “draft” operating concept/plan before recruiting members [“one shot rule”]. 4. Establish a uniform membership requirements & development strategy with potential members. 5. Establish dedicated management early in the effort /don’t reinvent the wheel. 20

10 Success Factors cont. 6. Organize well defined committees with high quality, dedicated leaders 7. Outsource…outsource… outsource to the best external advisors/ investigators via “performance based” contracts 8. Generate results that exceed expectations 9. Make it fun and say “thank you” in meaningful ways, and…. 10. Know when to fold … begin with an end in mind! 21

Learning #1 Have a Clear, Quality Objective which Unifies the Membership […Full Genome Representative SNP Map… Safety related markers in the public domain] • Needed Science! • Easily Understood • Measurable • Focused vs General • All essential for member recruitment 22

Learning #2 The focus should be “pre-competitive” and “pro-competitive. ” • Pre-Competitive (moving target) • Pro-Competitive (legal antitrust perspective) • “Public Good” orientation (Tax/Charity status) • “Neutral data access policy” “public” access to data 23
![Learning 3 Develop a highquality plan before recruiting membership Well organized formation phase Learning #3 Develop a high-quality plan [before recruiting membership] • Well organized “formation phase”](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-24.jpg)
Learning #3 Develop a high-quality plan [before recruiting membership] • Well organized “formation phase” with potential members • Scientific Plan [including necessary feasibility pilots] • Informatics Plan • Legal/IP Policies • Operating Plan/Performance Metrics/Contingencies • Budget [Proactive Risk Factor Mgnt] 24

Learning #4 Establish a Clear Membership Requirement & Development Strategy with potential members • High Priority a Must! • Fixed Dues Structure (Annual Sensitivity [$1 M down to <$. 4 M/year) • Do not vary membership tiers for industrial members • Non industrial members >> same $ terms for voting rights • Member Diversification has pros & cons 25

Learning #5 Establish dedicated management ASAP • Formation phase leadership • Members are investing in both the leader/project • Defining the Organization & Key Processes • Sourcing strategies • Pre-formation negotiations • “Closing” Membership • Communication is Key • Everything takes more Effort than Planned 26

Learning #6 Organize clear committees with high quality dedicated leaders • SMC, Legal/IP, & Subcommittees key • Functional/Scientific Leadership is vital • Time Commitment is Greater than Expected • Senior Management Support of Manager Commitment 27

Learning #7 Outsource…outsource… outsource • Increases expertise & skills • Leverage existing infrastructure/investments/skill • Performance based contracts no grants! • Reduces project risk • Increases management flexibility • Careful assessment of the risks of doing the Consortium if Outsourcing is not an Option

Learning #8 Generate results that exceed expectations! • Confirms membership investment creates additional consortia opportunities • “Consortia fatigue” is real major cause is poorly managed/under performing consortia • Winning together is fun … 29

Learning #9 Say “thank you” in meaningful ways and make the consortium a fun experience • Member help is essential • “Recognition starvation” is an epidemic • Everybody likes “special events” … but costeffectiveness is vital in today’s environment 30

Learning #10 Don’t try to make a consortium into something it shouldn't be … know when to fold • Finite objectives within a fixed time frame attractive to members • Consortia are strategic initiatives vs on-going concerns • New Consortia require different leadership • Effective BOD with the appropriate experience & seniority—Key!

Final Caveats • Wellcome Trust involvement and funding important validation to pharmcos • Involve stakeholder governmental bodies (TSC— NIH, i. SAEC—FDA, EMEA, PMDA), without slowing the effort. • Limits to company leverage in these “leaner times” … funding a quality consortium is very hard these days (more freeloaders) • Vital that we learn from best efforts take these into new, important consortia efforts to improve pharma productivity 32
![Acknowledgments SMC n Ann Daly DILIGEN Newcastle n Mariam Molokhia EUDRAGENE London Acknowledgments SMC n Ann Daly & DILIGEN [Newcastle] n Mariam Molokhia & EUDRAGENE [London]](https://slidetodoc.com/presentation_image/3460a9da3b5aac70b6f7ffea1d0b48fa/image-33.jpg)
Acknowledgments SMC n Ann Daly & DILIGEN [Newcastle] n Mariam Molokhia & EUDRAGENE [London] n Matt Nelson [GSK] n Sally John [Pfizer] n Yufeng Shen [Columbia] n Itsik Pe’er [Columbia] n Aris Floratos [Columbia] n Mark Daly [Harvard/Broad] n David Goldstein [Duke] n Eric Lai [ex-GSK] n Donald Halbert [Abbott] n Joe Walker [D-S] n Nadine Cohen, Quingqin Serena Li, & Adrian Thomas [J&J] n Joanne Meyer & Steve Lewitzky [Novartis] n Klaus Lindpaintner & Karen Wilcock [Roche] n Steven Kovacs [Sanofi-Aventis] n Leonardo Sahelijo [Takeda] n Ted Burczynski & Maha Karnoub [Wyeth] n Robert O'Neill & Steve Wilson [FDA] n Andrea Califano [Columbia] n Allen Roses [Duke] n John Senior [FDA] 33 Members n Brian Spear [Abbott] n Rick Scheyer [Daiichi Sankyo] n Lon Cardon [GSK] n Nadine Cohen [J&J] n Joanne Meyer [Novartis] n Aidan Power [Pfizer] n Klaus Lindpaintner [Roche] n Robert Dix [Sanofi-Aventis] n Leonardo Sahelijo [Takeda] n Michael Burczynski [Wyeth] n Janet Woodcock [FDA] n Sha. Avhree Buckman [FDA] n Michael Dunn [Wellcome Trust]

Appendix Slides

Industrial Consortia Some Operational Tenants • Unifying objective industry and public good [501 c 3] • Focused projects, with clear objectives & strong operational management • Strong “quality” and “time to result” orientation • Clear and uniform “membership requirements” • Extensive leverage of members’ skills via well organized sub-committees • Strive to collaborate with the best quality external advisors & investigators on an international basis • Public release of data after appropriate IP management actions to ensure “openness” 35