Review of Analytical Methods Part 2 Electrochemistry Roger

























- Slides: 40

Review of Analytical Methods Part 2: Electrochemistry Roger L. Bertholf, Ph. D. Associate Professor of Pathology Chief of Clinical Chemistry & Toxicology University of Florida Health Science Center/Jacksonville

Analytical methods used in clinical chemistry • • Spectrophotometry Electrochemistry Immunochemistry Other – Osmometry – Chromatography – Electrophoresis

Electrochemistry • Electrochemistry applies to the movement of electrons from one compound to another – The donor of electrons is oxidized – The recipient of electrons is reduced • The direction of flow of electrons from one compound to another is determined by the electrochemical potential

Electrochemical potential • Factors that affect electrochemical potential: – Distance/shielding from nucleus – Filled/partially filled orbitals

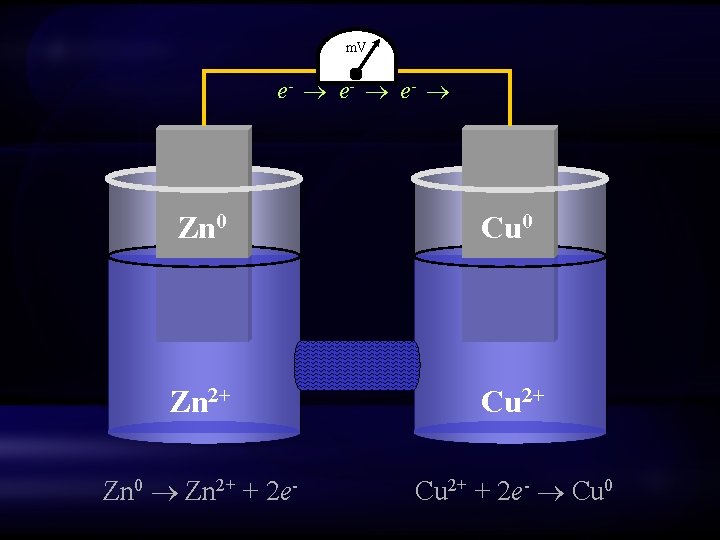
Relative potential Zn e- Cu • Copper is more electronegative than Zinc • When the two metals are connected electrically, current (electrons) will flow spontaneously from Zinc to Copper – Zinc is oxidized; Copper is reduced – Zinc is the anode; Copper is the cathode

m. V e- e- Zn 0 Cu 0 Zn 2+ Cu 2+ Zn 0 Zn 2+ + 2 e- Cu 2+ + 2 e- Cu 0

The Nernst Equation Where E = Potential at temperature T E 0 = Standard electrode potential (25ºC, 1. 0 M) R = Ideal gas constant F = Faraday’s constant n = number of electrons transferred

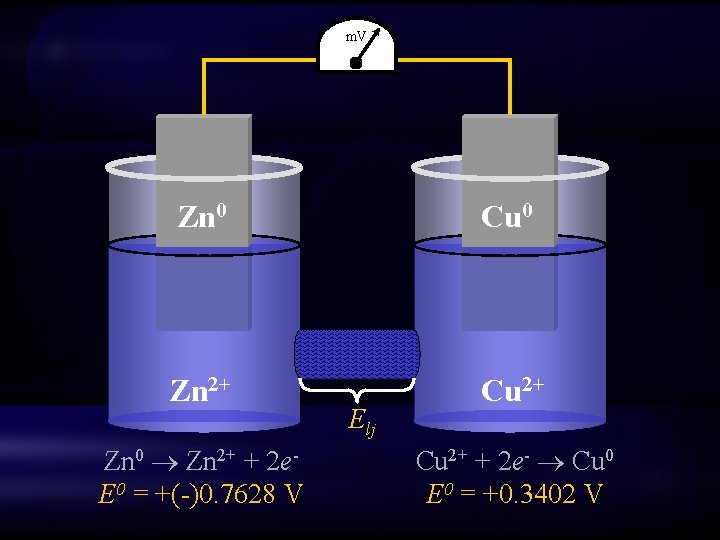
m. V Zn 0 Cu 0 Zn 2+ Cu 2+ Zn 0 Zn 2+ + 2 e. E 0 = +(-)0. 7628 V Elj Cu 2+ + 2 e- Cu 0 E 0 = +0. 3402 V

Electromotive force Ecell = Ecathode + Elj - Eanode Ecell = ECu(II), Cu + Elj – EZn(II), Zn Ecell = (+)0. 340 + Elj – (-)0. 763 Ecell = (+)1. 103 + Elj G = -n. FEcell

Would the reaction occur in the opposite direction? Ecell = Ecathode + Elj - Eanode Ecell = EZn(II) Zn + Elj – ECu(II) Cu Ecell = (-)0. 763 + Elj – (+)0. 340 Ecell = (-)1. 103 + Elj

How do we determine standard electrode potentials? • Absolute potential cannot be measured— only the relative potential can be measured • Standard electrode potentials are measured relative to a Reference Electrode • A Reference Electrode should be. . . – Easy to manufacture – Stable

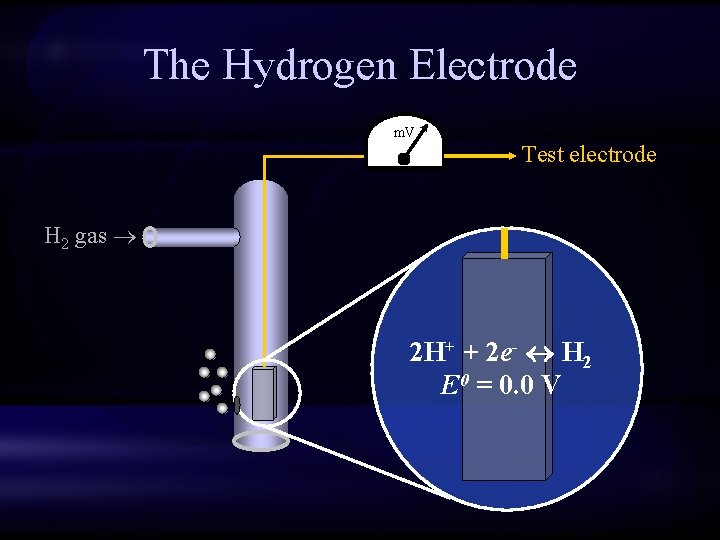
The Hydrogen Electrode m. V Test electrode H 2 gas 2 H+ + 2 e- H 2 E 0 = 0. 0 V

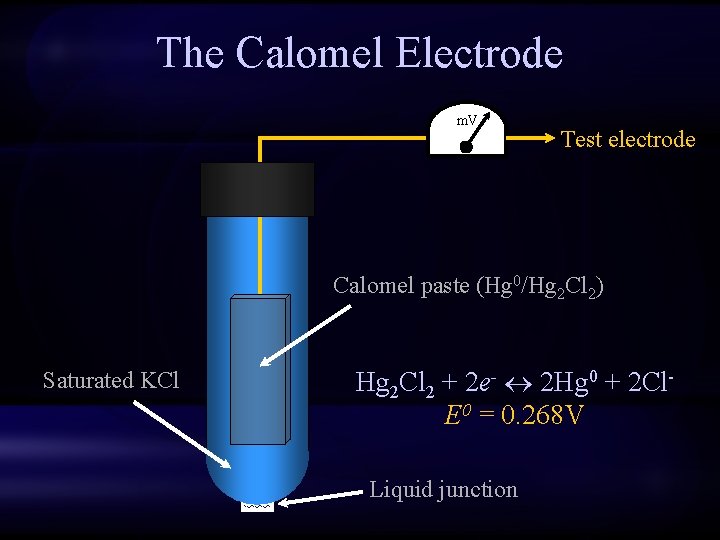
The Calomel Electrode m. V Test electrode Calomel paste (Hg 0/Hg 2 Cl 2) Saturated KCl Hg 2 Cl 2 + 2 e- 2 Hg 0 + 2 Cl. E 0 = 0. 268 V Liquid junction

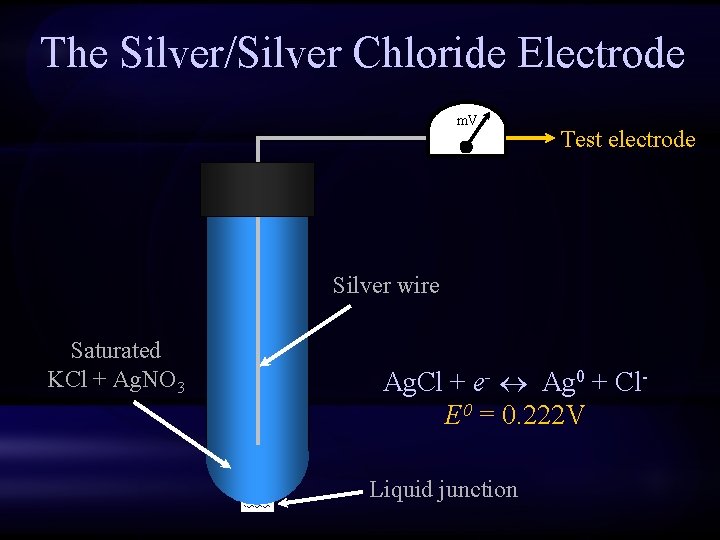
The Silver/Silver Chloride Electrode m. V Test electrode Silver wire Saturated KCl + Ag. NO 3 Ag. Cl + e- Ag 0 + Cl. E 0 = 0. 222 V Liquid junction

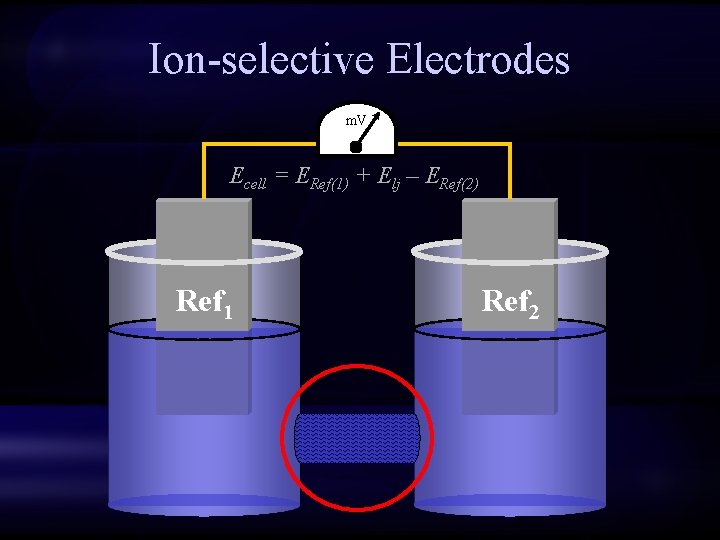
Ion-selective Electrodes m. V Ecell = ERef(1) + Elj – ERef(2) Ref 1 Ref 2

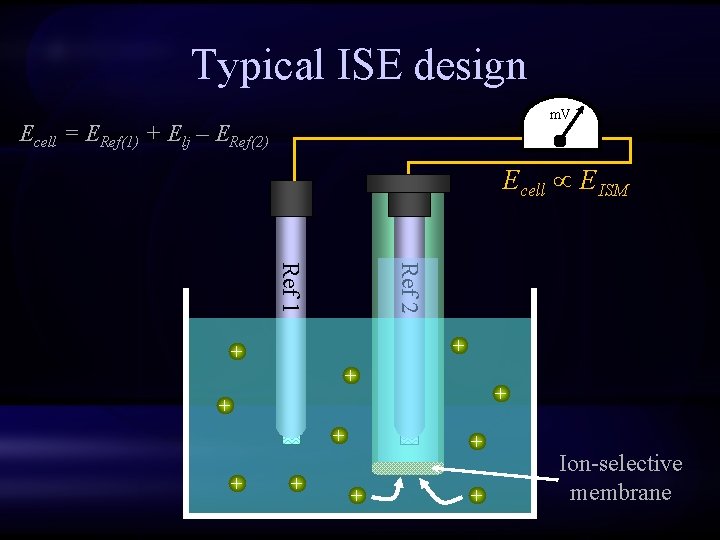
Typical ISE design m. V Ecell = ERef(1) + Elj – ERef(2) Ecell EISM Ref 2 Ref 1 + + + Ion-selective membrane

Activity and concentration • ISEs do not measure the concentration of an analyte, they measure its activity. – Ionic activity has a specific thermodynamic definition, but for most purposes, it can be regarded as the concentration of free ion in solution. – The activity of an ion is the concentration times the activity coefficient, usually designated by :

The activity coefficient • Solutions (and gases) in which none of the components interact are called ideal, and have specific, predictable properties • Deviations from ideal behavior account for the difference between concentration and activity • Dilute solutions exhibit nearly ideal behavior ( 1)

Types of ISE • Glass – Various combinations of Si. O 2 with metal oxides • Solid-state – Involve ionic reaction with a crystalline (or crystal doped) membrane (example: Cl-/Ag. Cl) • Liquid ion-exchange – A carrier compound is dissolved in an inert matrix • Gas sensors – Usually a combination of ISE and gas-permeable membrane

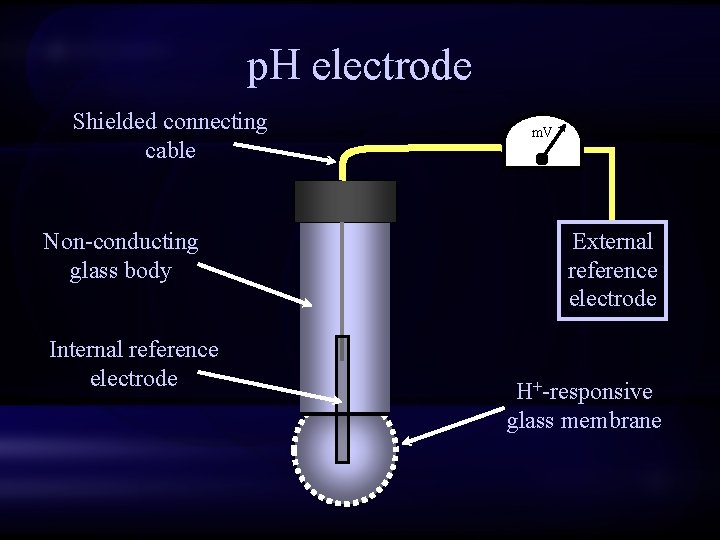
p. H electrode Shielded connecting cable Non-conducting glass body Internal reference electrode m. V External reference electrode H+-responsive glass membrane

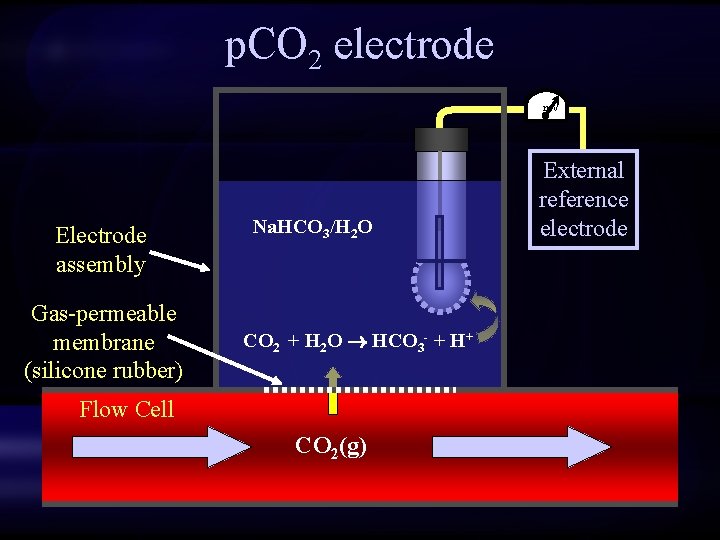
p. CO 2 electrode m. V Electrode assembly Gas-permeable membrane (silicone rubber) Na. HCO 3/H 2 O CO 2 + H 2 O HCO 3 - + H+ Flow Cell CO 2(g) External reference electrode

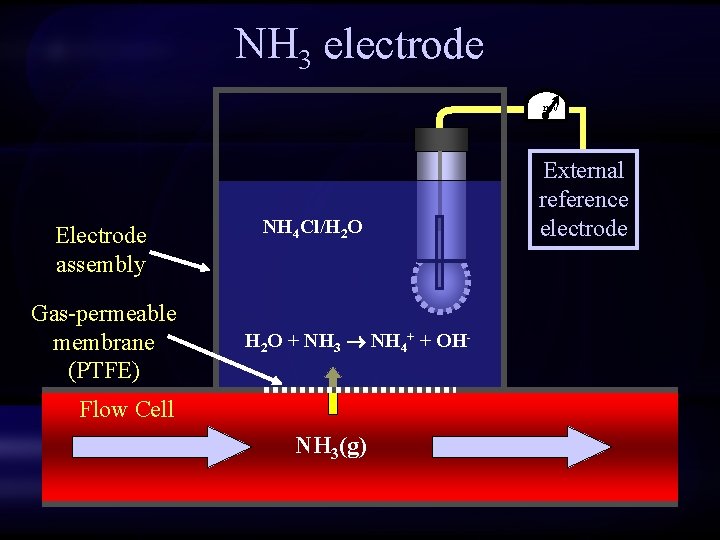
NH 3 electrode m. V Electrode assembly Gas-permeable membrane (PTFE) NH 4 Cl/H 2 O + NH 3 NH 4+ + OH- Flow Cell NH 3(g) External reference electrode

Other glass electrodes • Glass electrodes are used to measure Na+ – There is some degree of cross-reactivity between H+ and Na+ • There are glass electrodes for K+ and NH 4+, but these are less useful than other electrode types

The Sodium Error (or, direct vs. indirect potentiometry) m. V Lipids, proteins Na+ Aqueous phase Plasma Cells (45%) Whole blood Since potentiometry measures the activity of the ion at the electrode surface, the measurement is independent of the volume of sample.

The Sodium Error (or, direct vs. indirect potentiometry) m. V Na+ In indirect potentiometry, the concentration of ion is diluted to an activity near unity. Since the concentration will take into account the original volume and dilution factor, any excluded volume (lipids, proteins) introduces an error, which usually is insignificant.

So which is better? • Direct potentiometry gives the true, physiologically active sodium concentration. • However, the reference method for sodium is atomic emission, which measures the total concentration, not the activity, and indirect potentiometry methods are calibrated to agree with AE. • So, to avoid confusion, direct potentiometric methods ordinarily adjust the result to agree with indirect potentiometric (or AE) methods.

Then what’s the “sodium error” all about? • When a specimen contains very large amounts of lipid or protein, the dilutional error in indirect potentiometric methods can become significant. • Hyperlipidemia and hyperproteinemia can result in a pseudo-hyponatremia by indirect potentiometry. • Direct potentiometry will reveal the true sodium concentration (activity).

Sodium error Na+ 138 m. M 130 m. M Na+ 140 m. M

But. . . why does it only affect sodium? • It doesn’t only affect sodium. It effects any exclusively aqueous component of blood. • The error is more apparent for sodium because the physiological range is so narrow.

Solid state chloride electrode • Ag. Cl and Ag 2 S are pressed into a pellet that forms the liquid junction (ISE membrane) • Cl- ions diffuse into vacancies in the crystal lattice, and change the membrane conductivity

Liquid/polymer membrane electrodes • Typically involves an ionophore dissolved in a water-insoluble, viscous solvent • Sometimes called ion-exchange membrane electrodes • The ionophore determines the specificity of the electrode

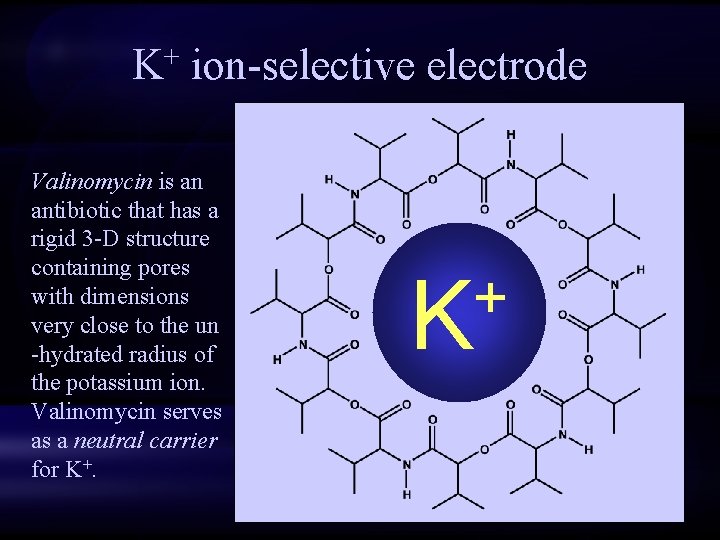
K+ ion-selective electrode Valinomycin is an antibiotic that has a rigid 3 -D structure containing pores with dimensions very close to the un -hydrated radius of the potassium ion. Valinomycin serves as a neutral carrier for K+. + K

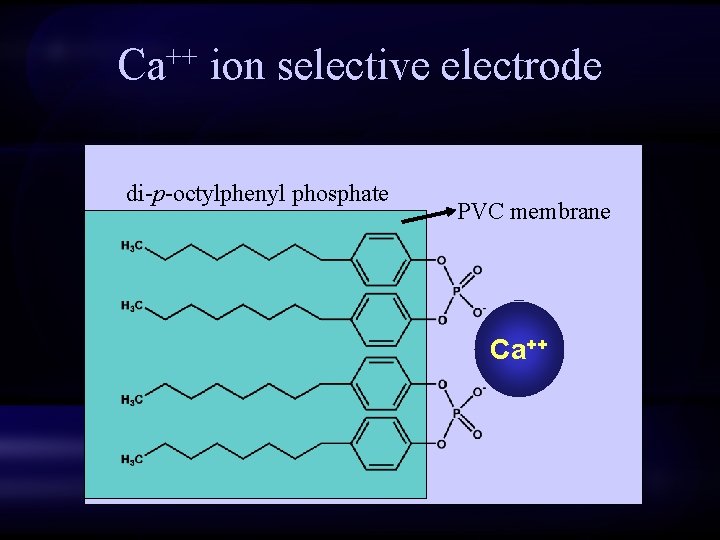
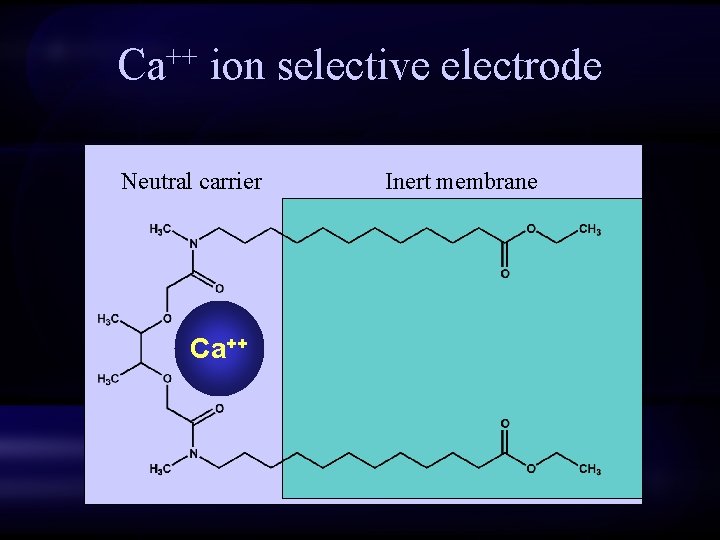
Ca++ ion selective electrode di-p-octylphenyl phosphate PVC membrane Ca++

Ca++ ion selective electrode Neutral carrier Ca++ Inert membrane

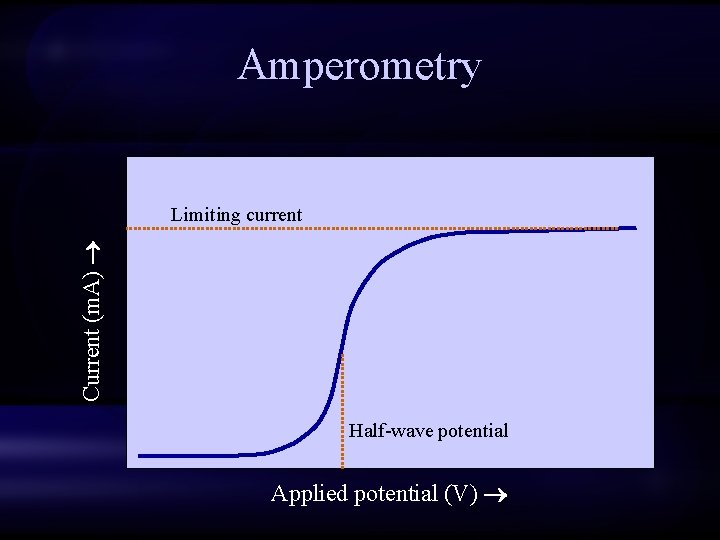
Amperometry • Whereas potentiometric methods measure electrochemical potential, amperometric methods measure the flow of electrical current • Potential (or voltage) is the driving force behind current flow • Current is the amount of electrical flow (electrons) produced in response to an electrical potential

Amperometry Current (m. A) Limiting current Half-wave potential Applied potential (V)

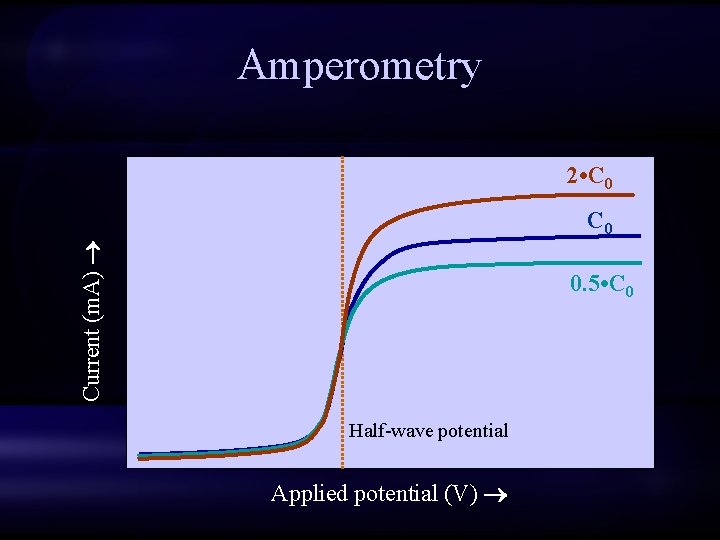
Amperometry 2 • C 0 Current (m. A) C 0 0. 5 • C 0 Half-wave potential Applied potential (V)

Oxygen (p. O 2) electrode -0. 65 V Reference electrode (anode) Platinum wire (cathode) Gas-permeable membrane Flow cell O 2

Reaction at the platinum electrode • The amount of current (e-) is proportional to the concentration of O 2

The glucose electrode Glucose + O 2 Glucose oxidase H 2 O 2 + Gluconic acid 2 e- O 2 + 2 H- O 2 electrode (+0. 6 V) • Note that the platinum electrode now carries a positive potential