ELECTROCHEMISTRY Lithiumion battery 1 Electrochemistry the area of

ELECTROCHEMISTRY Lithium-ion battery. 1

Electrochemistry = the area of chemistry that deals with the interconversion of electrical energy and chemical energy. l Electrochemical processes are redox reactions in which the energy released by a spontaneous reaction is converted to electricity or in which electricity is used to drive a non-spontaneous chemical reaction. 2

Galvanic or Voltaic cell = an electrochemical cell that produces electricity as a result of spontaneous chemical change 3
Why Does a Voltaic Cell Work? The spontaneous reaction occurs as a result of the different abilities of materials (such as metals) to give up their electrons and the ability of the electrons to flow through the circuit. Ecell > 0 for a spontaneous reaction 1 Volt (V) = 1 Joule (J)/ Coulomb (C) 4

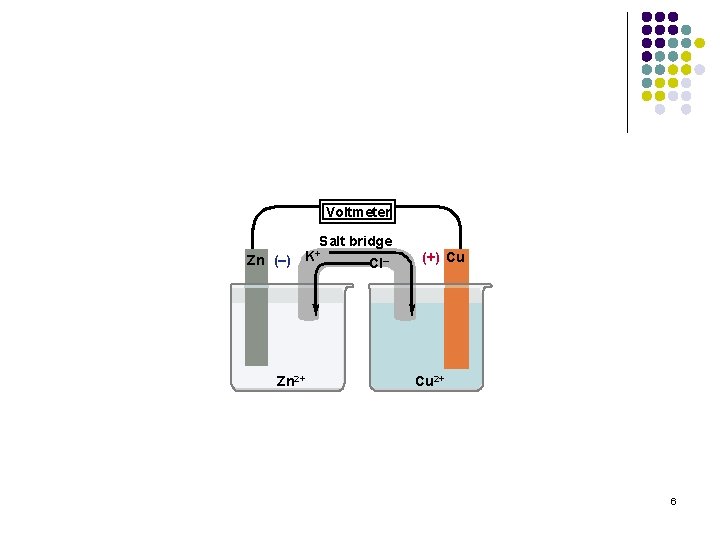
Example of Voltaic Cell Voltmeter Salt bridge Zn (–) Zn 2+ K+ Cl– (+) Cu Cu 2+ Zn and Cu cell 5
Voltmeter Salt bridge Zn (–) Zn 2+ K+ Cl– (+) Cu Cu 2+ 6
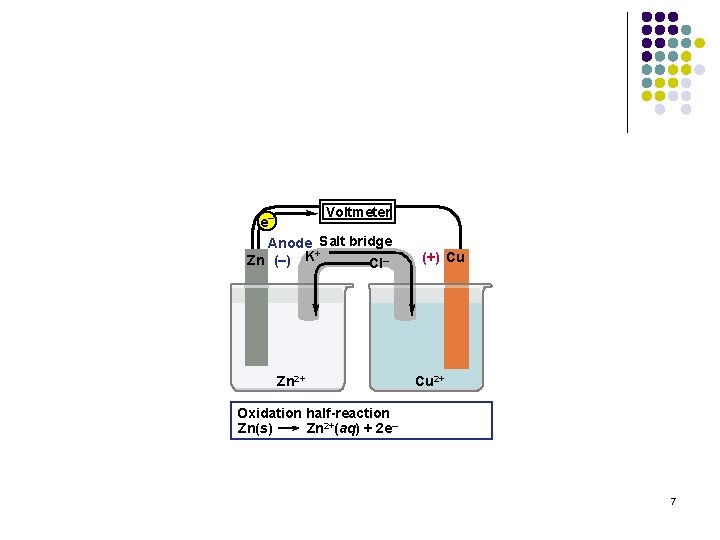
Voltmeter e– Anode Salt bridge + Zn (–) K Cl– Zn 2+ (+) Cu Cu 2+ Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e– 7
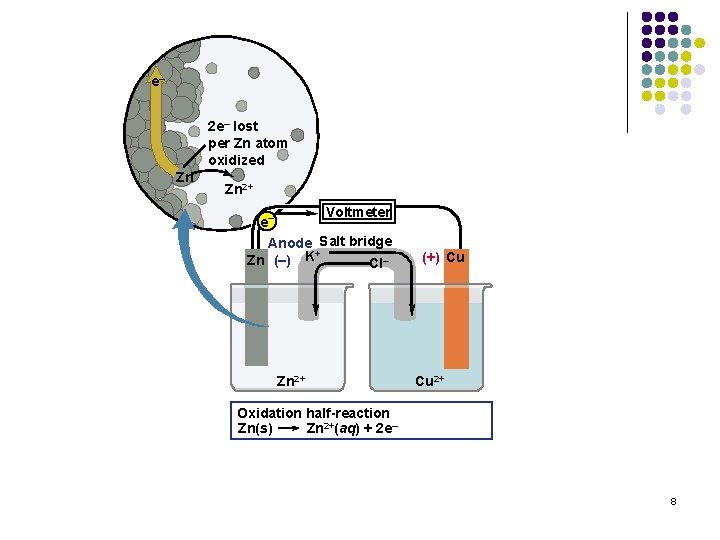
e– 2 e– lost per Zn atom oxidized Zn Zn 2+ Voltmeter e– Anode Salt bridge + Zn (–) K Cl– Zn 2+ (+) Cu Cu 2+ Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e– 8
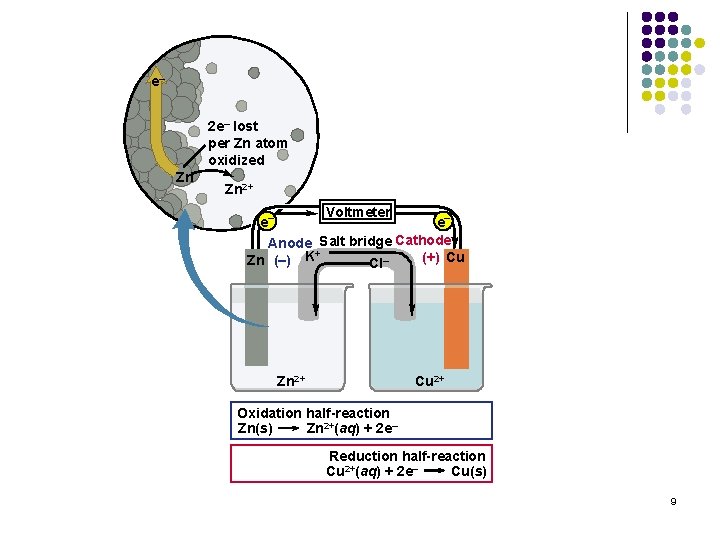
e– 2 e– lost per Zn atom oxidized Zn Zn 2+ Voltmeter e– e– Anode Salt bridge Cathode + (+) Cu Zn (–) K Cl– Zn 2+ Cu 2+ Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e– Reduction half-reaction Cu 2+(aq) + 2 e– Cu(s) 9
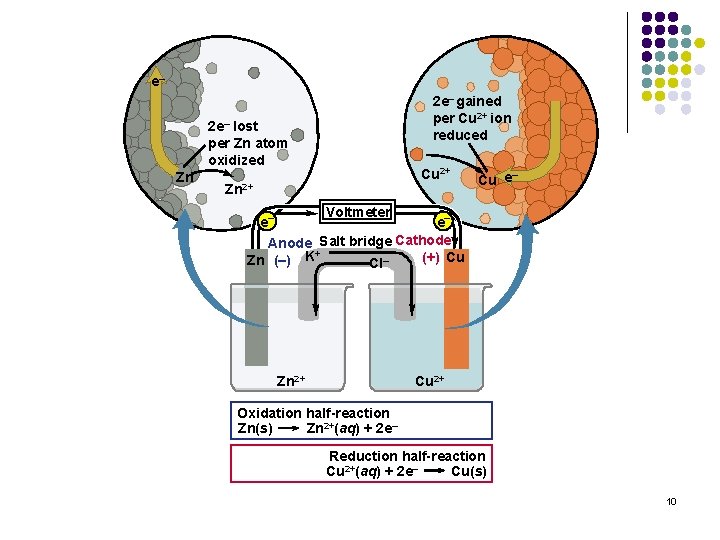
e– 2 e– gained per Cu 2+ ion reduced 2 e– lost per Zn atom oxidized Zn Cu 2+ Zn 2+ – Cu e Voltmeter e– e– Anode Salt bridge Cathode + (+) Cu Zn (–) K Cl– Zn 2+ Cu 2+ Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e– Reduction half-reaction Cu 2+(aq) + 2 e– Cu(s) 10
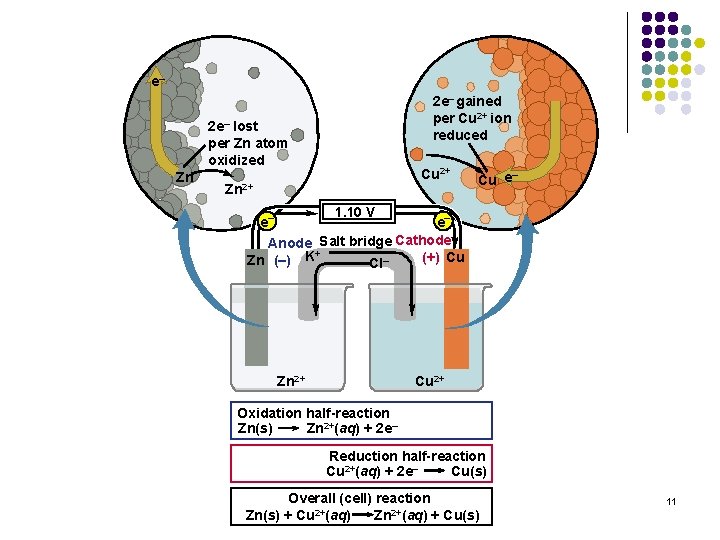
e– 2 e– gained per Cu 2+ ion reduced 2 e– lost per Zn atom oxidized Zn Cu 2+ Zn 2+ – Cu e 1. 10 V e– e– Anode Salt bridge Cathode + (+) Cu Zn (–) K Cl– Zn 2+ Cu 2+ Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e– Reduction half-reaction Cu 2+(aq) + 2 e– Cu(s) Overall (cell) reaction Zn 2+(aq) + Cu(s) Zn(s) + Cu 2+(aq) 11
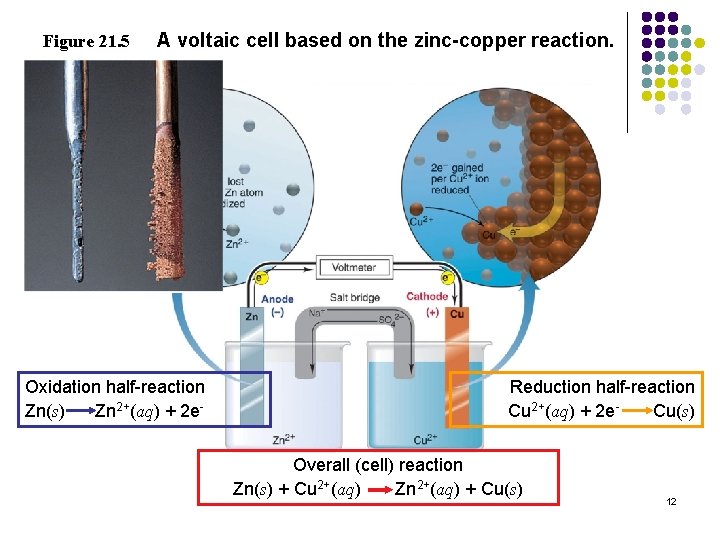
Figure 21. 5 A voltaic cell based on the zinc-copper reaction. Oxidation half-reaction Zn(s) Zn 2+(aq) + 2 e- Reduction half-reaction Cu 2+(aq) + 2 e. Cu(s) Overall (cell) reaction Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s) 12

13

Symbolic representation of cell diagram 14

Notation for a Voltaic Cell components of anode compartment components of cathode compartment (oxidation half-cell) (reduction half-cell) phase of lower phase of higher oxidation state phase of lower oxidation state phase boundary between half-cells Examples: Zn(s) | Zn 2+(aq) || Cu 2+(aq) | Cu (s) Zn 2+(aq) + 2 e- Cu(s) | = represents boundary between an electrode and another phase (solution or gas) || = signifiess that solutions are joined by a salt bridge 15

* Another way of representing the cell diagram: Zn(s) | Zn 2+(aq) | KCl | Cu 2+(aq) | Cu (s) 16
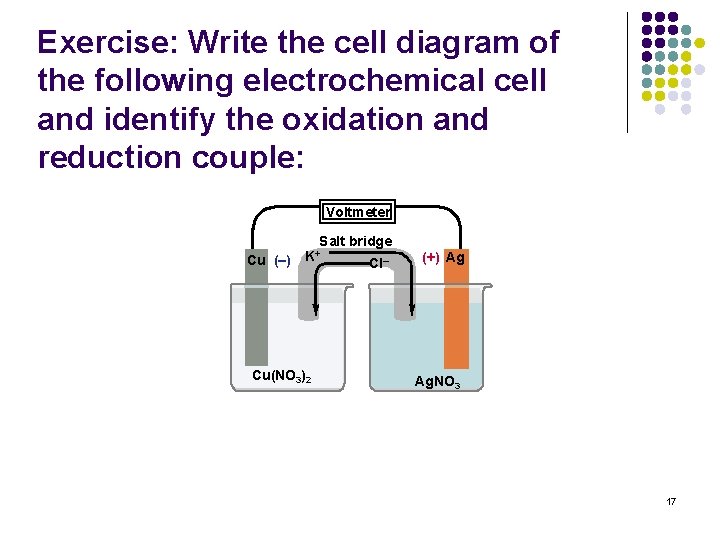
Exercise: Write the cell diagram of the following electrochemical cell and identify the oxidation and reduction couple: Voltmeter Salt bridge Cu (–) K+ Cu(NO 3)2 Cl– (+) Ag Ag. NO 3 17
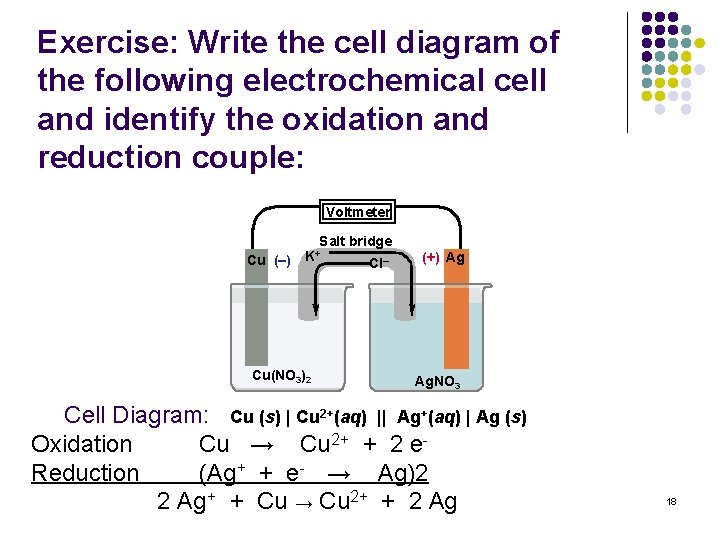
Exercise: Write the cell diagram of the following electrochemical cell and identify the oxidation and reduction couple: Voltmeter Salt bridge Cu (–) K+ Cu(NO 3)2 Cl– (+) Ag Ag. NO 3 Cell Diagram: Cu (s) | Cu 2+(aq) || Ag+(aq) | Ag (s) Oxidation Cu → Cu 2+ + 2 e. Reduction (Ag+ + e- → Ag)2 2 Ag+ + Cu → Cu 2+ + 2 Ag 18

Eo = Standard Electrode Potential = is based on the tendency for a reduction process to occur at the electrode 19

Rules: • By convention, electrode potentials are written as reductions. The Eo values apply to the half-cell reactions as read in the forward direction (left to right). • The more positive Eo, the greater the tendency to be reduced. • The half-cell reactions are reversible, (sign changes). The reduction halfcell potential and the oxidation half-cell potential are added to obtain the E 0 cell. • Eo values are unaffected by multiplying half-equations by constant coefficients. 20

Table 21. 2 Selected Standard Electrode Potentials (298 K) F 2(g) + 2 e 2 F-(aq) Cl 2(g) + 2 e 2 Cl-(aq) Mn. O 2(g) + 4 H+(aq) + 2 e. Mn 2+(aq) + 2 H 2 O(l) NO 3 -(aq) + 4 H+(aq) + 3 e. NO(g) + 2 H 2 O(l) Ag+(aq) + e. Ag(s) Fe 3+(g) + e. Fe 2+(aq) O 2(g) + 2 H 2 O(l) + 4 e 4 OH-(aq) Cu 2+(aq) + 2 e. Cu(s) 2 H+(aq) + 2 e. H 2(g) N 2(g) + 5 H+(aq) + 4 e. N 2 H 5+(aq) Fe 2+(aq) + 2 e. Fe(s) 2 H 2 O(l) + 2 e. H 2(g) + 2 OH-(aq) Na+(aq) + e. Na(s) Li+(aq) + e. Li(s) E 0(V) +2. 87 +1. 36 +1. 23 +0. 96 +0. 80 +0. 77 +0. 40 +0. 34 0. 00 -0. 23 -0. 44 -0. 83 -2. 71 -3. 05 strength of reducing agent strength of oxidizing agent Half-Reaction 21

Example: Cu (s) | Cu 2+(aq) || Ag+(aq) | Ag (s) Oxidation Reduction Cu → Cu 2+ + 2 e(Ag+ + e- → Ag)2 2 Ag+ + Cu → Cu 2+ + 2 Ag - 0. 34 0. 80 0. 46 V *reaction is spontaneous 22

Problem: 1) Write the cell reactions for the electrochemical cells diagrammed below and calculate Eo cell for each reaction. Will these reactions occur spontaneously or non-spontaneously? a) Zn(s) | Zn 2+(aq) | Na 2 SO 4 | Sn 2+(aq) | Sn (s) b) Pt(s) | Fe 2+(aq), Fe 3+(aq) | | Sn+4(aq), Sn 2+(aq) | Pt(s) c) Al(s) | Al 3+(aq) | NH 4 NO 3 | Cu 2+(aq) | Cu (s) 23

Answers: a) Ox: Zn → Zn 2+ + 2 e. Red: Sn 2+ + 2 e- → Sn Zn + Sn 2+ → Zn 2+ + Sn 0. 76 V -0. 14 V Eo = 0. 62 V, spontaneous b) Ox: 2 (Fe 2+ → Fe 3+ + 1 e-) -0. 77 V Red: Sn 4+ + 2 e- → Sn 2+ 0. 15 V 2 Fe 2+ + Sn 4+ → 2 Fe 3+ + Sn 2+ Eo = -0. 62 V, non-spo c) Ox: 2 (Al → Al 3+ + 3 e-) Red: 3 (Cu 2+ + 2 e- → Cu) 2 Al + 3 Cu 2+ → 3 Cu + 2 Al 3+ 1. 66 V 0. 34 V Eo = 2. 00 V, spontaneous 24

Problem: 2) Give the cell diagram and determine the Eo cell for the reaction: Zn(s) + Cl 2(g) Zn. Cl 2(aq). Answers: Cell diagram: Zn(s) | Zn 2+(aq) | | Cl 2(g), Cl-(aq) | Pt (s) Ox: Zn → Zn 2+ + 2 e. Red: Cl 2 + 2 e- → 2 Cl. Zn + Cl 2 → Zn 2+ + 2 Cl- 0. 76 V 1. 36 V 2. 12 V = Eo 25

Free Energy = ▲G ▲G = ▲H - T▲S ▲G = 0 , the system is at equilibrium ▲G < 0 , spontaneous reaction ▲G > 0 , non-spontaneous reaction 26

Free Energy = ▲G ▲G = ▲H - T▲S ▲G = ( - ), always a spontaneous process in ELECTROCHEMISTRY ▲G = Wmax (max. amt. of work that can be done) 27

in a voltaic cell, chemical energy is converted into electrical energy Voltages of Some Voltaic or Galvanic Cells Voltaic Cell Voltage (V) Common alkaline battery 1. 5 Lead-acid car battery (6 cells = 12 V) 2. 0 Calculator battery 1. 3 Electric eel 0. 15 28

Electrical energy = volts X coulombs = Joules The total charge is determined by the n of electrons that pass through the circuit. By definition: total charge = n F (1 F = 96, 500 C/mol) since 1 J = 1 C X 1 V We can also express the units of Faraday as 1 F = 96, 500 J/V. mol 29

The total work done is the product of 3 terms: 1. 2. 3. The emf (voltage) of the cell The n of e-s transferred between the electrodes The electric charge per mole of e-s. Welec = (n) (F) (Eocell) 30

Welec = (n) (F) (Eocell) Since for a spontaneous reaction ▲G < 0 ▲G = - (n) (F) (Eocell) 31

Problem: Determine ▲G for the reaction: Alo(s) | Al 3+(aq) | | Cu 2+(aq) | Cuo (s) oxi: Al → Al 3+ + 3 e. X 2 red: Cu 2+ + 2 e- → Cu X 3 2 Al + 3 Cu 2+ → 2 Al 3++ 3 Cu 1. 66 V 0. 34 V 2. 00 V = Eo ▲G = - (n F Eocell) = - ( 6 mol e- X 96500 C/mol e- X 2. 00 V ) = - 1, 158, 000 J or = - 1, 158 k. J 32

Problem: 2) Give the cell diagram and determine ▲G for the reaction: Zn(s) + Cl 2(g) Zn. Cl 2(aq). Answers: Cell diagram: Zn(s) | Zn 2+(aq) | | Cl 2(g) , Cl-(aq) | Pt (s) Ox: Zn → Zn 2+ + 2 e. Red: Cl 2 + 2 e- → 2 Cl. Zn + Cl 2 → Zn 2+ + 2 Cl- 0. 76 V 1. 36 V 2. 12 V = Eo ▲G = - (n F Eocell) = - ( 2 mol e- X 96500 C/mol e- X 2. 12 V ) = - 409, 160 J or - 409. 2 k. J 33

EXERCISES: 34

I. Give the following: a) cell diagram, b) anode and cathode reactions, 1. 2. 3. 4. 5. 6. c) Eo for the net reaction and d) ▲G for the following reactions Ag+(aq) + Mgo(s) → Mg 2+(aq) + Ago(s) I 2(s) + Cl-(aq) → Cl 2(g) + I-(aq) Br 2 o(aq) + Fe 2+(aq) → Fe. Br 3(aq) Pb 2+(aq) is displaced from solution by Al(s) Mg. Br 2(aq) is produced from Mg(s) and Br 2(l) Cl 2(g) is reduced to Cl-(aq) and Fe(s) is oxidized to Fe 2+(aq) 35

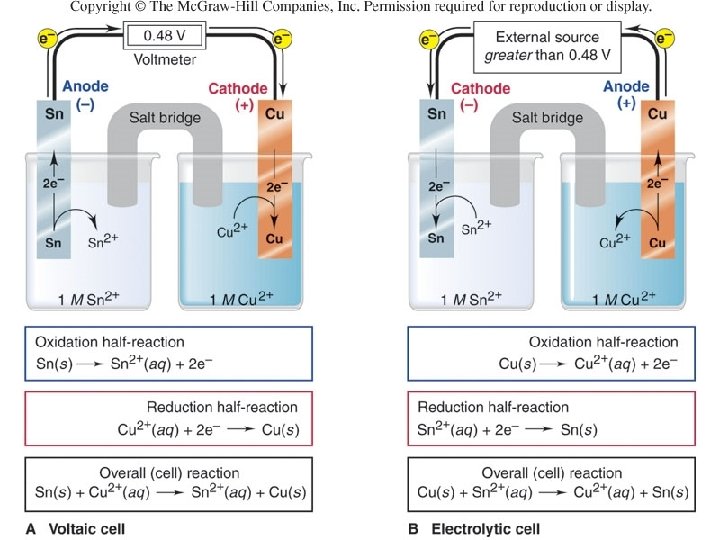
Sketch or draw and label the voltaic cell from the spontaneous reaction of Cu 2+ and Sn+2 solutions. Indicate the following: the a) anode, b) cathode, c) oxidation reaction, d) reduction reaction, e) final equation, and f) direction of electron flow. 36

Arrange the following metals in the order of decreasing oxidizing strength: Ni, Au, Ag, Al, and Cr. 37

38
CORROSION OF IRON 39

CORROSION OF IRON l No indication of corrosion l l l Basic or alkaline solutions (Na. OH, KOH, Mg(OH)2) With indication of corrosion l l Neutral solutions (including water, Na 2 SO 4, K 3 PO 4) Acidic solutions (HCl, H 2 SO 4, NH 4 Cl) 40

Table 21. 2 Selected Standard Electrode Potentials (298 K) F 2(g) + 2 e 2 F-(aq) Cl 2(g) + 2 e 2 Cl-(aq) Mn. O 2(g) + 4 H+(aq) + 2 e. Mn 2+(aq) + 2 H 2 O(l) NO 3 -(aq) + 4 H+(aq) + 3 e. NO(g) + 2 H 2 O(l) Ag+(aq) + e. Ag(s) Fe 3+(g) + e. Fe 2+(aq) O 2(g) + 2 H 2 O(l) + 4 e 4 OH-(aq) Cu 2+(aq) + 2 e. Cu(s) 2 H+(aq) + 2 e. H 2(g) N 2(g) + 5 H+(aq) + 4 e. N 2 H 5+(aq) Fe 2+(aq) + 2 e. Fe(s) Cr 3+(aq) + 3 e. Cr(s) Zn 2+(aq) + 2 e. Zn(s) Mg 2+(aq) + 2 e. Mg(s) E 0(V) +2. 87 +1. 36 +1. 23 +0. 96 +0. 80 +0. 77 +0. 40 +0. 34 0. 00 -0. 23 -0. 44 -0. 76 -2. 37 strength of reducing agent strength of oxidizing agent Half-Reaction 41
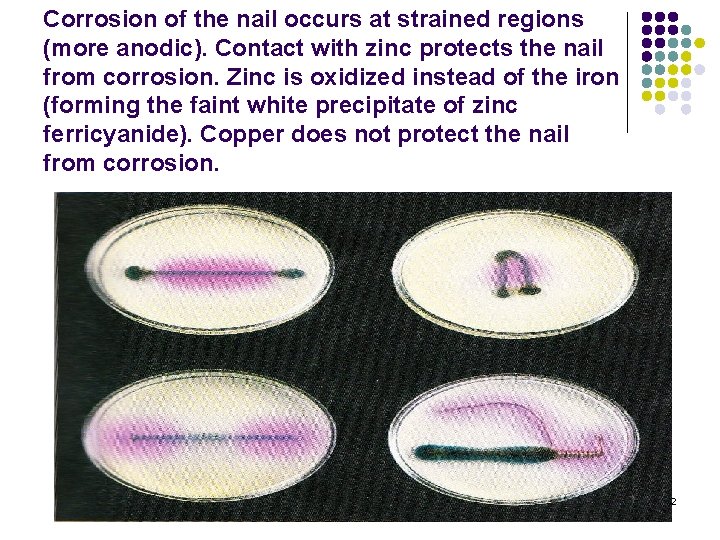
Corrosion of the nail occurs at strained regions (more anodic). Contact with zinc protects the nail from corrosion. Zinc is oxidized instead of the iron (forming the faint white precipitate of zinc ferricyanide). Copper does not protect the nail from corrosion. 42
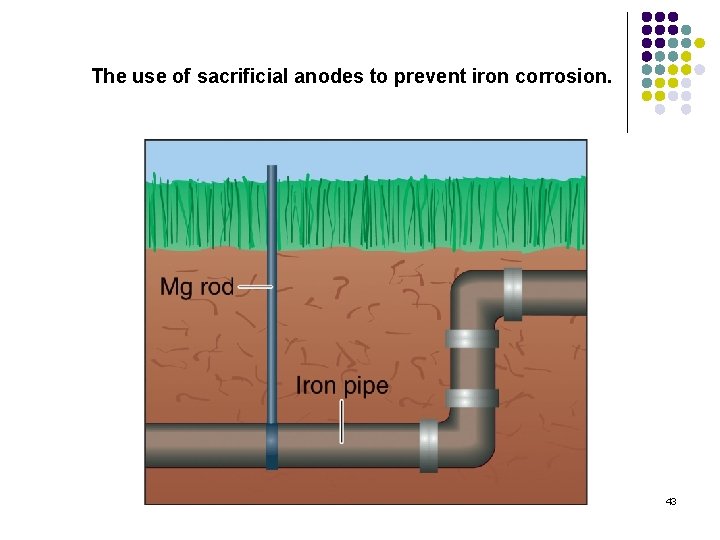
The use of sacrificial anodes to prevent iron corrosion. 43
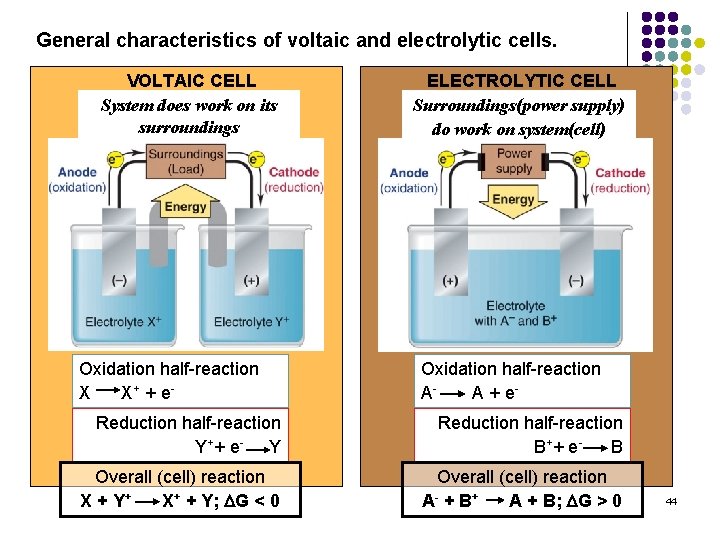
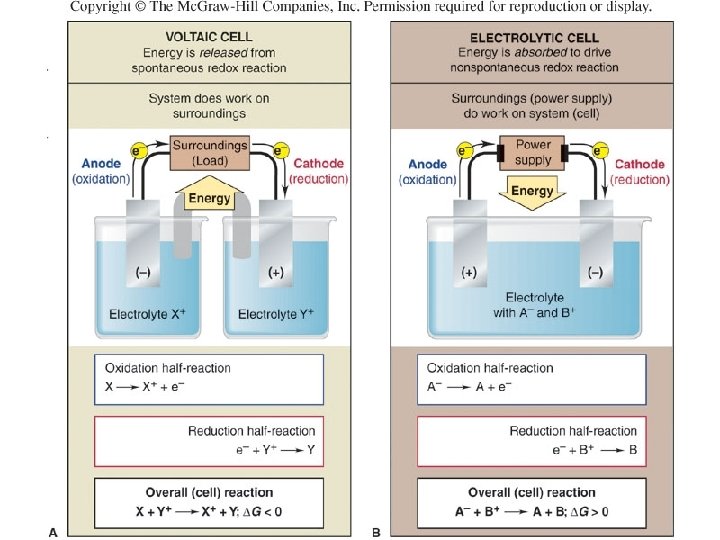
General characteristics of voltaic and electrolytic cells. VOLTAIC CELL System Energydoes is released work on from its spontaneous surroundings redox reaction Oxidation half-reaction X X + + e- ELECTROLYTIC CELL Surroundings(power Energy is absorbed tosupply) drive a nonspontaneous redox reaction do work on system(cell) Oxidation half-reaction AA + e- Reduction half-reaction Y+ + e - Y Reduction half-reaction B+ + e B Overall (cell) reaction X + Y+ X+ + Y; DG < 0 Overall (cell) reaction A- + B + A + B; DG > 0 44
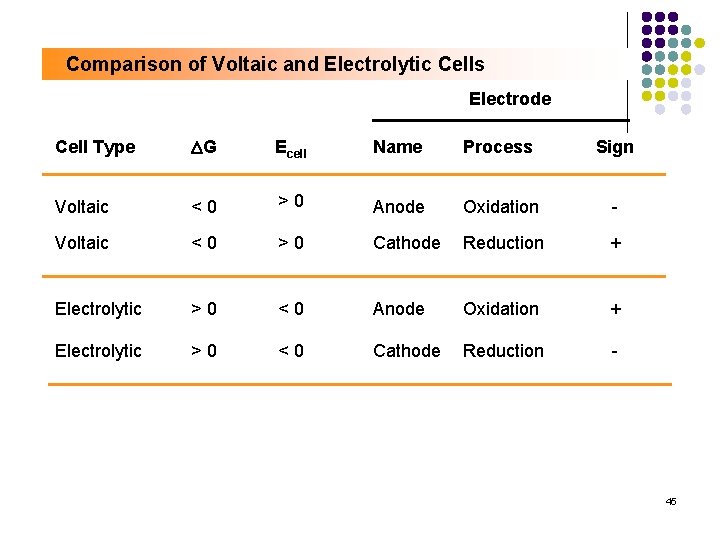
Comparison of Voltaic and Electrolytic Cells Electrode Cell Type DG Ecell Name Process Sign Voltaic <0 >0 Anode Oxidation - Voltaic <0 >0 Cathode Reduction + Electrolytic >0 <0 Anode Oxidation + Electrolytic >0 <0 Cathode Reduction - 45

Electrolysis of Pb. Br 2(s) Oxi: 2 Br- → Br 2(g) + 2 e. Red: Pb 2+ + 2 e- → Pb(s) Final: Pb 2+ + 2 Br- → Pb(s) + Br 2(g) 46

Electrolysis of Dilute H 2 SO 4 l l l Electrolysis of water H 2 SO 4 serves as a catalyst As the electrolysis proceeds, the H 2 SO 4 will become more and more concentrated as the water is used up. 47

The electrolysis of water. Oxidation half-reaction 2 H 2 O(l) 4 H+(aq) + O 2(g) + 4 e- Reduction half-reaction 2 H 2 O(l) + 4 e 2 H 2(g) + 2 OH-(aq) Overall (cell) reaction 2 H 2 O(l) 2 H 2(g) + O 2(g) 48

Electrolysis of KI(aq) 49

Electrolysis of KI(aq) Oxi: Red: Final: 2 I - → I 2 + 2 e 2 H + + 2 e - → H 2 2 H + + 2 I - → H 2 + I 2 50
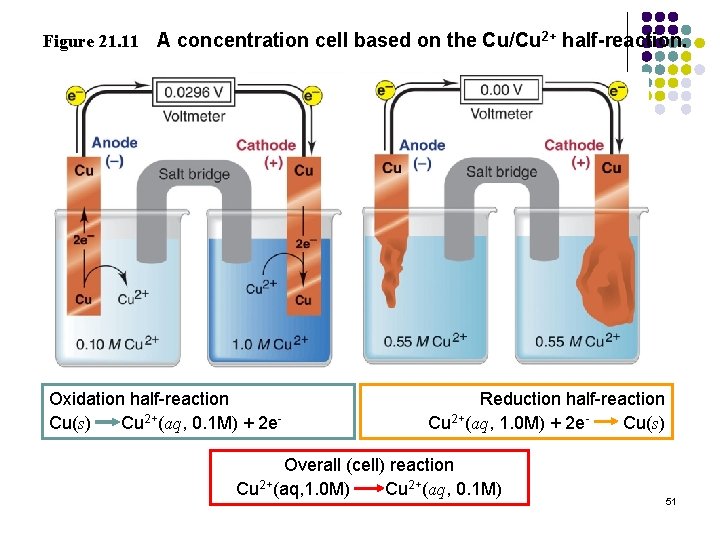
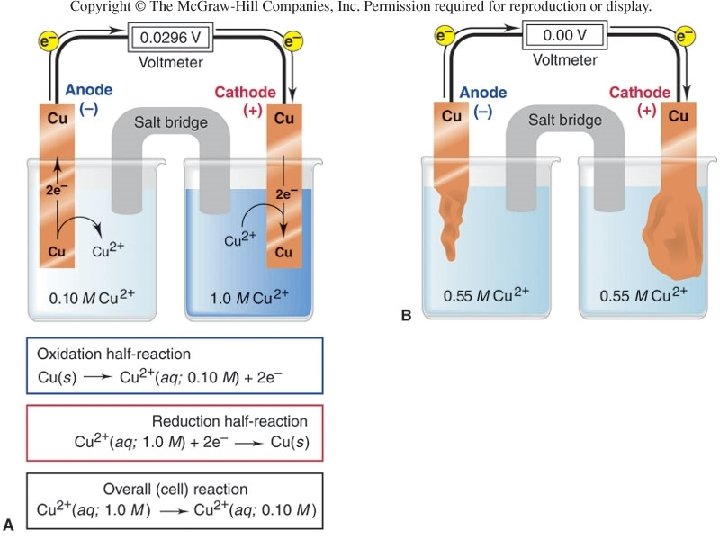
Figure 21. 11 A concentration cell based on the Cu/Cu 2+ half-reaction. Oxidation half-reaction Cu(s) Cu 2+(aq, 0. 1 M) + 2 e- Reduction half-reaction Cu 2+(aq, 1. 0 M) + 2 e. Cu(s) Overall (cell) reaction Cu 2+(aq, 1. 0 M) Cu 2+(aq, 0. 1 M) 51

Quantitative Aspects of Electrolysis l Faraday’s Law: the mass of product formed (or reactant consumed) at an electrode is proportional to both the amount of electricity transferred at the electrode and the molar mass of the substance in question. 52

E. g. 1) cathode reaction for Cu. SO 4 solution Cu 2+ + 2 e- → Cuo 2) cathode reaction for the electrolysis of molten Na. Cl Na+ + 1 e- → Nao l a) b) According to Faraday’s law: The mass of Na produced is proportional to its atomic mass divided by one, and the mass of Cu produced is proportional to its atomic mass divided by two. 1 mole of Cu ion reacts with 2 F of electricity to form 1 mole of Cu atom, and 1 mole of Na ion reacts with 1 F of electricity to form 1 mole of Na atom. 53

l l l Cu+2 + 2 e- → Cuo Na+ + 1 e- → Na Al 3+ + 3 e- → Alo 2 F = 1 mole Cu 2+ 1 F = 1 mole Na+ 3 F = 1 mole Al+3 54

In an electrolysis experiment, we generally measure the current (in amperes) that passes through an electrolytic cell in a given period of time. 1 C=1 AX 1 s that is, a coulomb is the quantity of electrical charge passing any point in the circuit in 1 second when the current is 1 ampere. 55
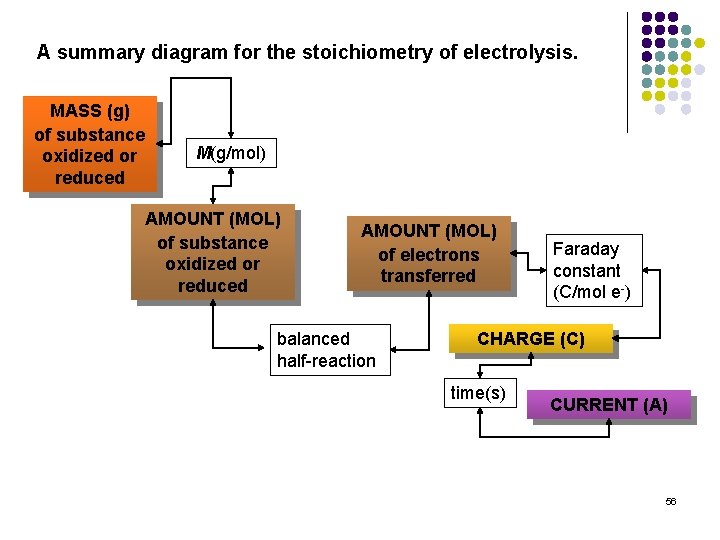
A summary diagram for the stoichiometry of electrolysis. MASS (g) of substance oxidized or reduced M(g/mol) AMOUNT (MOL) of substance oxidized or reduced AMOUNT (MOL) of electrons transferred balanced half-reaction Faraday constant (C/mol e-) CHARGE (C) time(s) CURRENT (A) 56

Problems: 1. A current of 0. 452 A is passed through an electrolytic cell containing molten Ca. Cl 2 for 1. 50 hours. Write the electrode reactions and calculate the quantity of products (in grams) formed at the electrodes. Anode: 2 Cl- → Cl 2 + 2 e. Cathode: Ca 2+ + 2 e- → Ca Final: Ca 2+ + 2 Cl- → Ca + Cl 2 57

Answers: ? C = 0. 452 A X 1. 50 hr X 3600 s 1 hr = 2. 44 X 103 C X 1 C 1 AXs ? g Ca = 2. 44 X 103 C X 1 F X 1 mol Ca 96, 500 C 2 F = 0. 507 g Ca X 40. 08 g Ca 1 mol Ca ? g Cl 2 = 2. 44 X 103 C X 1 F X 96, 500 C = 0. 896 g Cl 2 X 70. 90 g Cl 2 1 mol Cl 2 2 F 58

Problems: 2. If you wish to silver plate a spoon will you make it the cathode or the anode? How much Ag will be deposited by 0. 3 F of electric charge? Ans. The object to be plated is always made the cathode. Ag+ + 1 e- → Ago x = (0. 3 F) (1 mol Ag+ / 1 F) (107. 87 g Ag / 1 mol Ag+) x = 32. 4 g of Ag 59

Problems: 3. How many moles of electrons are needed to deposit 0. 45 g of aluminum from fused Al. Cl 3? Ans. Al 3+ + 3 e- → Alo x = (0. 45 g) (1 mol) 27 g Al x = 0. 05 mol e- (3 mol e-) 1 mol 60

Problems: 4. Calculate the time required for a current of 1. 8 Amperes to deposit 12. 5 grams of copper from Cu. SO 4 solution. Ans. Cu+2 + 2 e- → Cuo time = (g) (n) (F) (A) (M) = (12. 5 g) (2 e-) (96, 500 C) (1. 8 A) (64 g/mole) = 20, 942 secs. Or 5. 82 hours 61

Problems: 5. A constant current is passed through an electrolytic cell containing molten Mg. Cl 2 for 18 hours. If 4. 8 X 105 grams of Cl 2 are obtained, what is the current in Amperes? Ans. = 2. 0 X 104 A 62

63
64
65
66
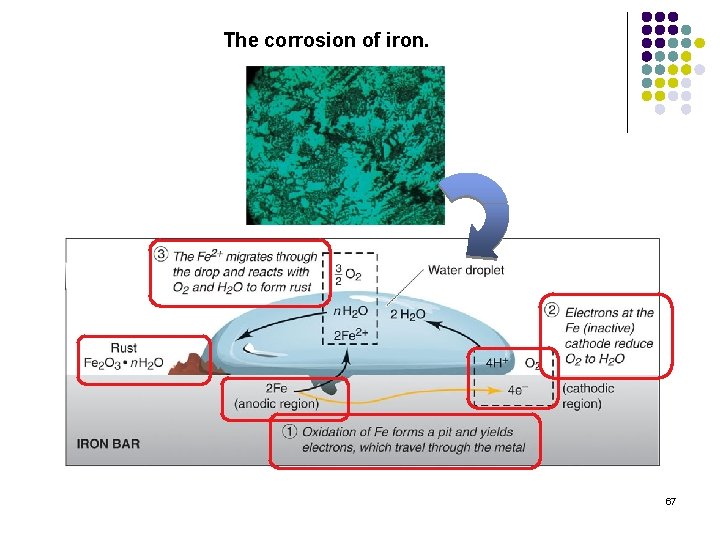
The corrosion of iron. 67

Enhanced corrosion at sea. 68
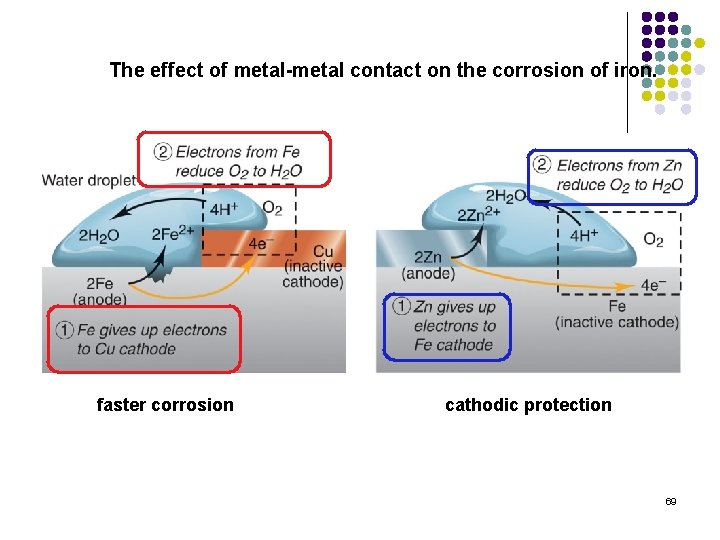
The effect of metal-metal contact on the corrosion of iron. faster corrosion cathodic protection 69
- Slides: 69