Optical Constants of Uranium Nitride Thin Films in






















- Slides: 40

Optical Constants of Uranium Nitride Thin Films in the EUV (80 -182 e. V) Marie K. Urry EUV Thin Film Group Brigham Young University

Acknowledgements Thanks to: Dr. David D. Allred Dr. R. Steven Turley Kristi R. Adamson Luke J. Bissell Jennie Guzman Elke Jackson Winston Larsen Mindy Tonks Department Funding

Outline Ø Why We Do What We Do Ø Making Thin Films Ø Studying Thin Films Ø Finding Optical Constants l Reflectometer

Why Extreme Ultraviolet (EUV)? Ø Astronomy l Ø Lithography l l Ø Our IMAGE Satellite Mirror Projection Imagining Scheduled for 2009 Medicine l High Resolution Imaging Microscopes Images courtesy of http: //euv. lpl. arizona. edu/euv/, www. schott. com/magazine/english/info 99/ and www. schott. com/magazine/english/info 99/.

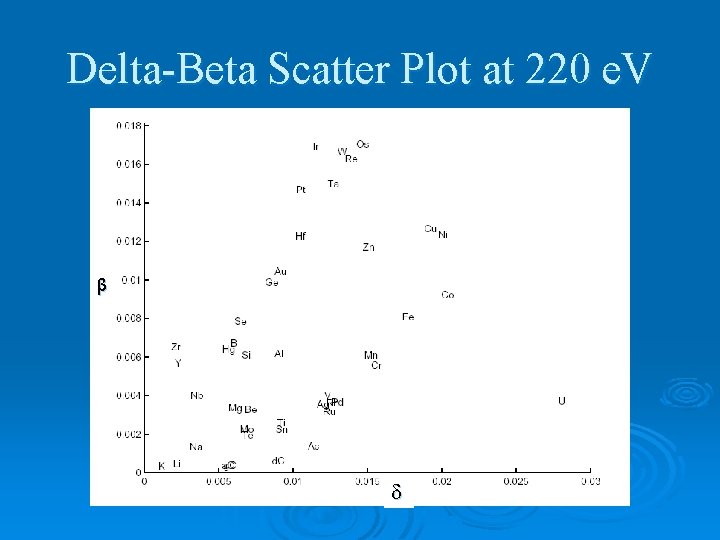
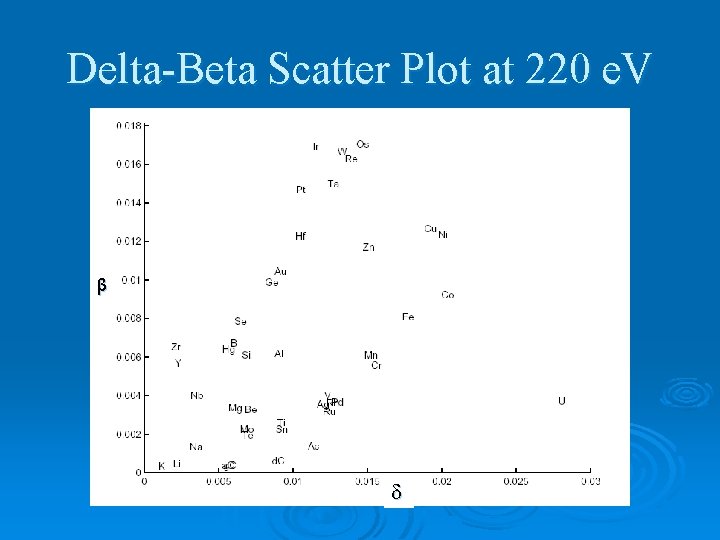
Optical Constants Ø Index of refraction: N = n + i k Ø In EUV, n ≈ 1 and k is huge. → n = 1 - k = Ø High and low for maximum reflection for multilayers.

Delta-Beta Scatter Plot at 220 e. V β

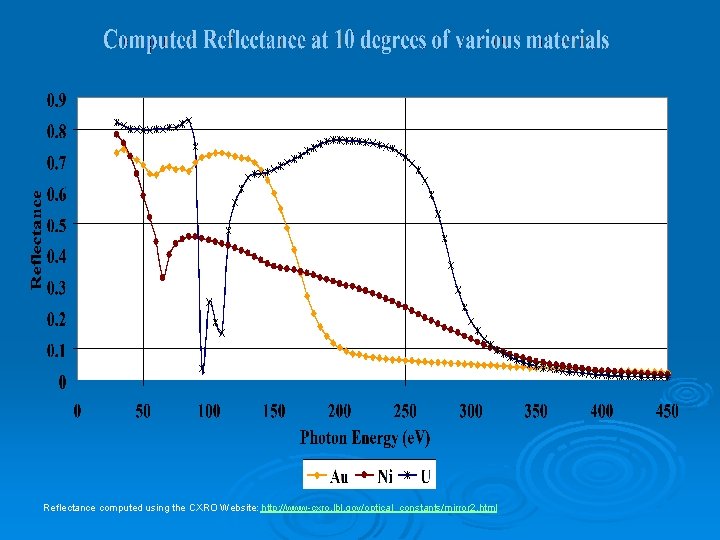
Why Uranium Nitride? Ø Uranium l High theoretical reflectivity due to high Ø Problem: Oxidation Ø Nitride l l Little effect on reflectivity Prevents oxidation

Reflectance computed using the CXRO Website: http: //www-cxro. lbl. gov/optical_constants/mirror 2. html

Reflectance computed using the CXRO Website: http: //www-cxro. lbl. gov/optical_constants/mirror 2. html

Delta-Beta Scatter Plot at 220 e. V β

Making Thin Films Ø Sputtering l l Bombard target, uranium, with argon ions Uranium atoms leave target due to collisions Nitrogen partial pressure in plasma introduces N atoms U and UN molecules deposit on our samples

Making Thin Films Ø 10 -30 nm thick Ø Deposited on: l l l silicon wafers quartz slides polyimide films Si. N membranes carbon coated TEM grids Ø Low pressure sputtering l smooth, dense, low stress films

Side Note for Clarification Ø Samples 002 – 005 l l 002 and 003 are U 2 N 3 004 and 005 are UN • 005 is new and unmeasured

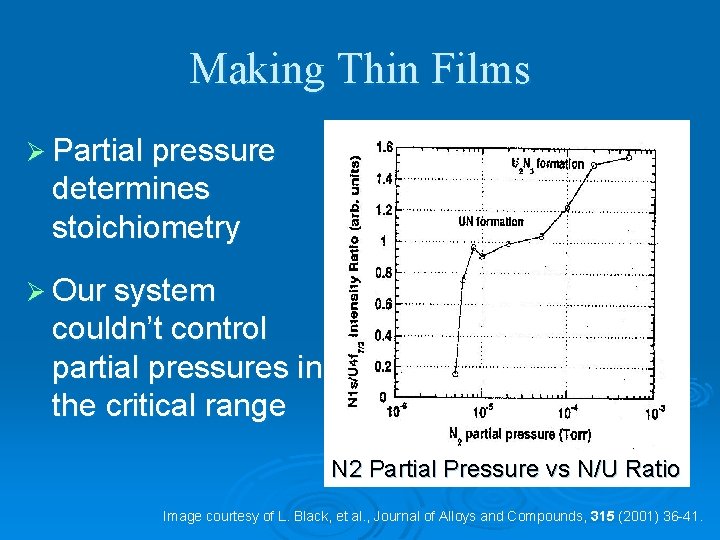
Making Thin Films Ø Partial pressure determines stoichiometry Ø Our system couldn’t control partial pressures in the critical range N 2 Partial Pressure vs N/U Ratio Image courtesy of L. Black, et al. , Journal of Alloys and Compounds, 315 (2001) 36 -41.

Learning About the Samples Ø Composition l Depends on partial pressure in system Ø Thickness l Crystal monitor is to the side of the film and gets less accurate with time Ø Roughness Ø Optical Constants

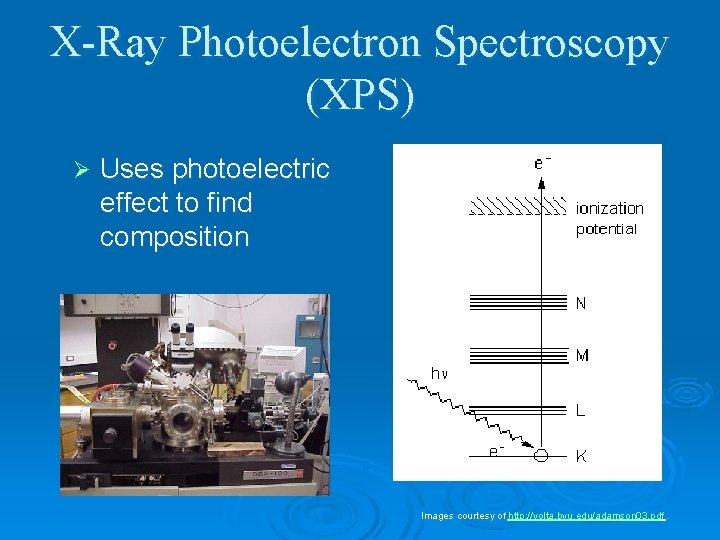
X-Ray Photoelectron Spectroscopy (XPS) Ø Uses photoelectric effect to find composition Images courtesy of http: //volta. byu. edu/adamson 03. pdf.

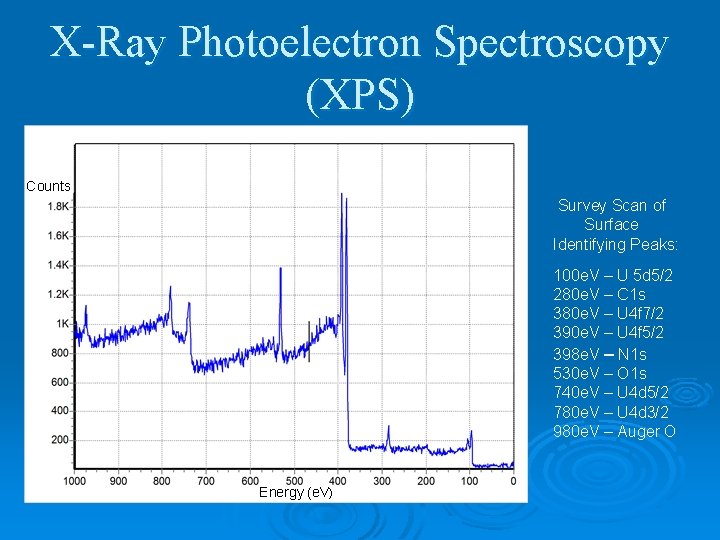
X-Ray Photoelectron Spectroscopy (XPS) Counts Survey Scan of Surface Identifying Peaks: 100 e. V – U 5 d 5/2 280 e. V – C 1 s 380 e. V – U 4 f 7/2 390 e. V – U 4 f 5/2 398 e. V – N 1 s 530 e. V – O 1 s 740 e. V – U 4 d 5/2 780 e. V – U 4 d 3/2 980 e. V – Auger O Energy (e. V)

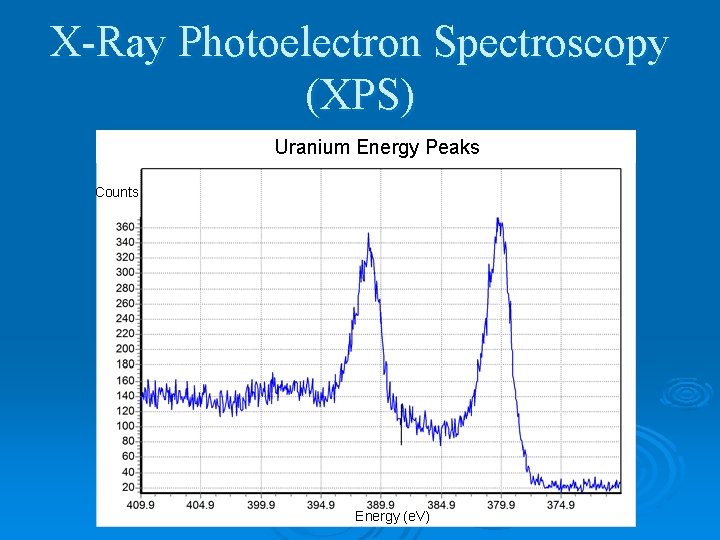
X-Ray Photoelectron Spectroscopy (XPS) Uranium Energy Peaks Counts Energy (e. V)

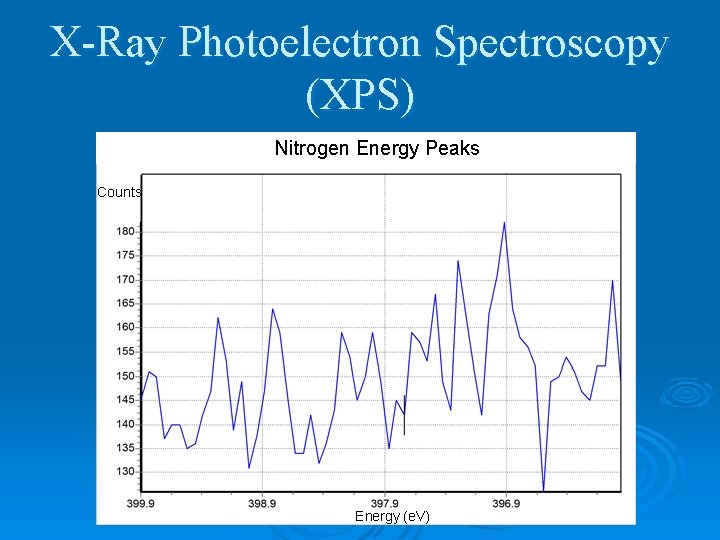
X-Ray Photoelectron Spectroscopy (XPS) Nitrogen Energy Peaks Counts Energy (e. V)

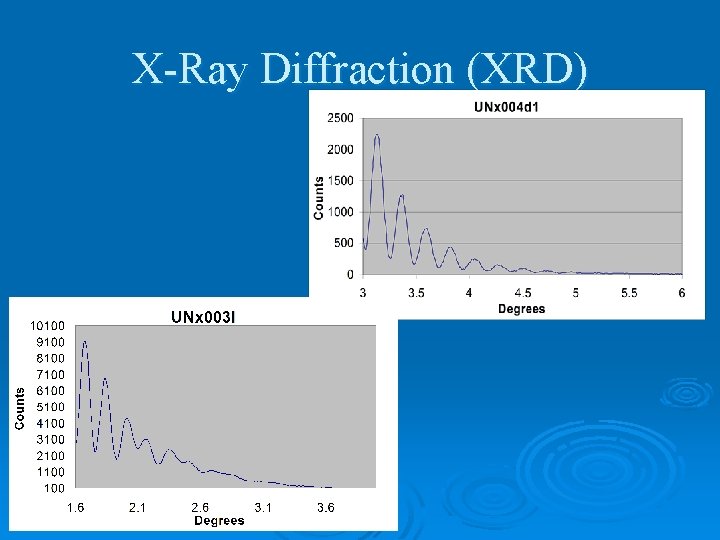
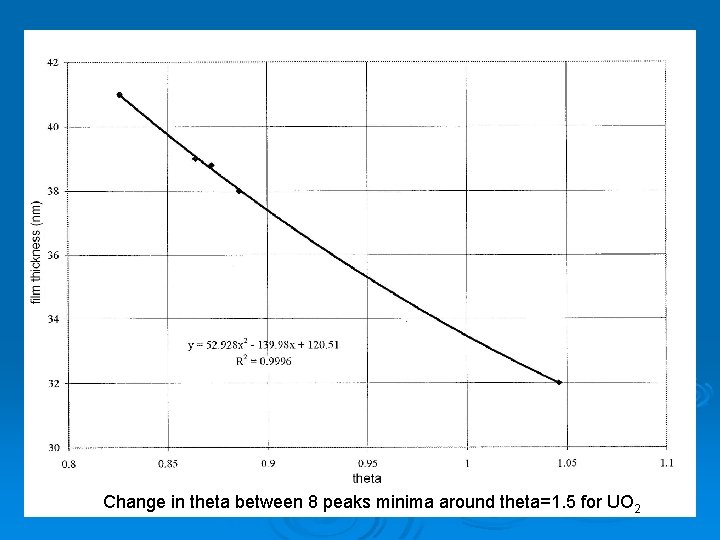
X-Ray Diffraction (XRD) To find thickness Ø m λ = 2 d = 2 sin θ sin Ø

XRD Data Change in theta between 8 peaks minima around theta=1. 5 for UO 2

XRD Data

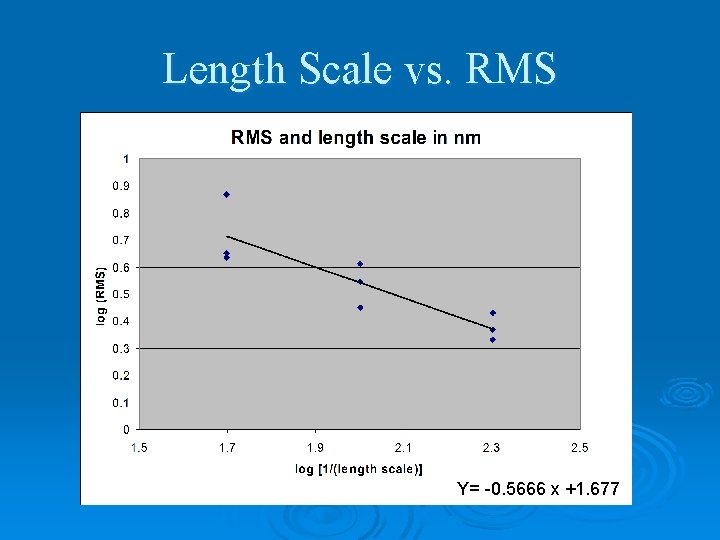
Atomic Force Microscopy (AFM) Ø To Measure Roughness Ø Result: RMS roughness Images courtesy of http: //www. weizmann. ac. il/surflab/peter/afmworks/.

Length Scale vs. RMS Y= -0. 5666 x +1. 677

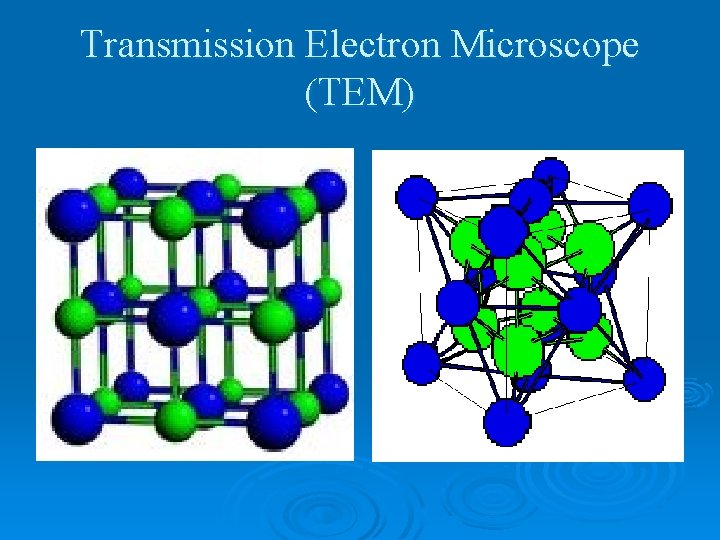
Transmission Electron Microscope (TEM)

Transmission Electron Microscope (TEM)

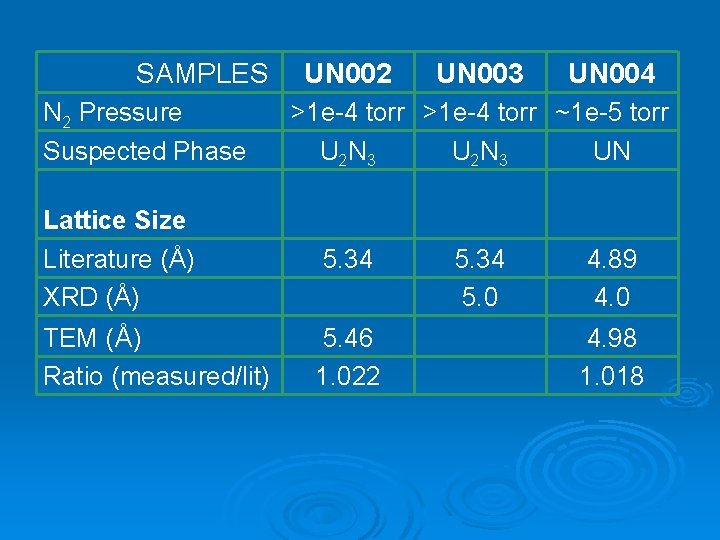
SAMPLES N 2 Pressure Suspected Phase UN 002 UN 003 UN 004 >1 e-4 torr ~1 e-5 torr U 2 N 3 UN Lattice Size Literature (Å) XRD (Å) 5. 34 5. 0 4. 89 4. 0 TEM (Å) Ratio (measured/lit) 5. 46 1. 022 4. 98 1. 018

Ellipsometry Ø Optical constants are different for different polarizations of light Ø If we know the substance and a model for the optical constants, we can find thickness and optical constants in UV Images courtesy of http: //www. swt. edu/~wg 06/manuals/Gaertner 117/ellipsometer. Home. htm.

Finding Optical Constants Ø Advanced Light Source at Berkeley l l Light created by synchrotron Measures reflectance at different angles and wavelengths Image courtesy of http: //www. lbl. gov/.

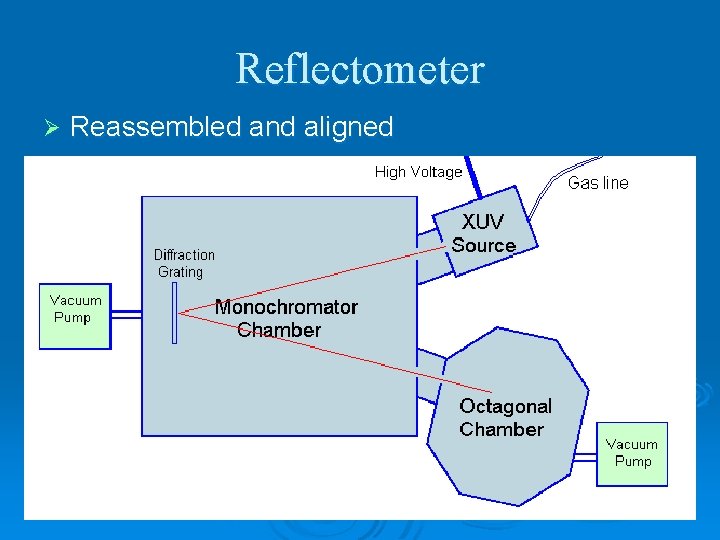
Reflectometer Ø Reassembled and aligned

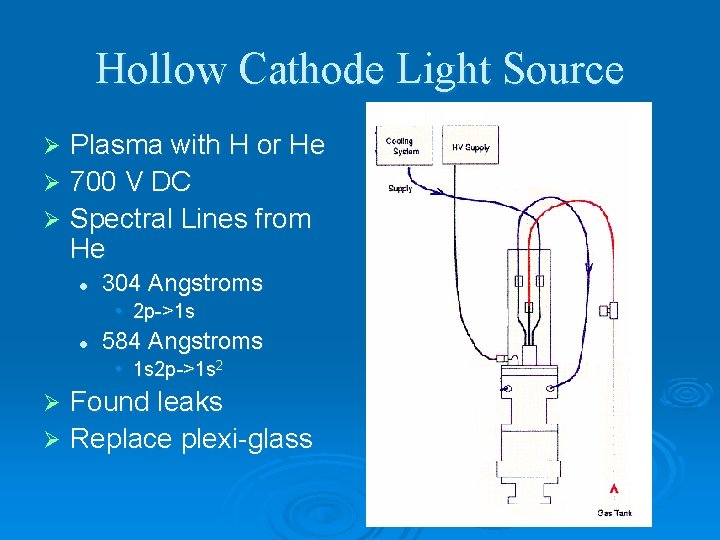
Hollow Cathode Light Source Plasma with H or He Ø 700 V DC Ø Spectral Lines from He Ø l 304 Angstroms • 2 p->1 s l 584 Angstroms • 1 s 2 p->1 s 2 Found leaks Ø Replace plexi-glass Ø

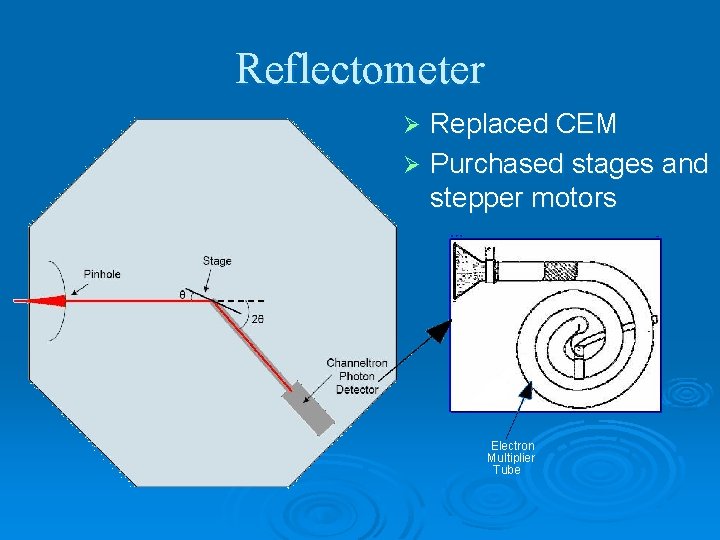
Reflectometer Replaced CEM Ø Purchased stages and stepper motors Ø Electron Multiplier Tube

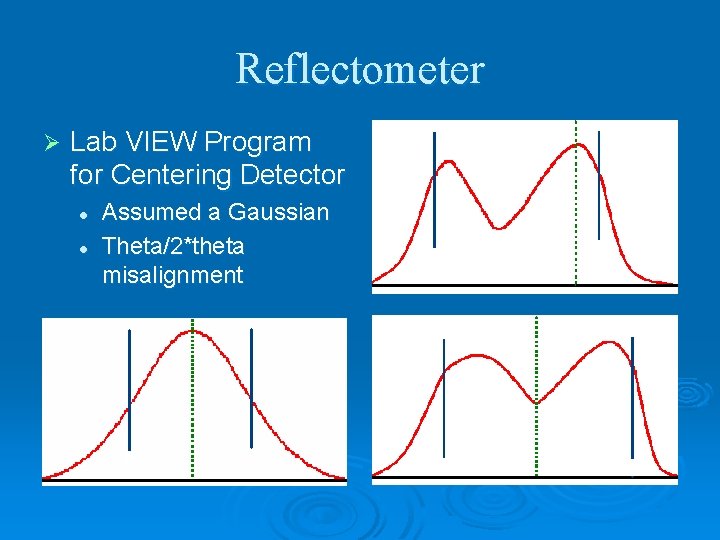
Reflectometer Ø Lab VIEW Program for Centering Detector l l Assumed a Gaussian Theta/2*theta misalignment

Reflectometer Ø Other Improvements l l Ø Circuit diagrams SOP’s Still working l Fixing virus problems

On to Berkeley!


Outline Ø Background (9 minutes) l l l Ø Why EUV? Optical Constants Why Uranium? Making & Studying Thin Films (13 minutes) l l l Ø Sputtering XPS XRD AFM TEM Ellipsometry Finding Optical Constants (10 minutes) l l l What we want to know ALS Reflectometer/ Monochromator Results/Continuing Research (8 minutes) Ø Acknowledgements (1 minute) Ø

To Do Ø Learn about light source (internet) Ø Ellipsometry stuff (Dr. Allred) Ø Update “Problem!!” and data slides (Dr. Allred)

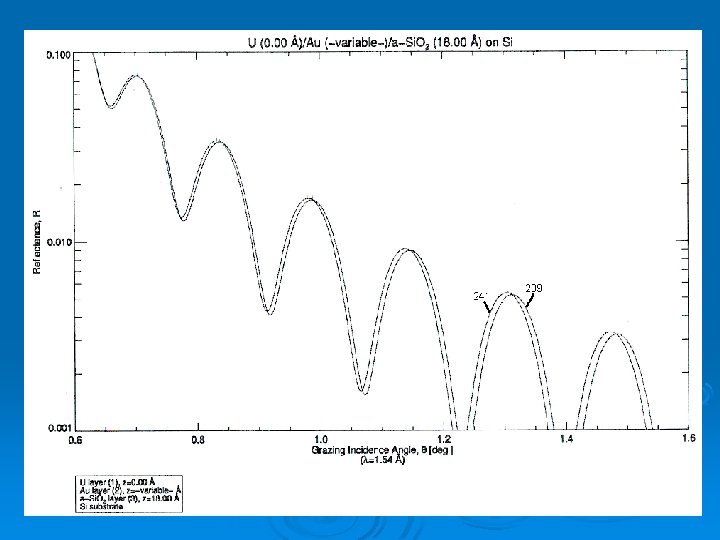
IMD Ø Written by David Wendt Ø Computes reflectivities of materials based on their optical constants Ø We used UO model because of similar densities Ø (Insert graph here)

Problem!! Ø Our samples change with time. l The peaks seen in XRD move. Ø Continuing research in this area.