MANAGEMENT OF CARDIAC SURGICAL PATIENTS RECEIVING PLATELET INHIBITORS

MANAGEMENT OF CARDIAC SURGICAL PATIENTS RECEIVING PLATELET INHIBITORS Jerrold H. Levy, MD Professor of Anesthesiology Emory University School of Medicine Director of Cardiothoracic Anesthesiology Emory Healthcare Atlanta, Georgia
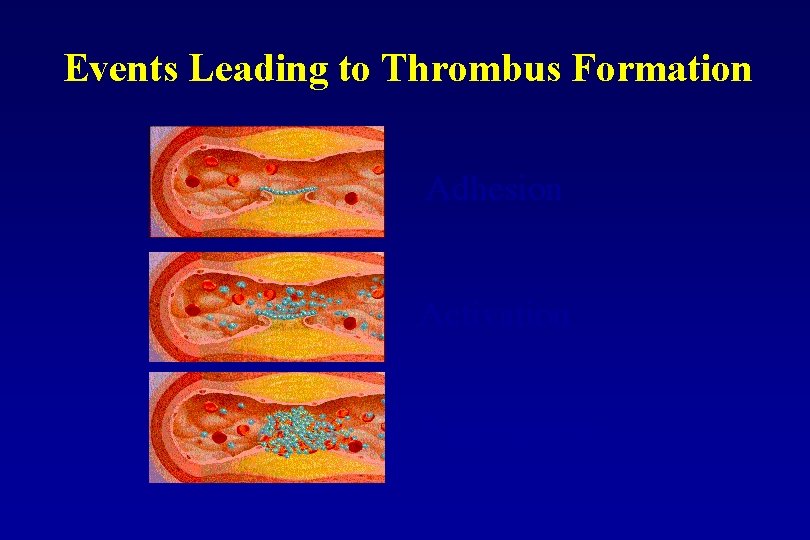
Events Leading to Thrombus Formation Adhesion Activation Aggregation
Platelet-fibrin clot

Gp IIb/IIIa ANTAGONISTS • Platelet Gp IIb/IIIa receptors play a pivotal role in platelet-mediated thrombus formation, binding to binds to fibrinogen and v. WF • IIb/IIIa antagonists differ in receptor affinity, reversibility, and specificity

PLATELET INHIBITORS • • • ASA Clopidogrel (Plavix), Ticlid Aggrastat (tirofiban) Reo. Pro (abciximab) Integrilin (eptifibatide)
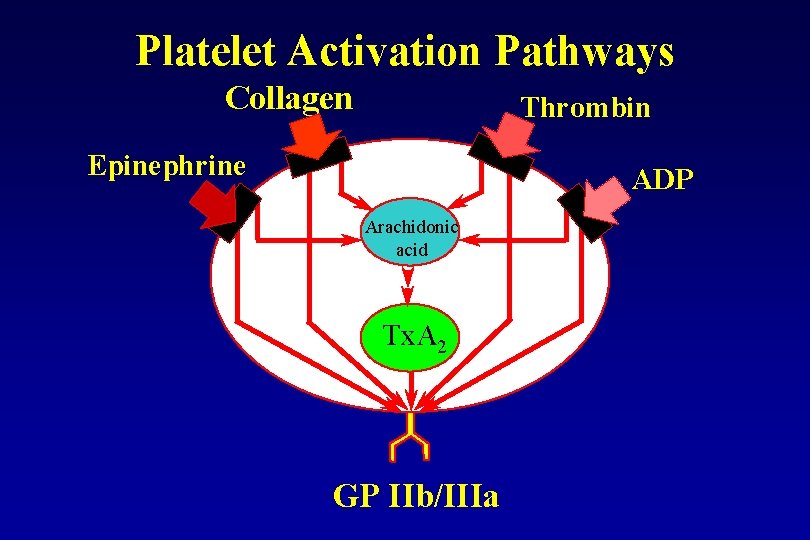
Platelet Activation Pathways Collagen Thrombin Epinephrine ADP Arachidonic acid Tx. A 2 GP IIb/IIIa
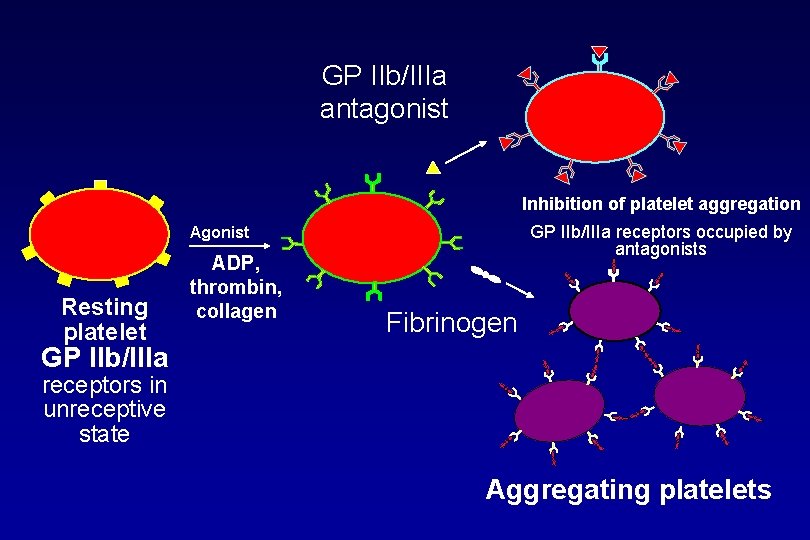
GP IIb/IIIa antagonist Inhibition of platelet aggregation GP IIb/IIIa receptors occupied by antagonists Agonist Resting platelet ADP, thrombin, collagen Fibrinogen GP IIb/IIIa receptors in unreceptive state Aggregating platelets

Tirofiban (Aggrastat) • Nonpeptide • KD 15 nmol/L • Indication: acute coronary syndrome

Eptifbatide (Integrelin) • Cyclic peptide • KD 120 nmol/L • Acute coronary syndrome

Abciximab (Reo. Pro) • Human/murine chimeric monoclonal antibody Fab • KD 5 nmol/L • Indication: PCI

• • • PLATELET DYSFUNCTION DURING CPB Hemodilution Contact activation Shear stresses Hypothermia Intrinsic/extrinsic defects Anticoagulation/reversal

PLATELET FUNCTION AGGREGATION • IIb/IIIa - fibrinogen interaction • Key step for hemostasis, part of final common pathway • Therapeutic target of inhibitors

PLATELET FUNCTION EVALUATION • • • Platelet count Bleeding time Aggregation TEG/Sono. Clot Platelet function assays Accumetrics

Accumetrics’ Ultegra System Step 1 Insert Cartridge Step 2 Insert whole blood sample Step 3 Read result in 60 seconds
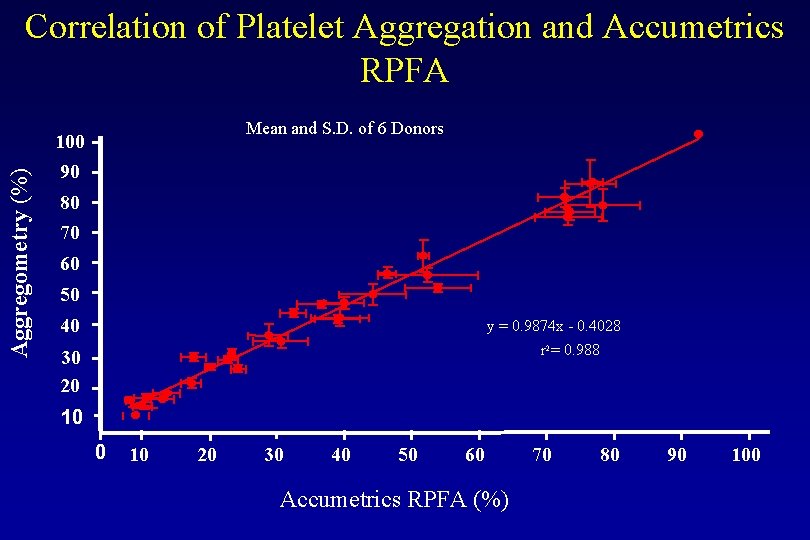
Aggregometry (%) Correlation of Platelet Aggregation and Accumetrics RPFA Mean and S. D. of 6 Donors 100 90 80 70 60 50 40 y = 0. 9874 x - 0. 4028 r 2 = 0. 988 30 20 10 20 30 40 50 60 Accumetrics RPFA (%) 70 80 90 100

Gammie: Abciximab and excessive bleeding in patients undergoing emergency cardiac operations. Ann Thor Surg 65: 465 -9, 1998 • 11 pts req emerg CABG, operated on <12 hr after abciximab (n = 6), or late >12 hr after abciximab (n = 5) • Postop drainage (1, 300 vs 400 m. L) • Tx p. RBC (6 versus 0 U; p = 0. 02), • Platelets transfused (20 versus 0 packs) • Max ACT (800 vs 528 sec; p = 0. 01)

Methods (EPILOG and EPISTENT Trials) • Patients undergoing CABG during index hospitalization • Data from both CRF andretrospective data collection at sites • Pooling of all abciximab tx groups and of all placebo groups in 39 sites • Most patients were unblinded undergoing CABG
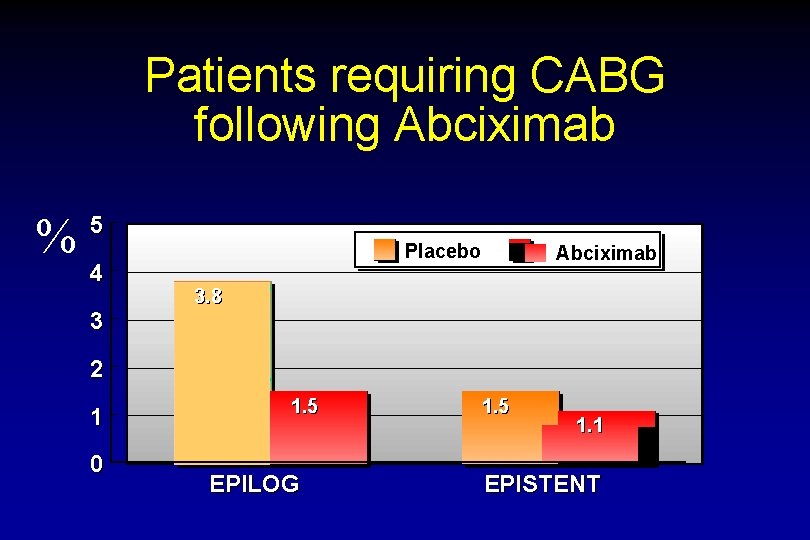
Patients requiring CABG following Abciximab % 5 4 3 Placebo Abciximab 3. 8 2 1 0 1. 5 EPILOG 1. 5 1. 1 EPISTENT
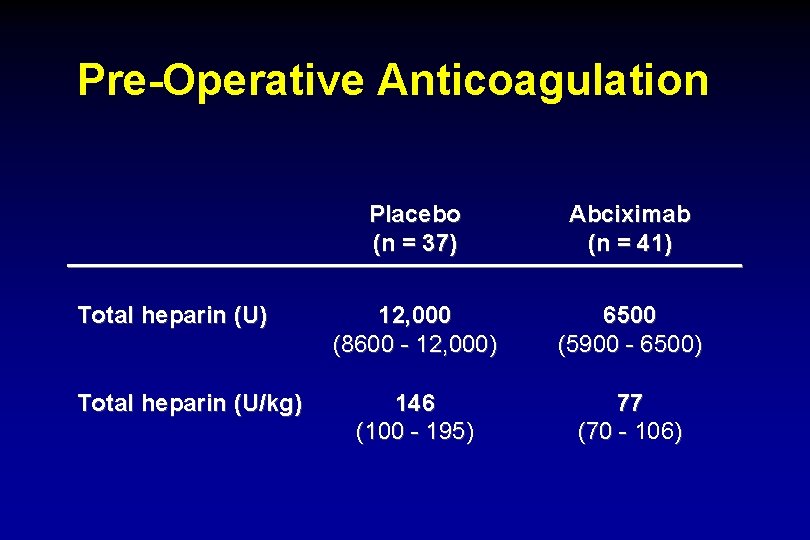
Pre-Operative Anticoagulation Total heparin (U) Total heparin (U/kg) Placebo (n = 37) Abciximab (n = 41) 12, 000 (8600 - 12, 000) 6500 (5900 - 6500) 146 (100 - 195) 77 (70 - 106)
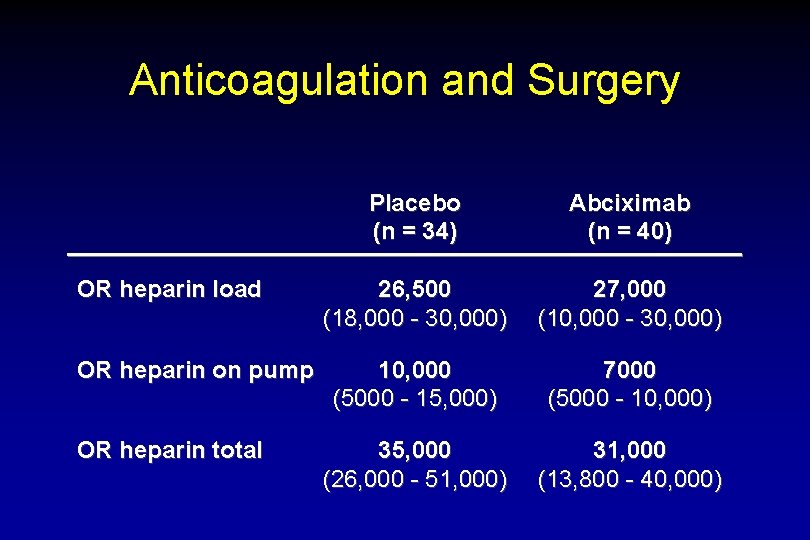
Anticoagulation and Surgery OR heparin load OR heparin on pump OR heparin total Placebo (n = 34) Abciximab (n = 40) 26, 500 (18, 000 - 30, 000) 27, 000 (10, 000 - 30, 000) 10, 000 (5000 - 15, 000) 7000 (5000 - 10, 000) 35, 000 (26, 000 - 51, 000) 31, 000 (13, 800 - 40, 000)
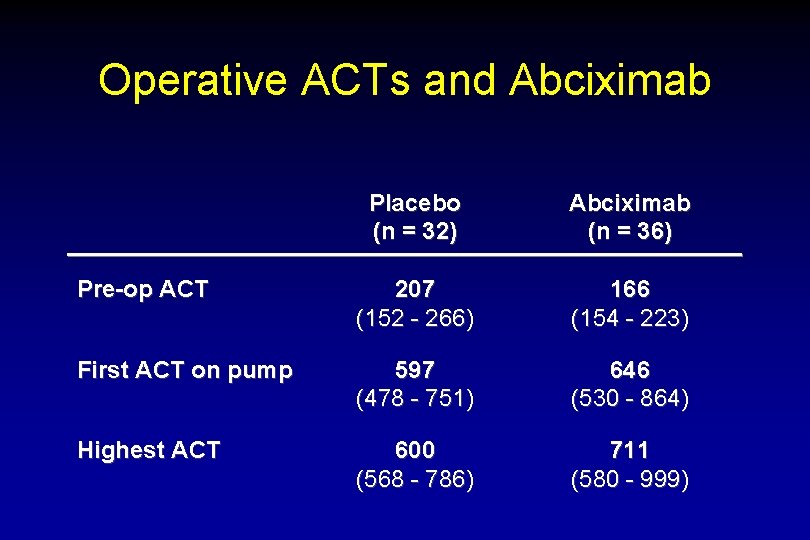
Operative ACTs and Abciximab Placebo (n = 32) Abciximab (n = 36) Pre-op ACT 207 (152 - 266) 166 (154 - 223) First ACT on pump 597 (478 - 751) 646 (530 - 864) Highest ACT 600 (568 - 786) 711 (580 - 999)
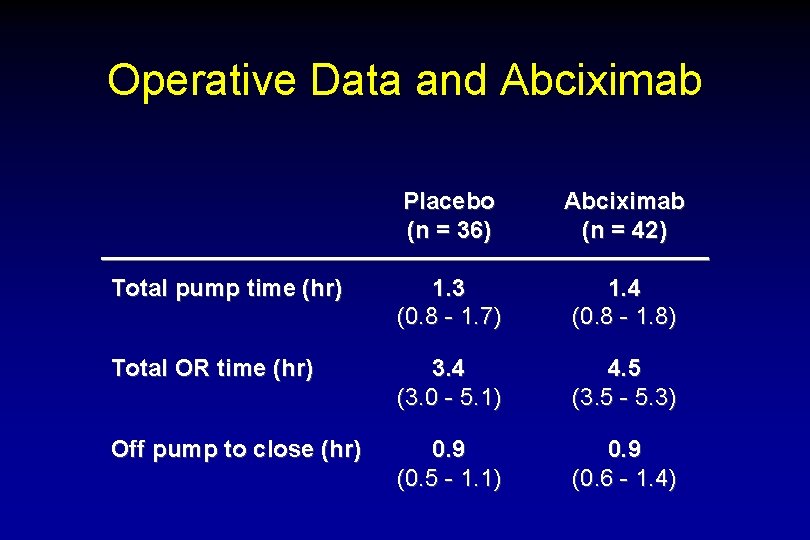
Operative Data and Abciximab Placebo (n = 36) Abciximab (n = 42) Total pump time (hr) 1. 3 (0. 8 - 1. 7) 1. 4 (0. 8 - 1. 8) Total OR time (hr) 3. 4 (3. 0 - 5. 1) 4. 5 (3. 5 - 5. 3) Off pump to close (hr) 0. 9 (0. 5 - 1. 1) 0. 9 (0. 6 - 1. 4)
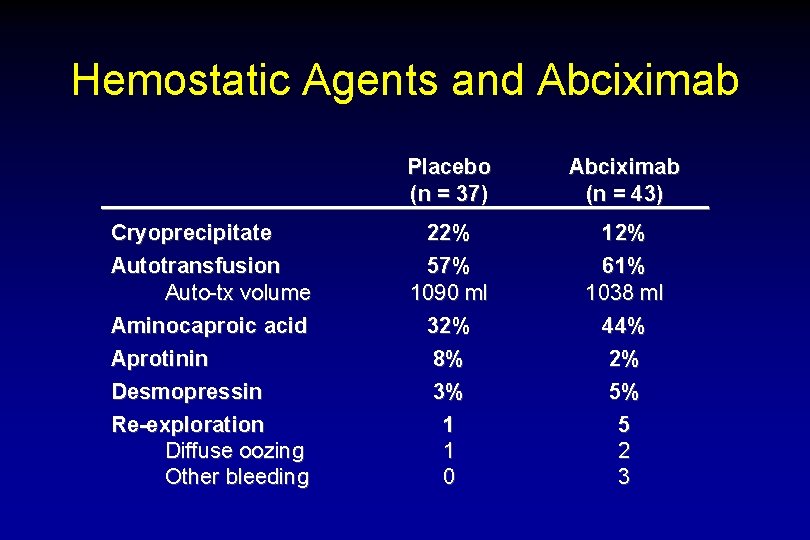
Hemostatic Agents and Abciximab Cryoprecipitate Autotransfusion Auto-tx volume Aminocaproic acid Aprotinin Desmopressin Re-exploration Diffuse oozing Other bleeding Placebo (n = 37) Abciximab (n = 43) 22% 57% 1090 ml 32% 8% 3% 1 1 0 12% 61% 1038 ml 44% 2% 5% 5 2 3
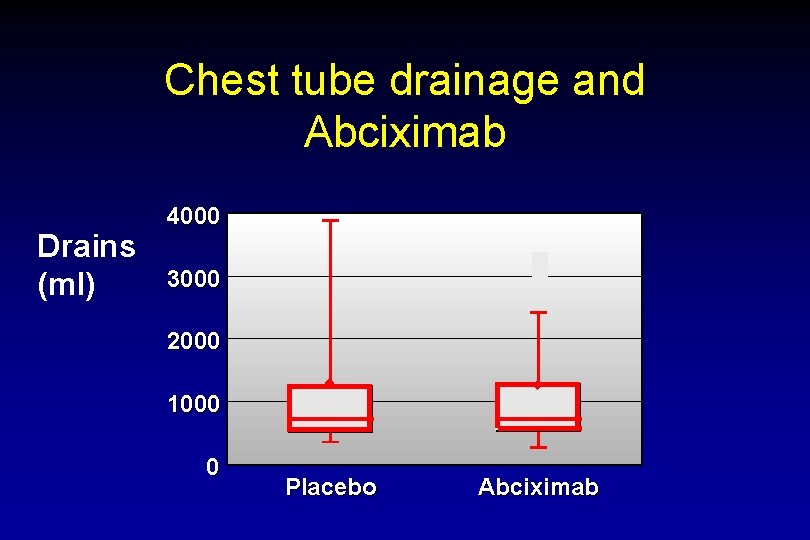
Chest tube drainage and Abciximab Drains (ml) 4000 3000 2000 1000 0 ¨ ¨ Placebo Abciximab

Abciximab and Bleeding Time to Surgery Time £ 12 hr Time > 12 hr Placebo Abciximab RBC Tx 78% 87% 69% 64% Plt Tx 44% 67% 13% 42% Major bleed 89% 96% 63% 75% Drain Blood Loss (ml) 730 870 1057 700 Hgb decrease (mg/dl) 7. 8 9. 4 7. 3 7. 1 72% 46% 17% 8% Death or MI
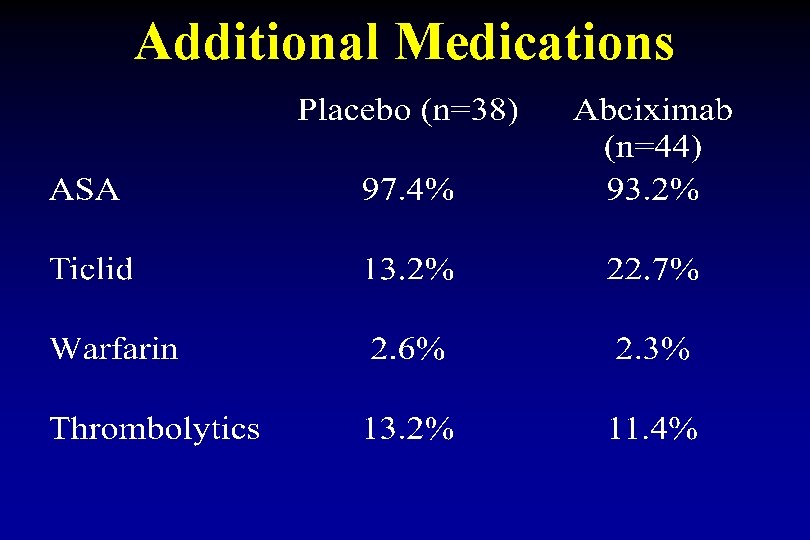
Additional Medications

Abciximab and CABG ¨Increased bleeding risk with urgent CABG ¨Abciximab therapy associated with minimal increase in blood loss with urgent CABG with conventional heparin dosing and platelet Tx transfusions ¨Patients requiring surgery in first 12 hours are at highest risk

TICLOPIDINE AND CLOPIDOGREL • Antiplatelet agents are used to treat, prevent arterial thrombosis. • Thienopyridine derivatives, inactive in vitro, requiring metabolism to achieve in vivo activity. • Inhibit binding of ADP to platelet receptor, inhibiting fibrinogen binding to the IIb/IIIa complex.

TICOLPIDINE/CLOPIDOGREL • In CAD stenting, ticlopidine reduces risk for subacute stent thrombosis • Clopidogrel reduces ischemic events with recent MI, stroke, or PVD • Clopidogrel + aspirin in stenting, is rapidly growing, given before stenting procedure • Bleeding variability for cardiac surgery relates to the duration of therapy

TICOLPIDINE and CABG Anesth Analg 1999; 88: SCA 105 • 96/1166 CABG pts receiving ticlopidine • 83% of ticlop pt also on ASA, 28% ticlop pt were urgent vs 9% • Blood loss >1500 ml/24 hr more frequent in ticlop (14% vs 5%) • 62% ticlop pts received allogneic blood vs 45% • p. RBC Tx 2 units vs 0 • Post op CT drain >30% in ticlop

HEMOSTATIC GOALS FOR CARDIAC SURGERY • Prevent clotting for cannulation and initiation of extracorporeal circulation • Reverse anticoagulation in a safe and complete manner. • Prevent the inflammatory effects of CPB and contact activation

FACTORS AFFECTING ACT • Factor deficiency: fibrinogen, XII, VIII • Contact activation inhibitors: aprotinin • Warfarin therapy • Heparin therapy • Hypothermia • Thrombocytopenia/cytosis • Platelet inhibitors

RECOMMENDATIONS FOR MANAGING PATIENTS RECEIVING ANTIPLATELET AGENTS AND REQUIRING CARDIAC SURGERY:

SAFETY Based on the data in press and published, urgent cardiac surgery can be safely performed on patients who have received abciximab or one of the other Gp. IIb/IIIa receptor inhibitors.

BLEEDING Although the relative risk of abciximab-related bleeding may be increased within 12 hrs, this should not preclude urgent CABG. Platelets may be needed, and should be available when operating on abciximab-tx pts.

HEPARIN DOSING There are no data to support reductions in heparin dosing during CPB and for cardiac surgery. Therefore, standardloading doses should be considered and additional heparin doses, based on time and duration of bypass or on actual heparin levels, should be maintained.

PLATELETS Platelets can be transfused to correct the bleeding defects associated with abciximab use. However, patients should not receive routine platelet transfusion prior to surgery and CPB. Rather, platelets should be administered after heparin reversal by protamine and after CPB.

SUMMARY: PLATELET INHIBITORS AND CARDIAC SURGERY • Do not transfuse with platelets before CPB • Normal heparin doses • Platelet transfusions when needed after CPB
- Slides: 38