LIGHT and QUANTIZED ENERGY Much of our understanding




























- Slides: 60

LIGHT and QUANTIZED ENERGY

Much of our understanding of the electronic structure of atoms has come from studying how substances absorb or emit light. To understand electric structure, therefore, we must first understand light.

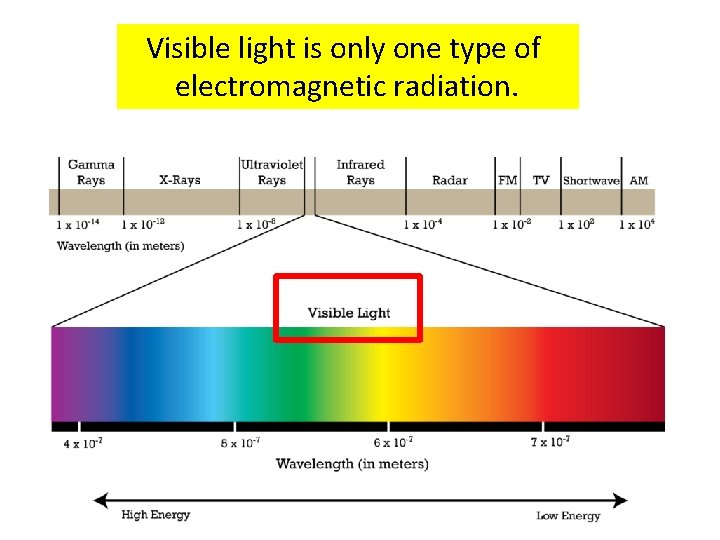
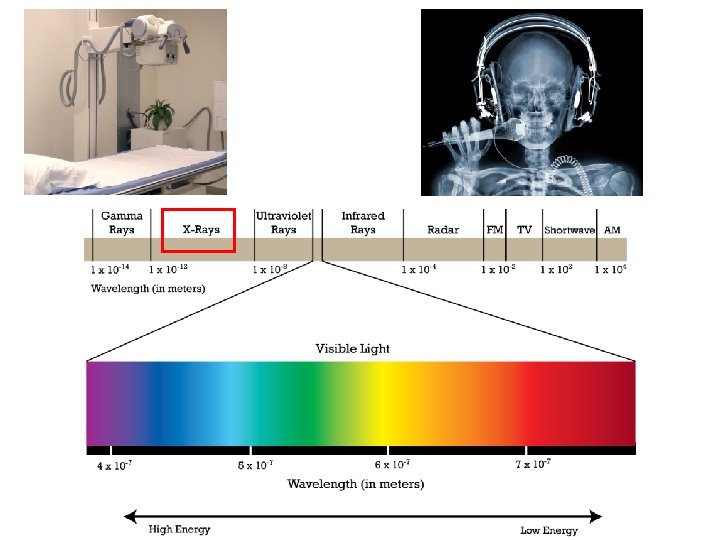
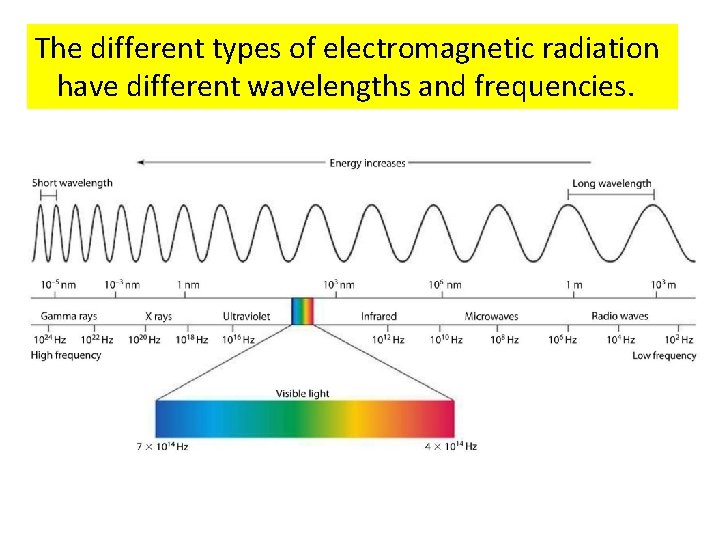
Visible light is only one type of electromagnetic radiation.

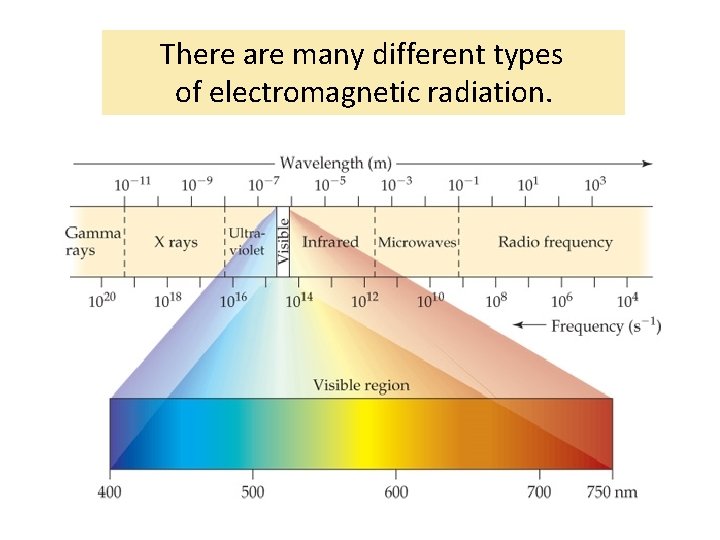
There are many different types of electromagnetic radiation.

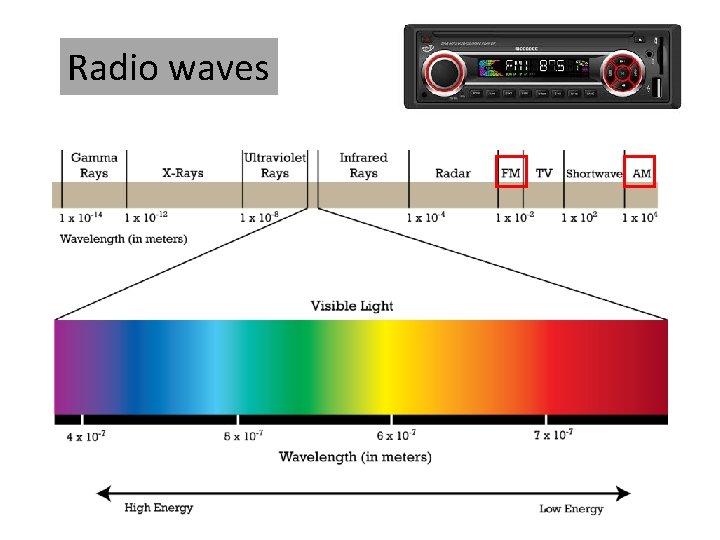
Radio waves



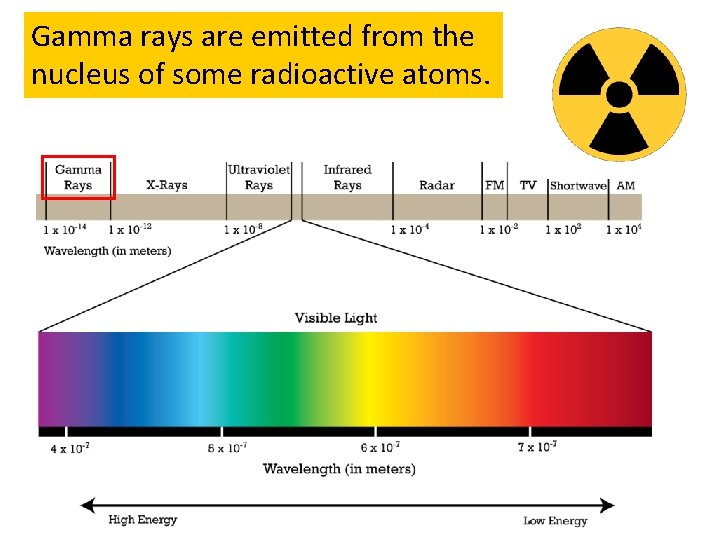
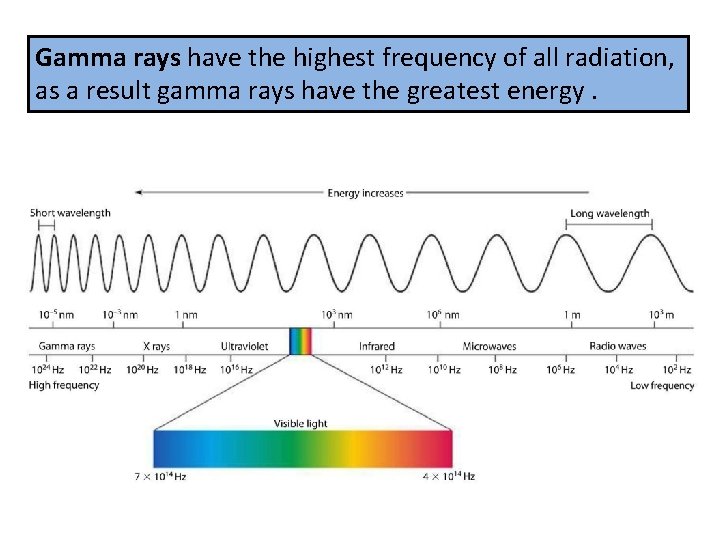
Gamma rays are emitted from the nucleus of some radioactive atoms.

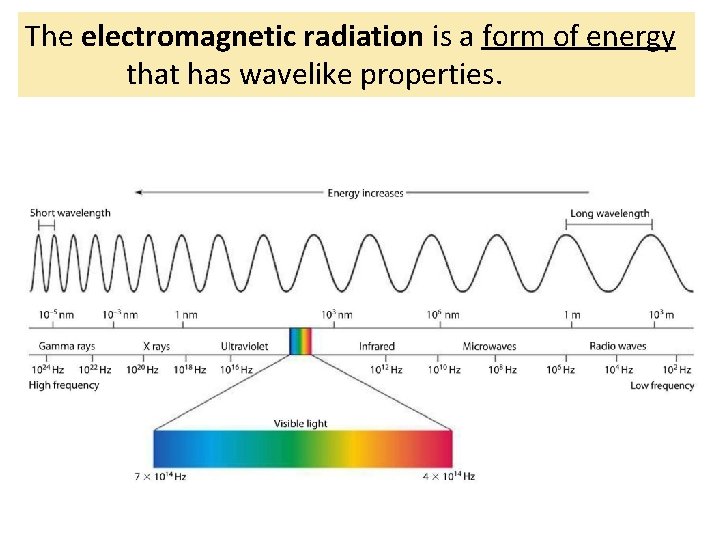
The electromagnetic radiation is a form of energy that has wavelike properties.

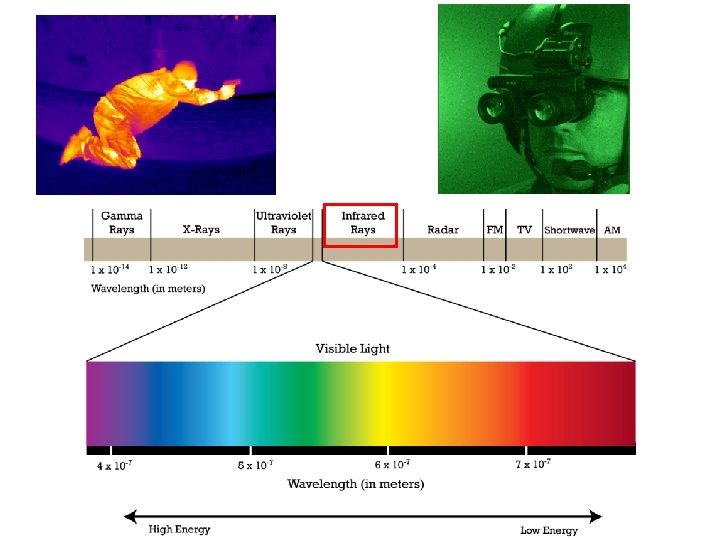
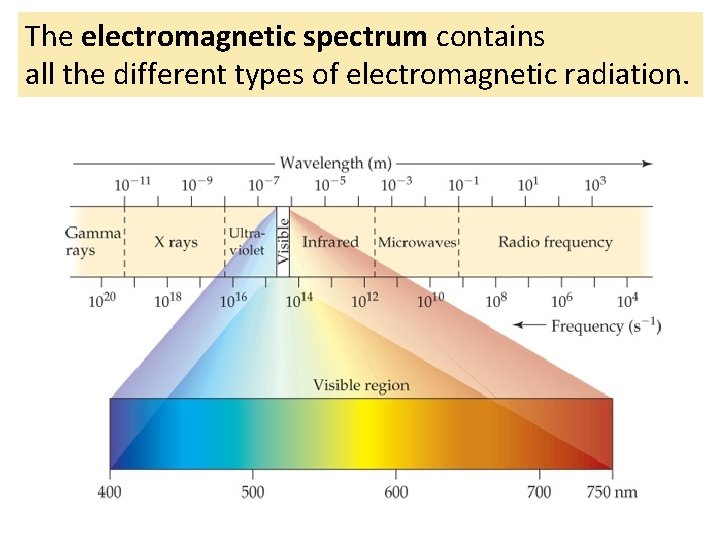
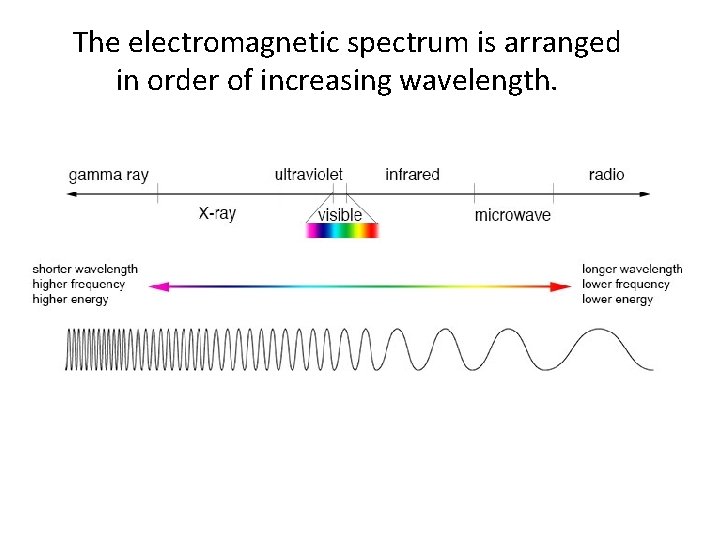
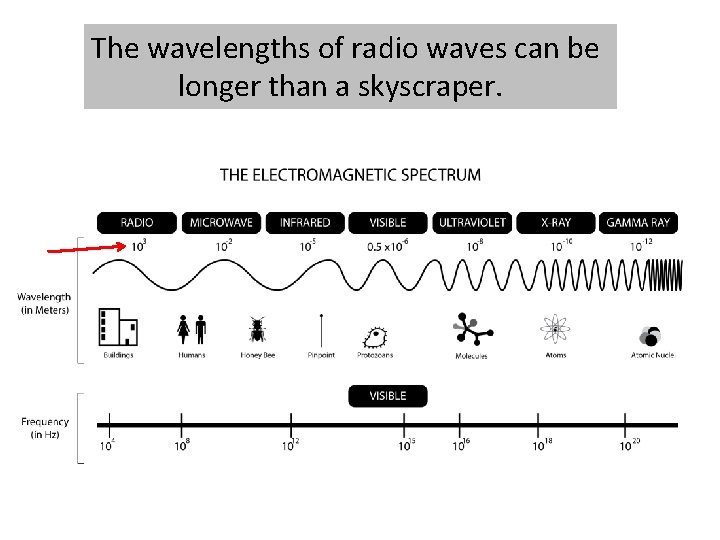
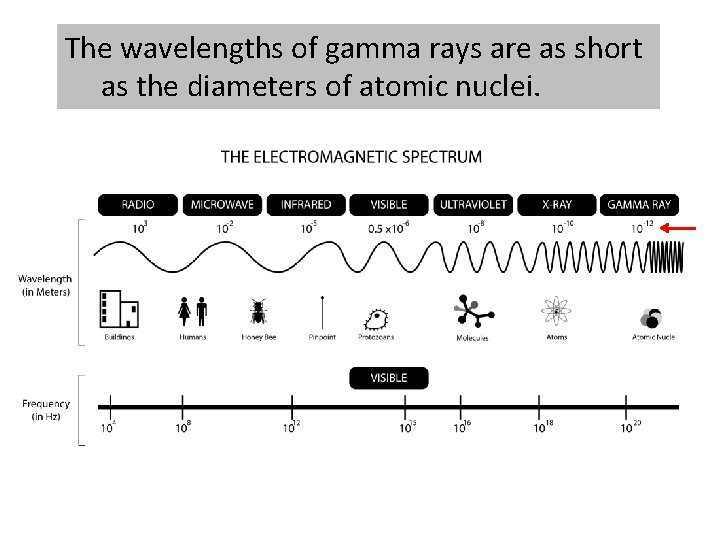
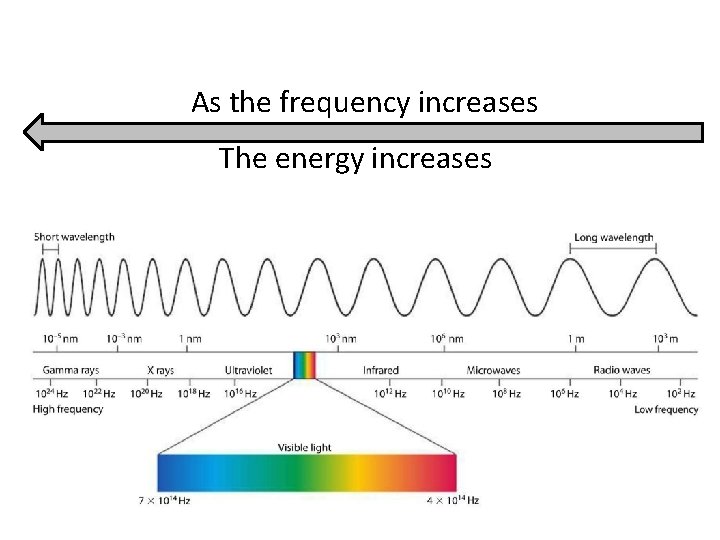
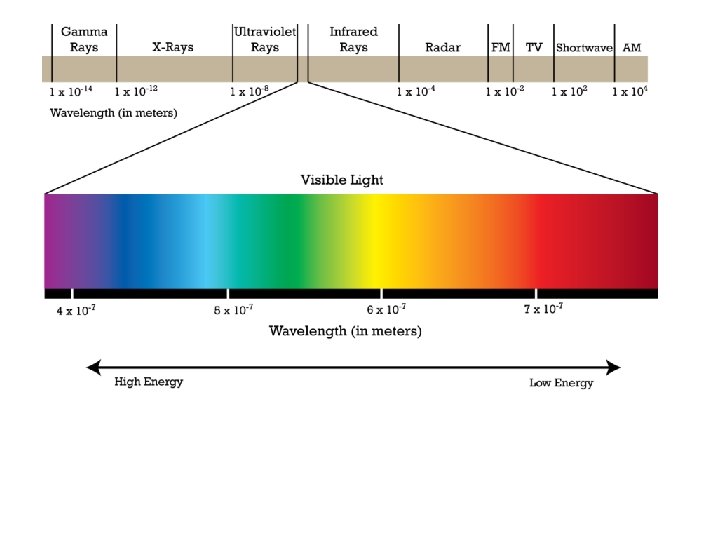
The electromagnetic spectrum contains all the different types of electromagnetic radiation.

The different types of electromagnetic radiation have different wavelengths and frequencies.

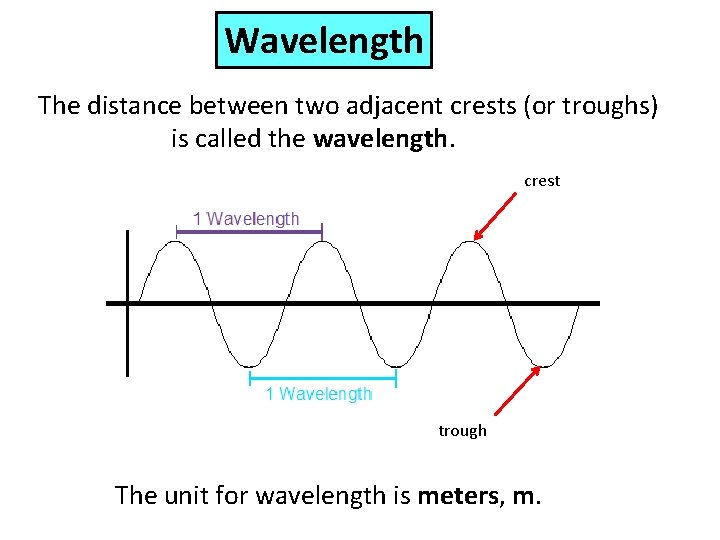
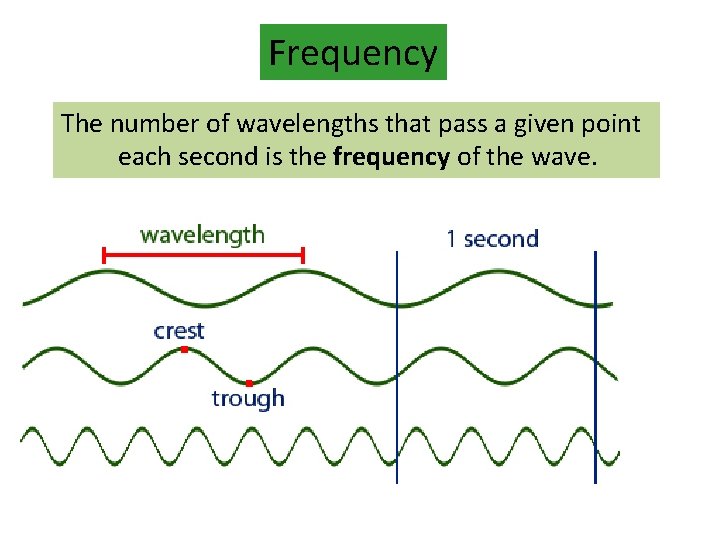
Wavelength The distance between two adjacent crests (or troughs) is called the wavelength. crest trough The unit for wavelength is meters, m.


The electromagnetic spectrum is arranged in order of increasing wavelength.

The wavelengths of radio waves can be longer than a skyscraper.

The wavelengths of gamma rays are as short as the diameters of atomic nuclei.

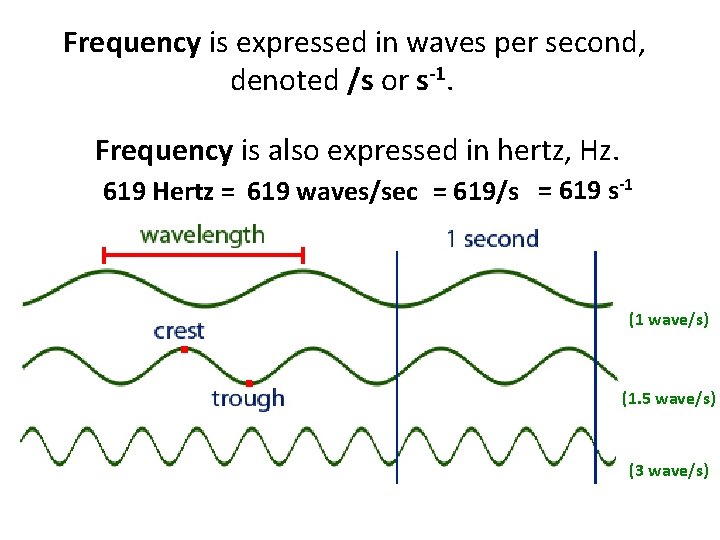
Frequency The number of wavelengths that pass a given point each second is the frequency of the wave. (1 wave/s) (1. 5 wave/s) (3 wave/s)

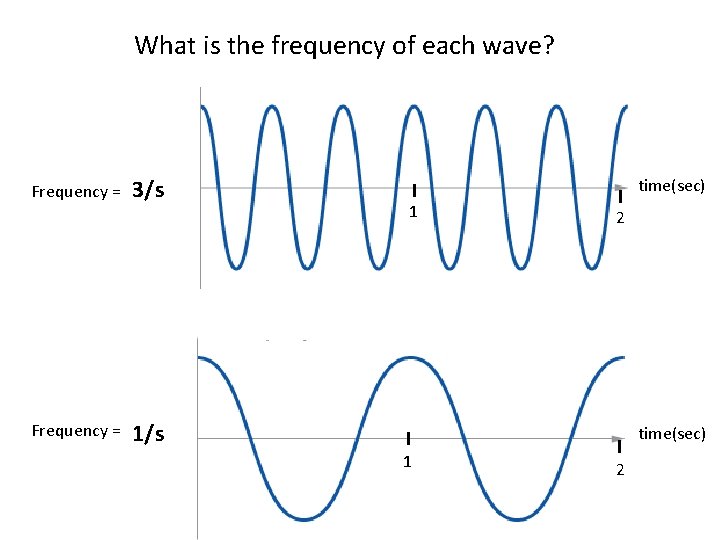
What is the frequency of each wave? Frequency = 3/s ? Frequency = time(sec) 1 2 ? 1/s time(sec) 1 2

Frequency is expressed in waves per second, denoted /s or s-1. Frequency is also expressed in hertz, Hz. 619 Hertz = 619 waves/sec = 619/s = 619 s-1 (1 wave/s) (1. 5 wave/s) (3 wave/s)

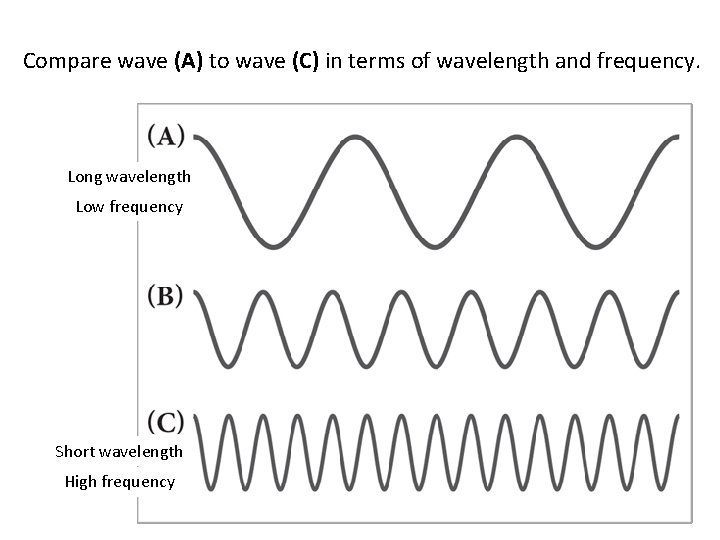
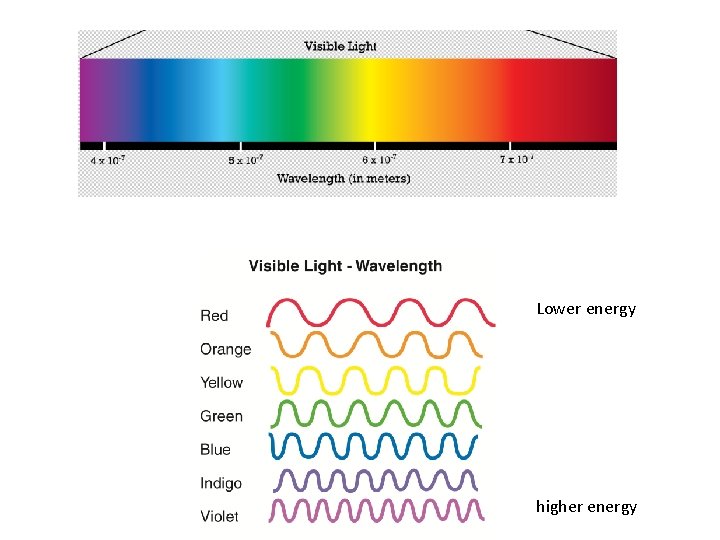
Compare wave (A) to wave (C) in terms of wavelength and frequency. Long wavelength Low frequency Short wavelength High frequency

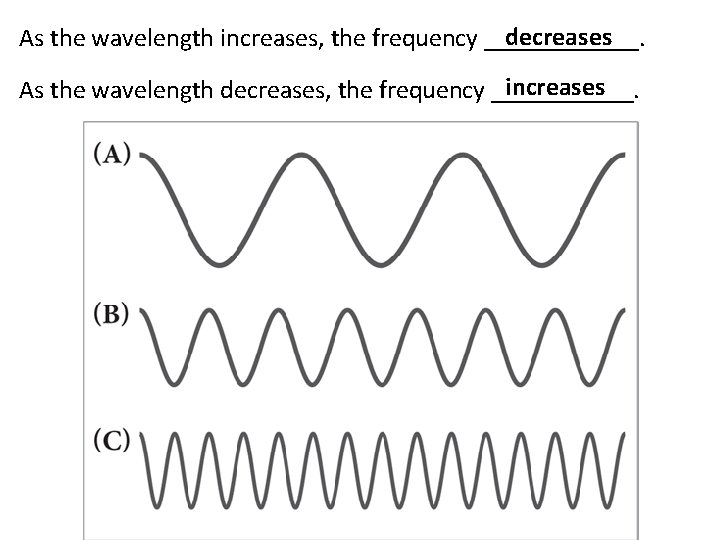
decreases As the wavelength increases, the frequency ______. increases As the wavelength decreases, the frequency ______.

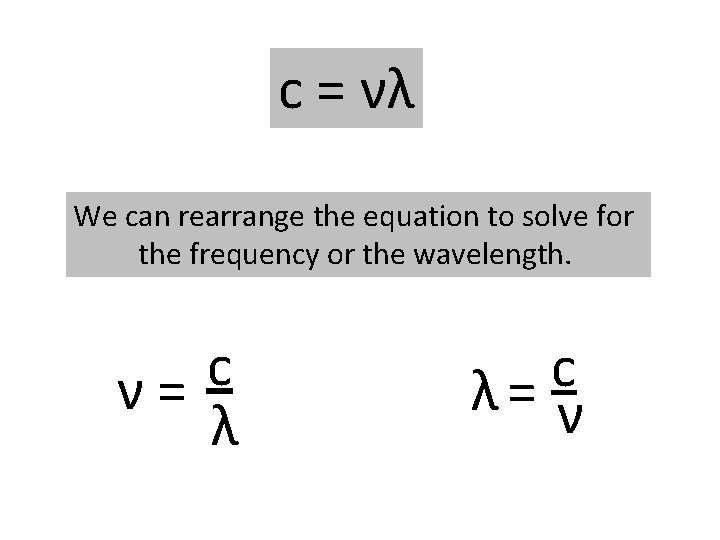
All types of electromagnetic radiation move through a vacuum at a speed of 3. 00 x 108 m/s, the speed of light. As a result, the wavelength and frequency of electromagnetic radiation are related by: Speed of light Wavelength (lambda) c = νλ Frequency (nu)

c = νλ We can rearrange the equation to solve for the frequency or the wavelength. c ν= λ c λ= ν

c ν= λ c λ= ν The wavelength and the frequency are inversely related. As one increases the other decreases. As one decreases the other increases.

c = νλ We can rearrange the equation to solve for the frequency or the wavelength.

Calculate the wavelength of the yellow light emitted by a sodium lamp if the frequency of the radiation is 5. 09 x 1014 Hz (5. 09 x 1014 /s). Given: ν = 5. 09 x 1014 /s c = 3. 00 x 108 m/s λ=? Equation: c λ= ν

An gamma ray has a wavelength of 4. 1 x 10 -12 m. What is the frequency? Given: Equation:

A radio station broadcasts at a frequency of 590 k. Hz. What is the wavelength of the radio waves? Given: Equation:

WHITEBOARD PRACTICE

1. What is the frequency of orange light, which has a wavelength of 6. 09 x 10 -7 m? Given: Equation:

2. An electromagnetic wave has a frequency of 8. 70 X 1018 /s. What is its wavelength? Given: Equation:

3. The yellow light given off by a sodium vapor lamp used for public lighting has a wavelength of 589 nm. What is the frequency of this radiation? Given: Equation:

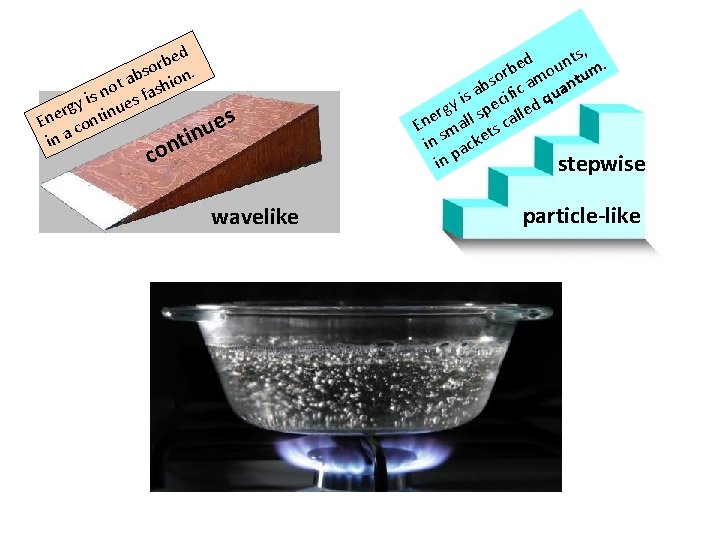
The Particle Nature of Light Thus far, we have learned that light and other radiation behave like waves. But light and other radiation also behave as if composed of particles or rather packets of energy. ed b r bso ion. a t no s fash s i gy inue r e s e En cont u tin in a con , d unts. e rb mo tum o s ab ific a quan s i y pec led g r s l e En mall ts ca s in acke p in stepwise

ed b r so on. b a t hi o s n a is sf y e g u r n s Ene conti e u a in tin con wavelike , d unts. e rb mo tum o s b fic a uan a s i eci d q y g er all sp calle n E m ts s e k in ac p in stepwise particle-like

Matter can gain or lose energy only in small, specific amounts called quanta (quantum). That is, a quantum is the minimum amount of energy that can be gained or lost by an atom. Radiant energy is quantized.

A Photon Electromagnetic radiation has both wavelike and particlelike characteristics. Electromagnetic radiation can be thought of as a stream of tiny particles, or bundles of energy, called photons. photon A photon is quantum of radiant energy.

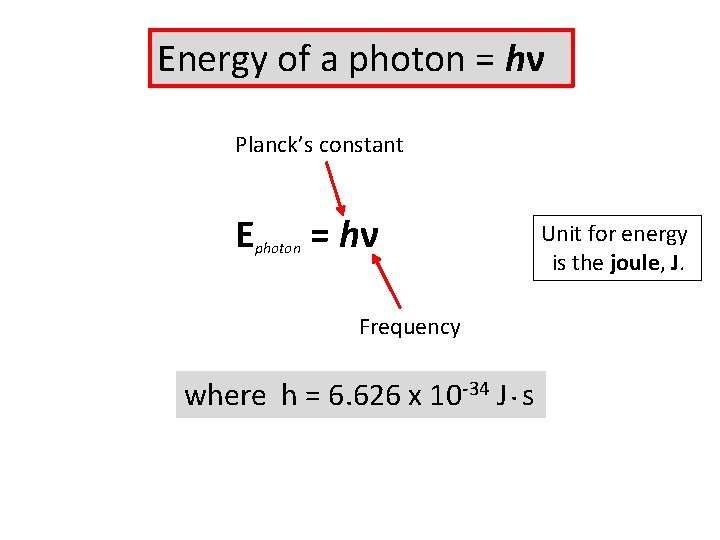
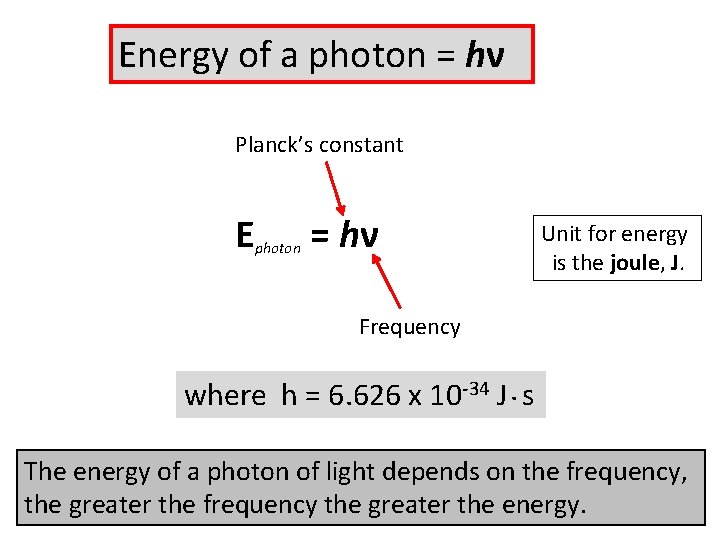
Energy of a photon = hν Planck’s constant E photon = hν Frequency where h = 6. 626 x 10 -34 J. s Unit for energy is the joule, J.

What is the energy of one photon of red light? The wavelength of red light is 690 nm. c ν= = λ E E 4. 35 x 1014 /s photon = hν photon = 2. 88 x 10 -19 J

Energy of a photon = hν Planck’s constant E photon = hν Unit for energy is the joule, J. Frequency where h = 6. 626 x 10 -34 J. s The energy of a photon of light depends on the frequency, the greater the frequency the greater the energy.

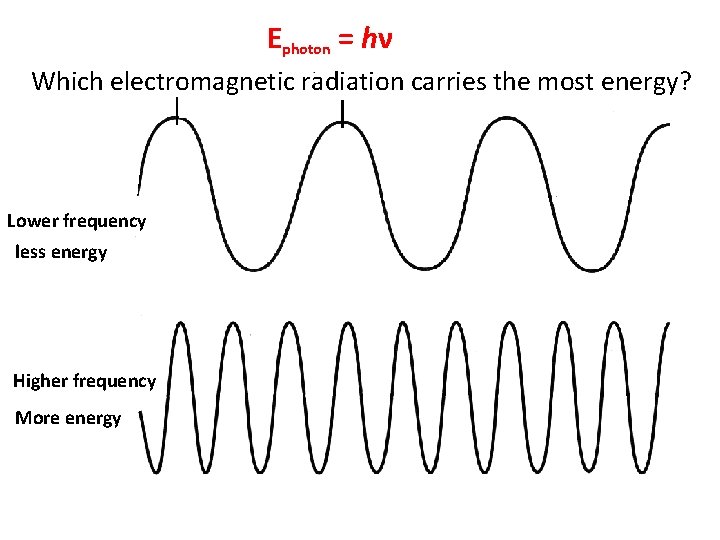
Ephoton = hν Which electromagnetic radiation carries the most energy? Lower frequency less energy Higher frequency More energy

As the frequency increases The energy increases

Gamma rays have the highest frequency of all radiation, as a result gamma rays have the greatest energy.

Tiny water droplets in the air disperse the white light of the sun into a rainbow. What is the energy of a photon from the violet portion of the rainbow if it has a frequency of 7. 23 x 1014 s-1? Given: ν = 7. 23 x 1014 /s h = 6. 626 x 10 -34 J. s E=? Equation: E = hν

Microwave ovens emit microwave energy with a wavelength of 12. 9 cm. What is the energy of exactly one photon of this microwave radiation? Given: Equation:

What is the frequency of UV light that has an energy of 2. 39 x 10 -18 J? . Given: Equation:

WHITEBOARD PRACTICE

4. What is the energy of radiation that has a frequency of 4. 2 X 1017 /s? Given: Equation:

5. Calculate the energy of one photon of orange light that has a wavelength of 605 nm. Given: Equation:

6. Calculate the energy and frequency of red light having a wavelength of 6. 80 x 10 -5 cm. Given: Equation:

7. A ruby laser produces red light that has a wavelength of 715 nm. Calculate its energy. Given: Equation:

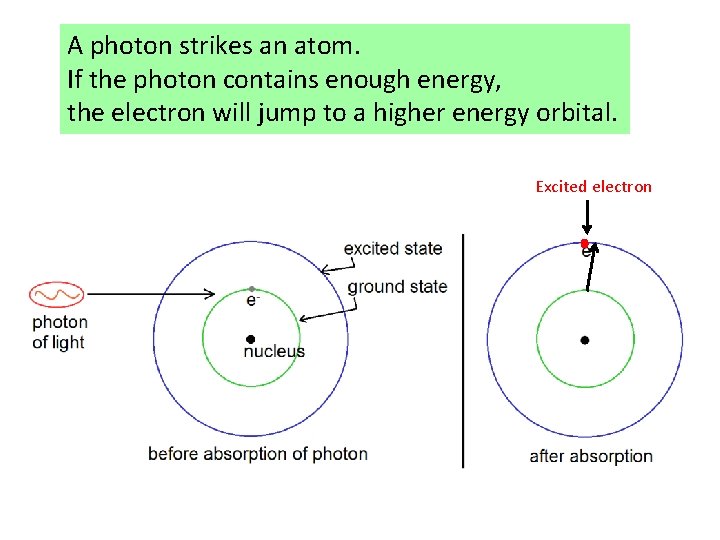
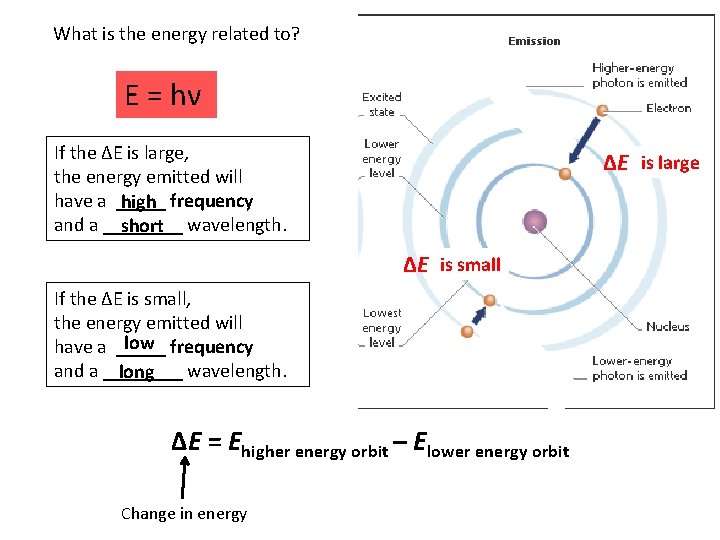
A photon strikes an atom. If the photon contains enough energy, the electron will jump to a higher energy orbital. Excited electron

If the photon doesn’t contain enough energy, the electron will remain in the ground state.


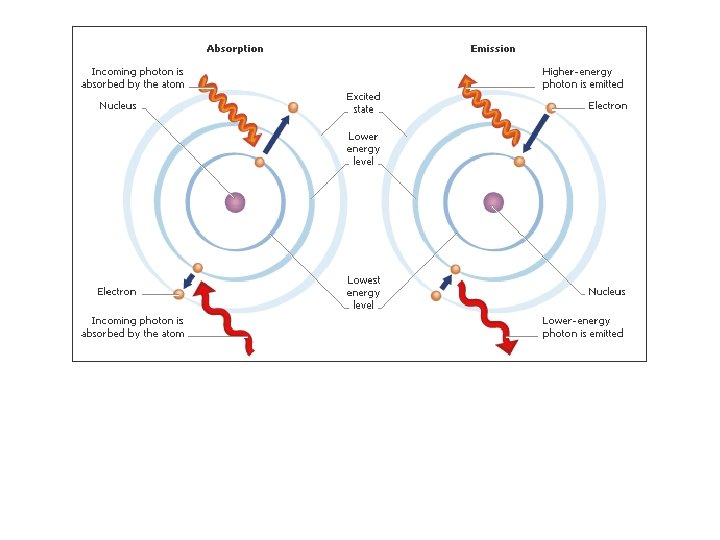
What is the energy related to? E = hν If the ∆E is large, the energy emitted will have a _____ high frequency and a ____ short wavelength. ∆E =is? large ∆E =is? small If the ∆E is small, the energy emitted will low frequency have a _____ and a ____ wavelength. long ∆E = Ehigher energy orbit – Elower energy orbit Change in energy

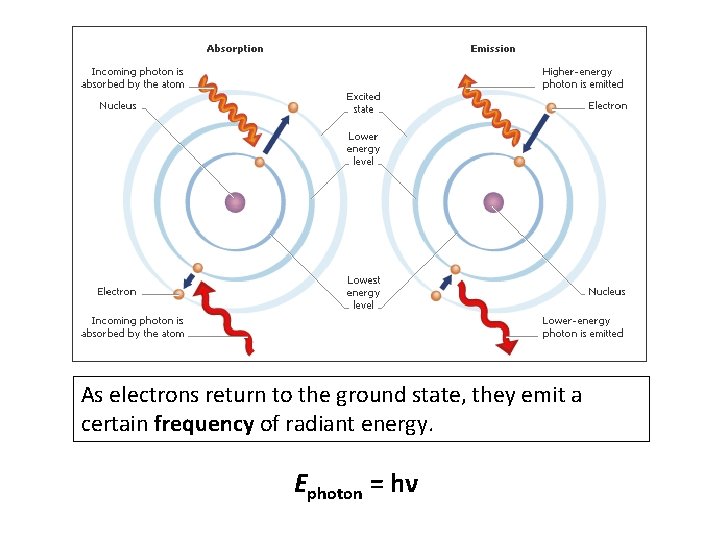
As electrons return to the ground state, they emit a certain frequency of radiant energy. Ephoton = hν


LAB: FLAME TEST

Each compound tested will produce a different color flame. Flame colors are produced from the movement of the electrons in the metal atoms present in these compounds. For example, a sodium atom in its ground state has the electronic configuration 1 s 22 p 6. When you heat the sodium atoms, the electrons gain energy and jump into any of the empty orbitals at higher levels - for example, into the 7 s or 6 p or 4 d.

Because the electrons are now at a higher and more energetically unstable level, they tend to fall back down to the ground state. As they return to the ground state, they emit photons of a specific energy. This energy corresponds to a particular wavelength of light, and so produces particular colors of light. Each metal has a unique electron configuration. The exact sizes of the possible jumps in energy terms vary from one metal to another. That means that each different metal will produce a different flame color.

Lower energy higher energy