Light and Quantized Energy Chapter 5 1 Objectives

Light and Quantized Energy Chapter 5. 1 Objectives: • Compare the wave and particle models of light • Define a quantum of energy and explain how it is related to an energy change of matter. • Contrast continuous electromagnetic spectra and atomic emission spectra.

Electromagnetic Radiation • Electromagnetic Radiation has wavelike characteristics as it travels through space. • Light rays act like waves as they travel. – Visible light – Microwaves – X-rays
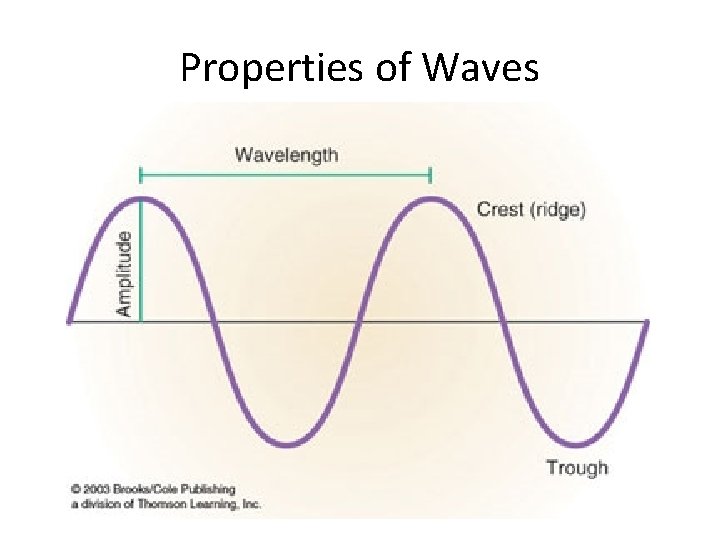
Properties of Waves
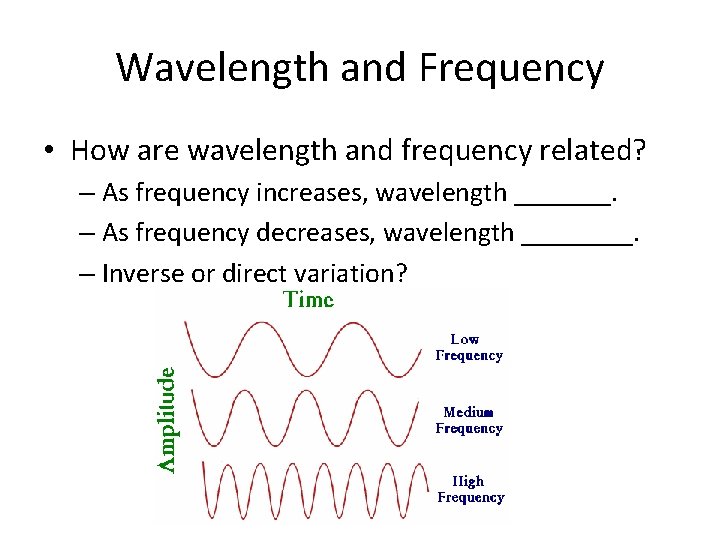
Wavelength and Frequency • How are wavelength and frequency related? – As frequency increases, wavelength _______. – As frequency decreases, wavelength ____. – Inverse or direct variation?
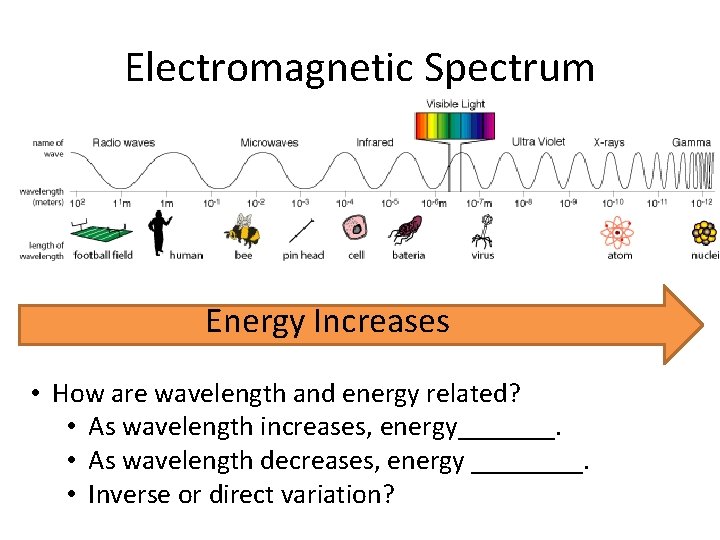
Electromagnetic Spectrum Energy Increases • How are wavelength and energy related? • As wavelength increases, energy_______. • As wavelength decreases, energy ____. • Inverse or direct variation?
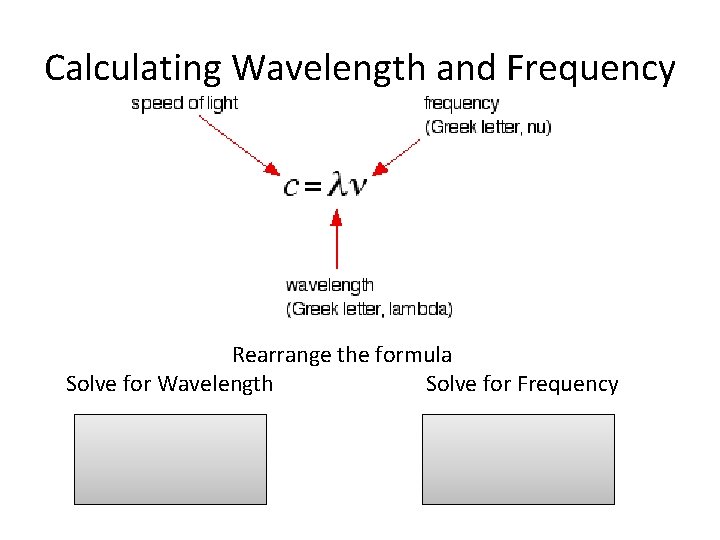
Calculating Wavelength and Frequency Rearrange the formula Solve for Wavelength Solve for Frequency

Practice • P. 121 2 -4

Quantum Concept • A quantum is the minimum amount of energy that can be gained or lost by an atom. • Think of a ladder. – Could you stand between rungs? – Could you climb more than one rung at a time?

Planck’s Constant • The energy of a quantum is related to the frequency of the emitted radiation. • Planck’s constant: h=6. 626 x 10 -34 J●s – Energy is measured in units called Joules

Photoelectric Effect • The photoelectric effect occurs when light shines on a metal surface and causes electrons to be emitted from the surface. – In this case, the electrons are called photoelectrons.

Photons • Light is sometimes depicted as a wave, but you can also think of it as a stream of tiny particles or bundles of energy, called photons. – A photon is a particle of electromagnetic radiation that • Has no mass • Carries a quantum of energy Ephoton = hν

Equations Ephoton = hν • What is h? • What is ν? Rearrange the formula. Solve for Frequency

Practice • P. 124 5 a-b

Atomic Emissions Spectra • Atomic Emissions Spectrum – the set of frequencies of the electromagnetic waves emitted by atoms of an element – Think of a prism • Each element emits different wavelengths – This is why elements appear different colors as they burn.

Quantum Theory of the Atom Section 5. 2 Objectives: • Compare the Bohr and quantum mechanical models • Explain the impact of Broglie’s wave-particle duality • Explain Heisenberg’s uncertainty principle • Identify the relationships among a H atom’s energy levels, sublevels, and atomic orbitals.

Bohr Model • Because the hydrogen atom’s emission spectrum is discontinuous, Bohr developed a quantum theory that each frequency emitted corresponds to the change in energy levels between orbitals. ΔE = E higher level orbit – E lower lever orbit = E photon = hν
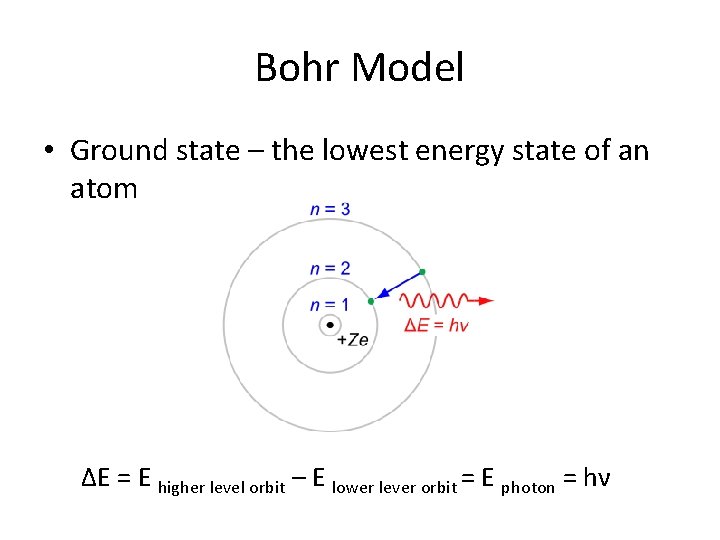
Bohr Model • Ground state – the lowest energy state of an atom ΔE = E higher level orbit – E lower lever orbit = E photon = hν

Quantum Mechanical Model • De Broglie thought Bohr’s model of quantized electron orbits was similar to the characteristics of waves. • The de Broglie equation predicts that all moving particles have characteristics of waves. λ= _h_ mv λ = wavelength h = Planck’s constant m = mass of particle v = velocity
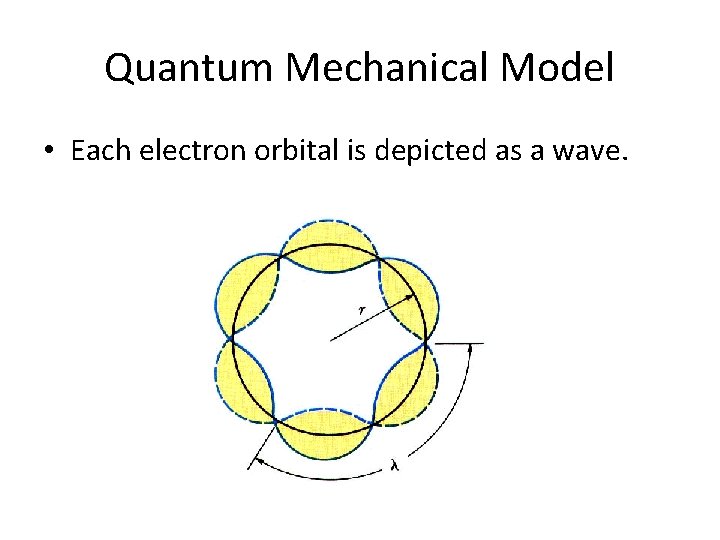
Quantum Mechanical Model • Each electron orbital is depicted as a wave.

Heisenberg Uncertainty Principal • It is virtually impossible to know the velocity and location of a particle at the same time. – We can’t know exactly where an electron is and how fast it is orbiting the nucleus of an atom. – That’s why we think of the electron clouds. – The atomic orbital describes the electron’s probable location. • Density diagram

Hydrogen’s Atomic Orbitals (Quantum Mechanical Model) • Principal Quantum Numbers (n) indicate the relative sizes and energies of atomic orbitals. • ‘n’ specifies the atoms major energy levels, called principal energy levels. – Lowest principal energy level, n=1 – Maximum n=7 for hydrogen

Continued • Each principal energy level has energy sublevels. – n = 1 has 1 sublevel – n = 2 has 2 sublevels • Sublevels are named s, p, d, and f based on their shapes.
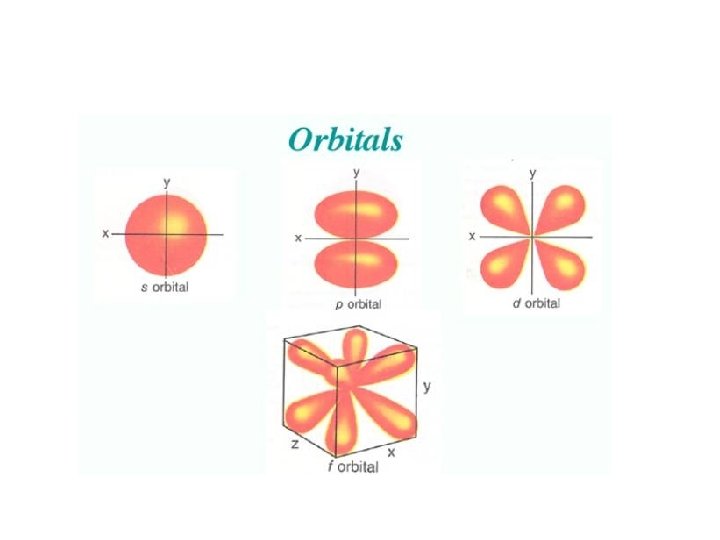

Electron Configurations Section 5. 3 • • Objectives: Apply the Pauli exclusion principal, the Aufbau principal, and Hund’s rule to write electron configurations Define valence electrons and draw electron-dot structures

Electron Configurations • The electron configuration shows the arrangement of electrons in the ground state. – The ground state is the lowest energy state, and remember low energy states are more stable than high energy states. – Aufbau Principal – Electrons will fill the lowest energy orbitals first.
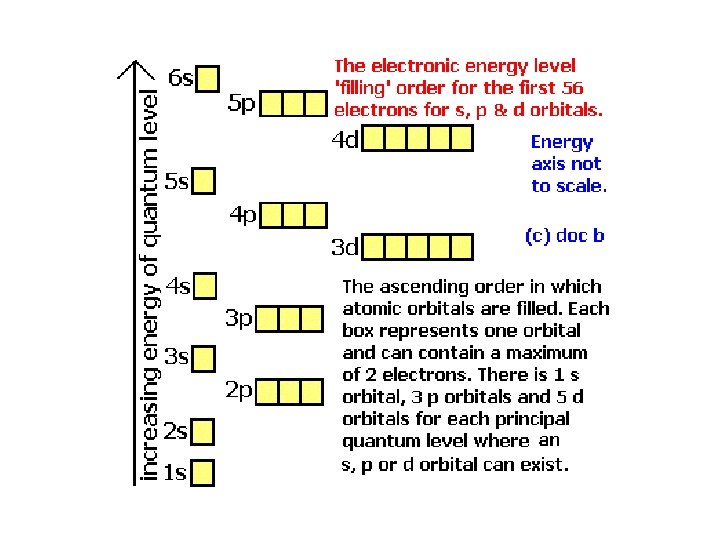
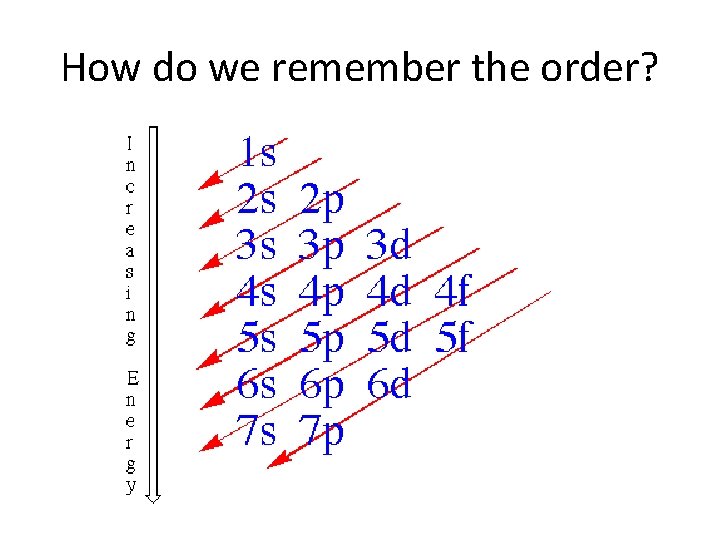
How do we remember the order?

How many electrons does each type of orbital hold? Sublevel s Maximum number of electrons 2 p 6 d 10 f 14
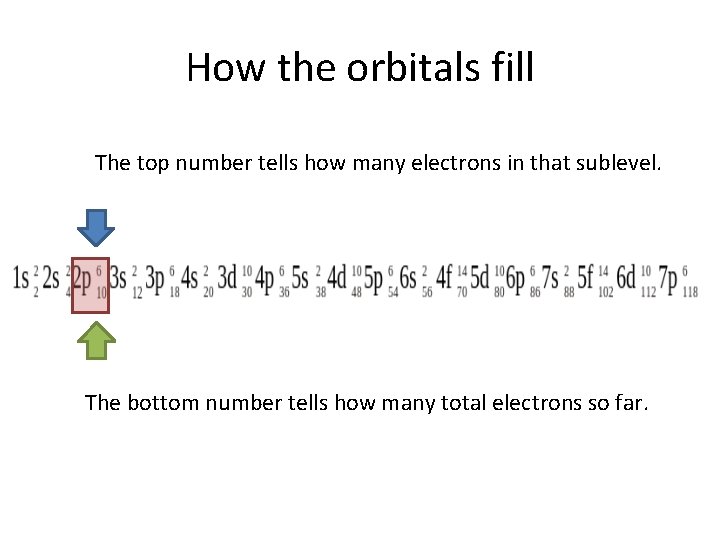
How the orbitals fill The top number tells how many electrons in that sublevel. The bottom number tells how many total electrons so far.
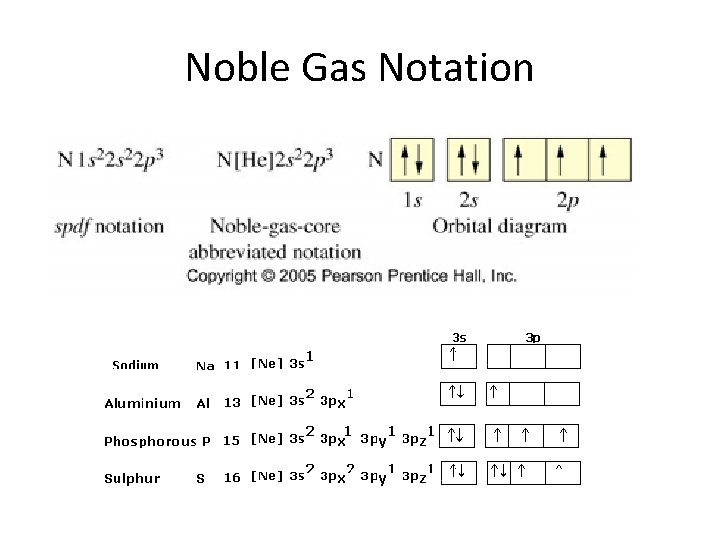
Noble Gas Notation

Pauli Exclusion Principal • How many electrons are in each box? – Each box represents an orbital. • What do you notice about the arrows? • Pauli Exclusion Principal – A maximum of 2 electrons will fill an orbital (box) if they have opposite spins.

Hund’s Rule • How are the boxes filled before each box is full? • Hund’s Rule – singe electrons with the same spin must occupy equal-energy obitals before electrons with opposite spins can occupy the same orbital. – Put up spins in each orbital for a given energy level, then pair them with down spins.

Examples

Practice • P. 139 18 a-b, 19 -21

Valence Electrons • Valence electrons – electrons in the atom’s outermost orbitals – Outer orbitals have the highest principle energy levels. – Valence electons determine an atom’s chemical properties (how it reacts). – Look at the additional electrons after the noble gas in noble gas notation. – Look at the periodic table!!!

Electron-Dot Structure • Electron-dot structure is the shorthand way to show the valence electrons. – We don’t draw the electrons in the inner shells because they don’t affect how the atom will react with other atoms.

- Slides: 37