Section 5 1 Light and Quantized Energy Objectives

Section 5. 1 Light and Quantized Energy

Objectives • Compare the wave and particle models of light • Define a quantum of energy and explain how it is related to an energy change of matter • Contrast continuous electromagnetic spectra and atomic emission spectra

Light and Quantized Energy • Wave Nature of Light – Electromagnetic radiation (ER) form of energy that exhibits wavelike behavior as it travels through space. – Exs: visible light, microwaves, x-rays

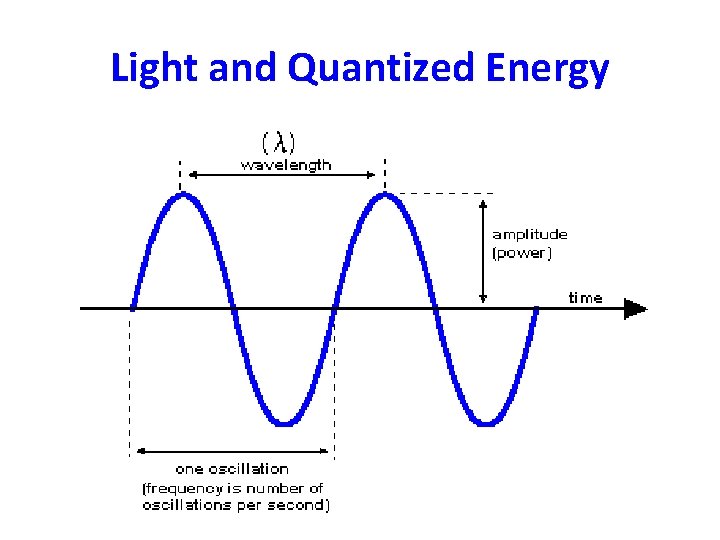
Characteristics of waves – Wavelength, : shortest distance between equivalent points on a continuous wave. • Usually in nm – Frequency, : number of waves that pass a given point per second. • Hz (Hertz)

Continued – Amplitude: wave’s height from the origin to a crest, or from the origin to a trough. – Speed of light = c = 3. 0 x 108 m/s
Light and Quantized Energy

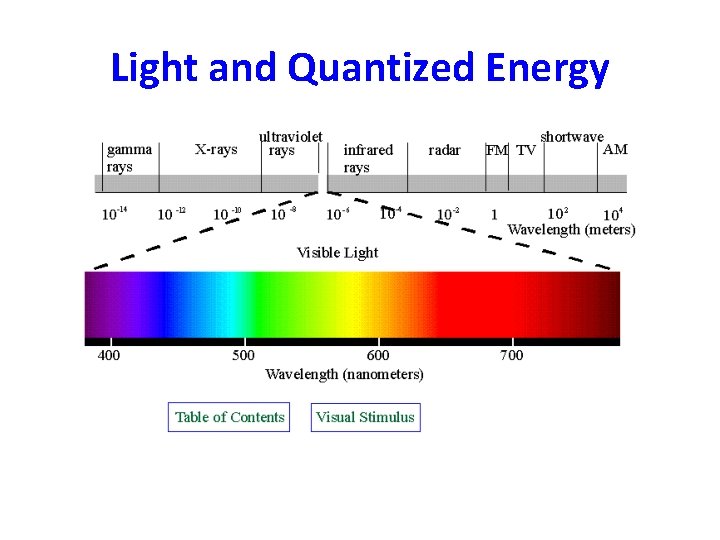
Electromagnetic Spectrum – Continuous spectrum of different types of ER. – No portion that does not correspond to a unique wavelength and frequency.
Light and Quantized Energy

Light and Quantized Energy • Calculating wavelength/frequency – Example P. 121

Particle Nature of Light • Besides having wave characteristics, light also acts like a particle. • The Quantum Concept: Energy can only be gained or lost in a specified amount Quantum • Equantum = hν • h is Plank’s Constant = 6. 626 x 10 -34 J • s

Blocks and Quanta • Think of energy transfer like building a wall of Legos®. • Only add/take away 1 Lego. • No ½ Lego.

Photoelectric Effect • Electrons, called photoelectrons, emit from a metal’s surface when light of a certain frequency shines on the surface. Photoelectrons Light

The Photon • Photon: – particle of electromagnetic radiation – no mass – carries a quantum of energy • Ephoton = hv

Neon Signs

Atomic Emission Spectra • Electricity passed through a tube of gas • Gas atoms absorb energy and become excited • Atoms release the energy absorbed as light.

Atomic Emission Spectra Hydrogen (H) Helium (He) Oxygen (O) http: //astro. u-strasbg. fr/~koppen/discharge/index. html

Atomic Emission Spectra Neon (Ne) Xenon (Xe) http: //astro. u-strasbg. fr/~koppen/discharge/index. html

Example Problem 5 -2 p. 124

Homework • Wave and Energy Worksheet
- Slides: 19