Kinetics Reaction Rates Rate Law Collision Theory and

















![The Initial Rate Method – Data Collection • Experiment [NH 4 NCO] M Rate The Initial Rate Method – Data Collection • Experiment [NH 4 NCO] M Rate](https://slidetodoc.com/presentation_image_h2/8ee9592bd78d78e1ab2930364c0864fc/image-18.jpg)





















- Slides: 41

Kinetics Reaction Rates, Rate Law, Collision Theory and Activation Energy (PLN 710)

PLN 7 • Important Concepts: – Reactions can occur at different rates – Factors that help determine the reaction rate – Reaction characteristics: • Mechanism of reaction (PLN 11) • Rate of Reaction • Rate Law (PLN 8)

Basic Kinetics • Reaction Rate – Speed that reactants disappear and products form – How fast reactants become/form products

Examples: •

What Determines the Rate? • • Temperature Pressure Concentration Catalyst (PLN 12) – Lowers activation energy • Surface Area – Not going to be covered on this test

Mechanism of Reaction • Lists the individual steps of a reaction • Describe reactions at a molecular level • Not all reactions occur in one step or all at once • Chemical equation is overall summary of the reaction

Rate of Reaction • The calculated rate at which reactants are used up/disappear or products are formed/appear • For general reaction: Where a, b, c and d are coefficients,

Rate Law •

Reaction Rates • Definition: – The rate of a reaction is the change in molar concentration of a reactant or product per unit of time in a reaction • Example: • Rate of decomposition of • However, this gives the average rate over the period of time Δt • The instantaneous rate can be calculated as the slope of the tangent line at a given point

Overall Rate of Reaction • The rate of reaction is more commonly described in terms of the equation • For the reaction: • For every 2 moles of N 2 O 5 lost: – 4 moles of NO 2 is formed – And 1 mole of O 2 is formed Note: The negative sign placed in front of the reactants is to count for the fact that their concentrations are decreasing

PLN 8 • Important Concepts: – Rate Laws – Rate Constant (k) – Order of Reaction – Initial Rate Method

Rate Laws for Chemical Reactions • Rates depend on concentrations of certain reactants and the concentration of the catalyst, if there is one • Definition: – A Rate Law is an equation that relates the rate of a reaction to the concentrations of the reactants (and catalyst, if used) raised to various powers, or exponents.

• Rate – Expressed in mol/L/time or M/time • k – Rate constant – Specific to a certain reaction at a specific temperature – Units depend on the overall reaction order (explained later) • [A] & [B] – Concentrations of reactants as mol/L or M • m & n – Orders of reaction with respect to reactants

k • The reaction constant, k, is called the rate constant and is dependent on the particular reaction as well as the specific temperature at which the reaction takes place • The units of k depend on the order of reaction

Orders of Reaction •

Determining the Rate Law Experimentally • The Initial Rate Method – Uses the relationship between the measured initial rate of a reaction and the concentrations of each reactant • The Integrated Rate Law Method – Uses the relationship between reactant or product concentration and its changes over time

The Initial Rate Method •
![The Initial Rate Method Data Collection Experiment NH 4 NCO M Rate The Initial Rate Method – Data Collection • Experiment [NH 4 NCO] M Rate](https://slidetodoc.com/presentation_image_h2/8ee9592bd78d78e1ab2930364c0864fc/image-18.jpg)
The Initial Rate Method – Data Collection • Experiment [NH 4 NCO] M Rate of loss of NH 4 NCO M/min 1 0. 14 2. 2 × 10 -4 2 0. 28 8. 8 × 10 -4

The Initial Rate Method – Calculations •

Initial Rate Method – Rate Law and k •

PLN 9 • Important Concepts: – Integrated Rate Law Method • 0 th, 1 st and 2 nd order reactions – Half-Life • 0 th, 1 st and 2 nd order reactions – Units for k • 0 th, 1 st and 2 nd order reactions

The Integrated Rate Law Method • Initial Rate Method describes change of rate as we change initial reactant concentrations • Using integral calculus, we can convert Rate Laws into equations that can give us concentrations of the reactant(s) or product(s) at anytime during the reaction • The Integrated Rate Law Method fits experimental data to a mathematical relationship

First Order Reactions •

First Order Reactions (cont. ) •

Explaining the Integration: 1 st Order •

Explaining the Integration: 1 st Order (cont. ) •

Half-Life of a Reaction •

Rate Law, k, Integrated Rate Law and Half-Life for 1 st, 2 nd and 3 rd Order • For only 1 st order reactions: The half-life doesn’t depend on the initial concentration Order Rate Law 0 Rate = k 1 Rate = k [B] 2 k Integrated Rate Law Half-Life n (number of half lives)

PLN 10 • Important Concepts: – Collision Theory • Pre-exponential constant (A) • f. KE – Importance of Correct Orientation – Arrhenius Equation – Activation Energy (EA) – Transition State Theory – Potential Energy Diagrams

What Affects Reaction Rates, Again? • Reaction rates are dependent upon: – Temperature – Pressure – Concentration – Catalyst – Surface Area

How Temperature influences Reaction Rates •

Collision Theory •

Changes in Temperature •

Importance of Correct Orientation •

Arrhenius Equation •

Calculating EA for an Equation •

Example • k (L mol-1 s-1) T (°C) 1. 1 550 6. 4 625

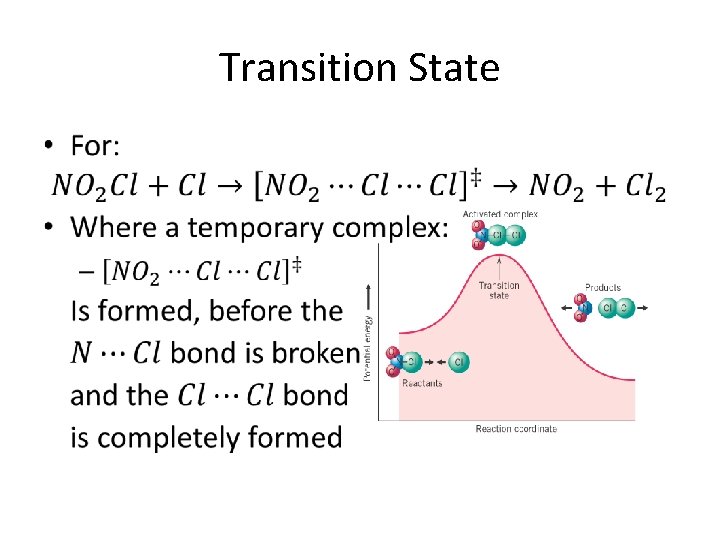
Transition State Theory • Transition State Theory describes what happens to the reactant molecules as a reaction proceeds • When the reactants collide, they form a temporary “substance” composed of a combination of the two reactants – This temporary “substance” is called the transition state or activated complex

Transition State •

Transition States (cont. ) •

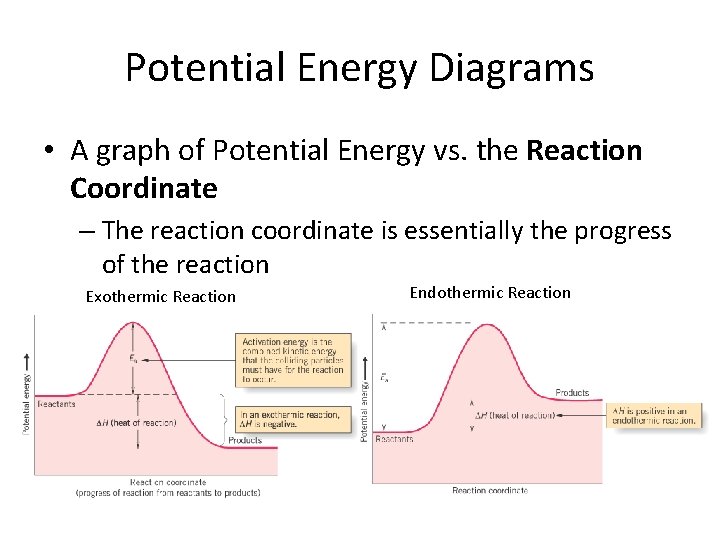
Potential Energy Diagrams • A graph of Potential Energy vs. the Reaction Coordinate – The reaction coordinate is essentially the progress of the reaction Exothermic Reaction Endothermic Reaction