Chemistry Chapter 19 Reaction Rates and Equilibrium Study

Chemistry Chapter 19 – Reaction Rates and Equilibrium Study Guide - Answers

Matching. 1. 2. 3. 4. 5. I activated complex A reaction rate J Le Châtelier’s principle C spontaneous G elementary reaction E chemical equilibrium 7. H entropy 8. D activation energy 9. F inhibitor 10. B free energy 6.
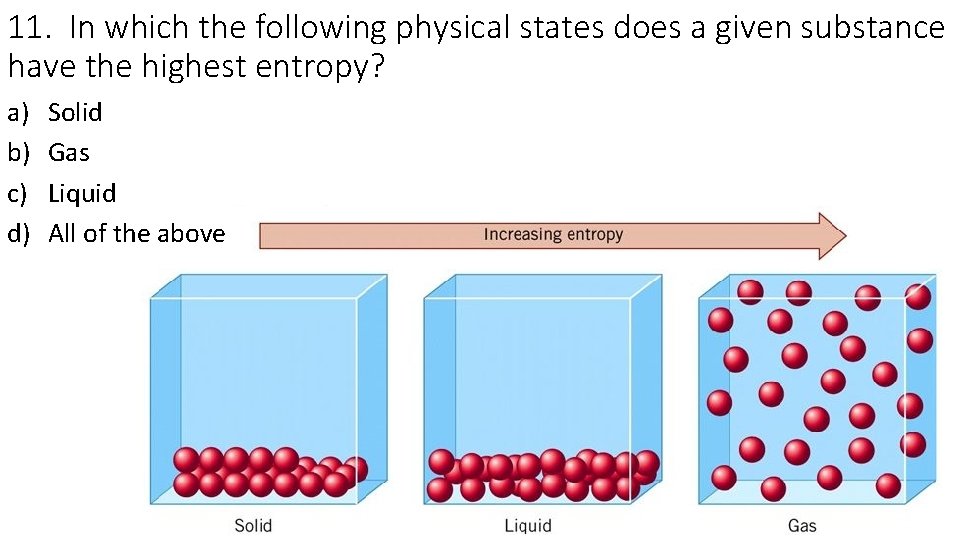
11. In which the following physical states does a given substance have the highest entropy? a) b) c) d) Solid Gas Liquid All of the above
12. A reaction that requires free energy a) b) c) d) Must be endothermic Is nonspontaneous. Must correspond to a decrease in entropy. Is spontaneous.

13. The two factors that determine whether a reaction is spontaneous or nonspontaneous are: a) b) c) d) Entropy and disorder. Entropy and enthalpy change. Electron configuration and energy change. Energy and the heat of reaction.
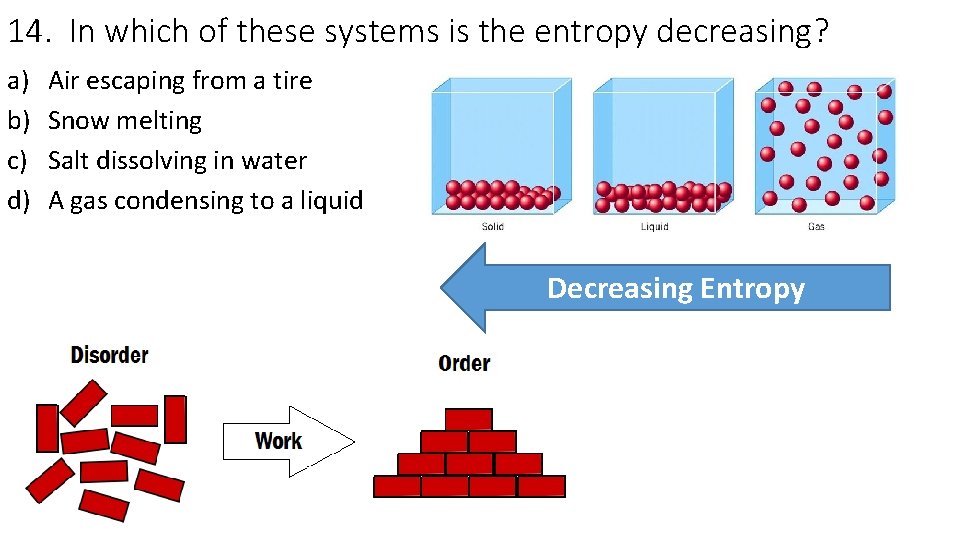
14. In which of these systems is the entropy decreasing? a) b) c) d) Air escaping from a tire Snow melting Salt dissolving in water A gas condensing to a liquid Decreasing Entropy
15. All spontaneous processes: a) b) c) d) Are exothermic Are endothermic Involve an increase in entropy Release free energy
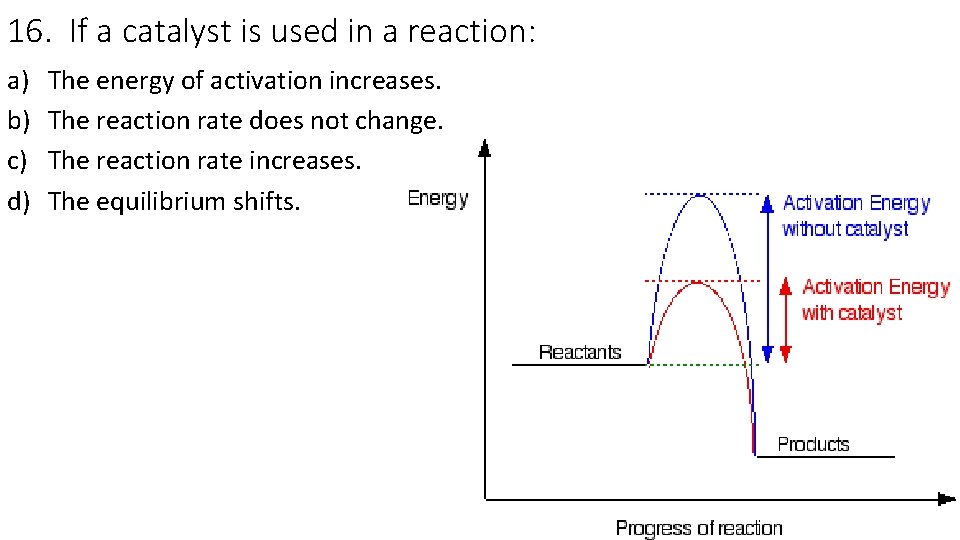
16. If a catalyst is used in a reaction: a) b) c) d) The energy of activation increases. The reaction rate does not change. The reaction rate increases. The equilibrium shifts.

17. Which of the following affects the rate of a chemical reaction? a) b) c) d) The presences of a catalyst The temperature The concentration of reactants All of the above.

18. What is the expression for Keq for this reaction? 2 H 2 O(g) ⇋ 2 H 2(g) + O 2(g) a) Keq = [2 H 2 O] [H 2]x[O 2] b) Keq = [H 2]2 x [O 2] [H 2 O]2 c) Keq = [2 H 2] x [O 2] [2 H 2 O] d) Keq = [H 2 O]2 [H 2]2 x [O 2]

19. In an equilibrium reaction with a Keq of 1 x 108 a) b) c) d) Reactants are favored. Reaction is nonspontaneous. Products are favored. Reaction is exothermic.
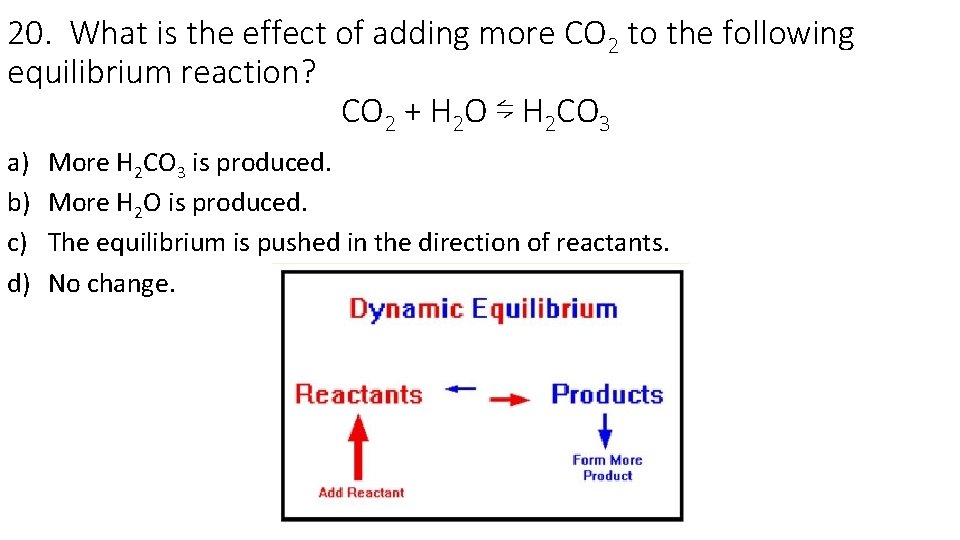
20. What is the effect of adding more CO 2 to the following equilibrium reaction? CO 2 + H 2 O ⇋ H 2 CO 3 a) b) c) d) More H 2 CO 3 is produced. More H 2 O is produced. The equilibrium is pushed in the direction of reactants. No change.
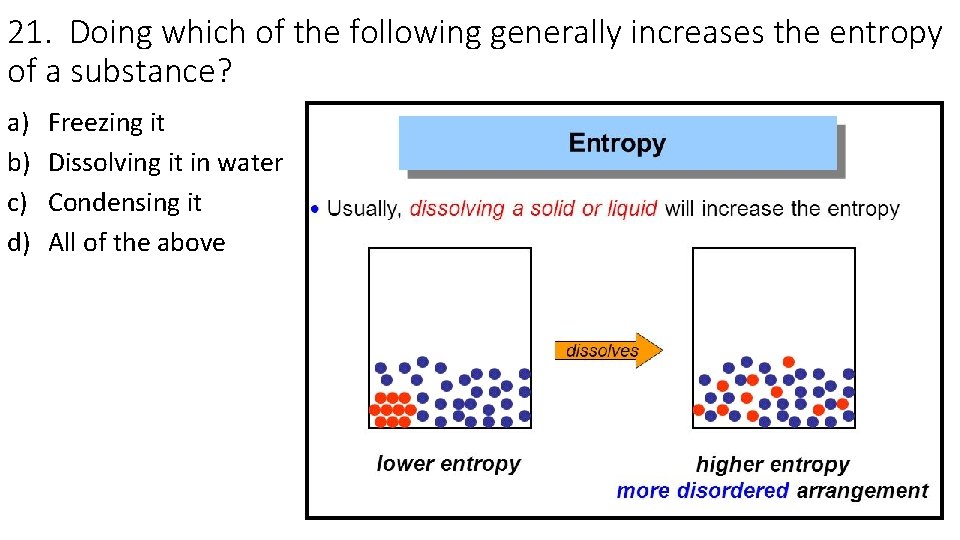
21. Doing which of the following generally increases the entropy of a substance? a) b) c) d) Freezing it Dissolving it in water Condensing it All of the above

22. The Keq of a reaction is 4 x 10 7. At equilibrium: a) b) c) d) The reactants are favored. The products are favored. The reactants and products are present in equal amounts. The rate of the reverse reaction is much greater than the rate of the forward reaction.
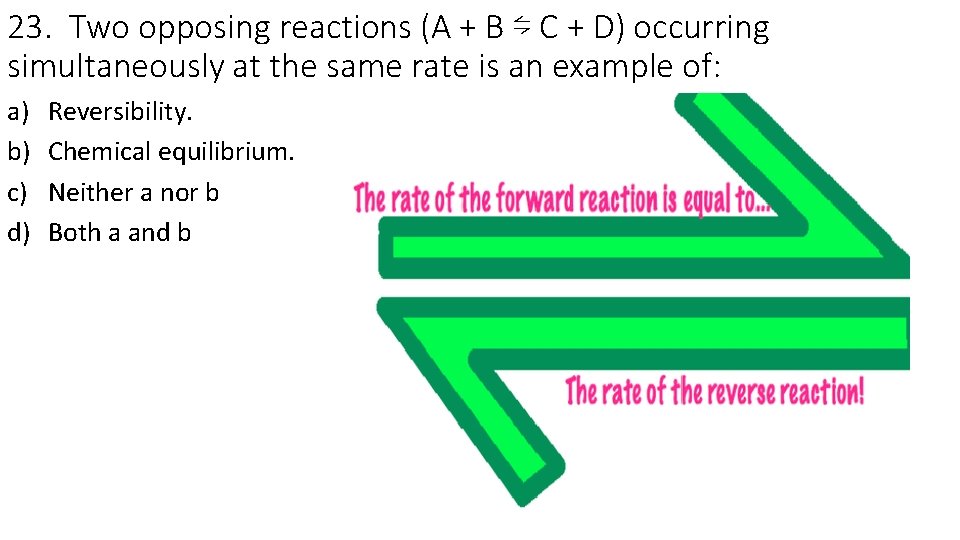
23. Two opposing reactions (A + B ⇋ C + D) occurring simultaneously at the same rate is an example of: a) b) c) d) Reversibility. Chemical equilibrium. Neither a nor b Both a and b

24. Calculate the value of Keq for this reaction at equilibrium. 2 NOCl(g) ⇋ 2 NO(g) + Cl 2(g) An analysis of the equilibrium mixture in a 1 L flask gives the following results: NOCl, 0. 30 mol; 1. 2 mol; Cl 2, 0. 60 mol. Keq = [NO]2 x [Cl 2] = (1. 2)2(0. 60) = 9. 6 [NOCl]2 (0. 30)2 Keq = 9. 6
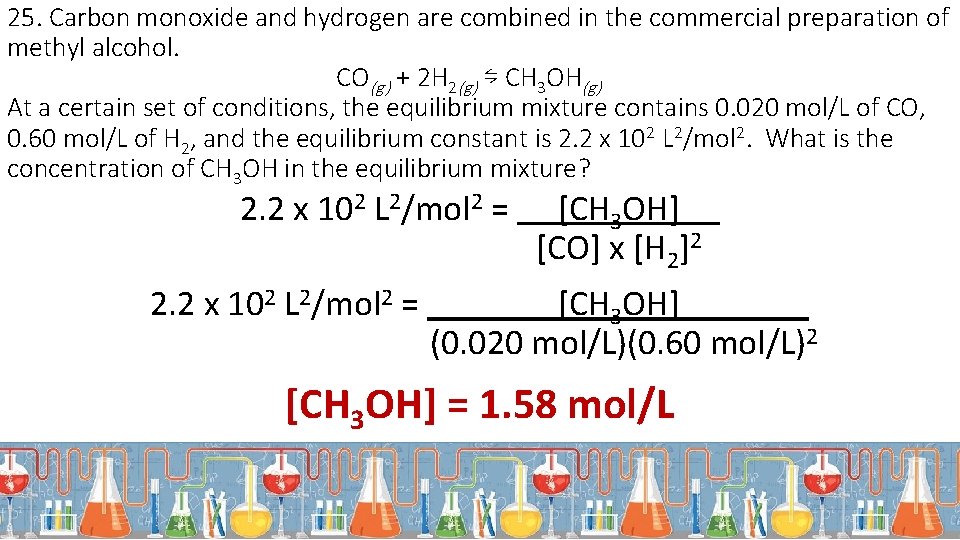
25. Carbon monoxide and hydrogen are combined in the commercial preparation of methyl alcohol. CO(g) + 2 H 2(g) ⇋ CH 3 OH(g) At a certain set of conditions, the equilibrium mixture contains 0. 020 mol/L of CO, 0. 60 mol/L of H 2, and the equilibrium constant is 2. 2 x 102 L 2/mol 2. What is the concentration of CH 3 OH in the equilibrium mixture? 2. 2 x 102 L 2/mol 2 = [CH 3 OH]. [CO] x [H 2]2 [CH 3 OH]. (0. 020 mol/L)(0. 60 mol/L)2 [CH 3 OH] = 1. 58 mol/L

26. The following reaction has an H 0 of 53 k. J/mol and a S 0 of 0. 070 k. J/(K x mol) at 25 °C. 2 A + B → 2 C Is this reaction spontaneous? (Calculate ΔG. ) 273 °C + 25° = 298 K ΔG 0 = ΔH 0 – TΔS 0 ΔG 0 = (53 k. J/mol) – (298 K)(0. 070 k. J/mol·K) ΔG 0 = (53 k. J/mol) – (21 k. J/mol) 0 ΔG = 32 k. J/mol This reaction is not spontaneous, because G 0 is positive!

27. What is the standard change in entropy for the following reaction when all reactants are in specified states? 2 H 2 O(l) + O 2(g) → 2 H 2 O 2(l) Standard entropies S 0 is J/(K x mol): H 2 O(l) = 69. 94, H 2 O 2(l) = 92. 0, O 2(g) = 205. 0 ΔS 0 = S 0(products) – S 0(reactants) ΔS 0 = (2 mol)(92. 0 J/mol·K) – [(2 mol)(69. 94 J/mol·K) + (1 mol)(205. 0 J/mol·K)] 1 mole ΔS 0 = -161 J/(mol·K)

28. Predict the changes in the equilibrium position for this reaction when the following changes are made: 2 A (g) + B(g) ⇋ 4 C(g) + heat A. Decrease the concentration of C; B. Add more heat; C. Increase the concentration of A; D. Increase the pressure. In each case, state whether the change causes a shift that favors the formation of reactants or of products. a) Favors Products b) Favors Reactants c) Favors Products d) Favors Reactants
![29. The rate law for the following reaction is: Rate = k[A]a x [B]b. 29. The rate law for the following reaction is: Rate = k[A]a x [B]b.](http://slidetodoc.com/presentation_image_h/2737ddb21c8c8d34a1472723a1f68495/image-21.jpg)
29. The rate law for the following reaction is: Rate = k[A]a x [B]b. a. A + b. B → c. C + d. D From the data in the following chart, find the kinetic order of the reaction with respect to A and B, as well as the overall order. Initial concentration of A (mol/L) Initial Concentration of B (mol/L) 0. 50 0. 10 0. 20 0. 01 0. 05 0. 10 0. 20 Initial Rate [mol/(L x s)] 2 x 10 -3 4 x 10 -3 8 x 10 -3 1 x 10 -3 8 x 10 -3 64 x 10 -3 Doubling A doubles the rate – first order in A. Doubling B increases the rate 8 times (23 = 8) – third order in B. First order + third order = fourth order overall.
- Slides: 21