Kinetics Reaction Rates Reaction Rates Collision theory Potential







- Slides: 14

Kinetics Reaction Rates

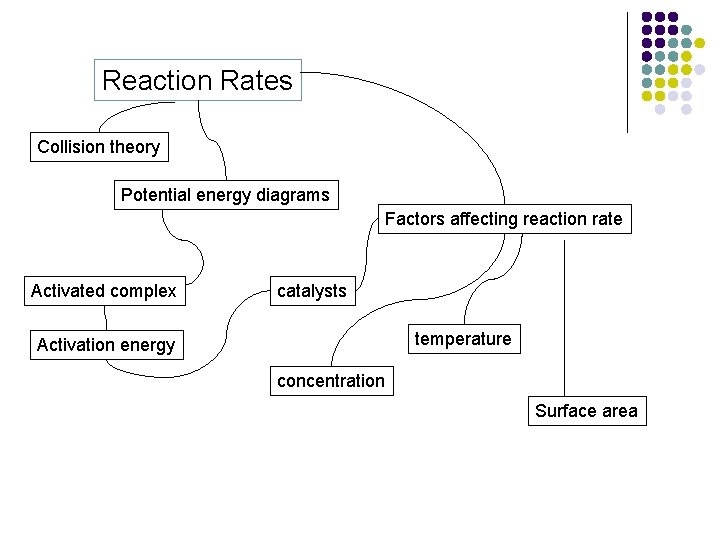
Reaction Rates Collision theory Potential energy diagrams Factors affecting reaction rate Activated complex catalysts temperature Activation energy concentration Surface area

Reaction Rates l l l A measure of how quickly a reaction occurs An experimental, measurable quantity Rate = change in property/change in time l l Example: Speed = miles/hour l Speed = Ddistance/Dtime Chemical kinetics: the study of reaction rates and the factors that affect them

What could we measure for a reaction? Easily measured properties include: l Change in mass of a solid l Change in concentration l Temperature changes l p. H changes l Gas volume changes l Color changes l We must also measure changes in TIME!

Writing Rate Expressions l l l For a general reaction a. A + b. B c. C + d. D General form: We need to modify the rate expression to compensate for stoichiometry l The reaction has only one rate for a given set of conditions l Convention: all reaction rates are positive

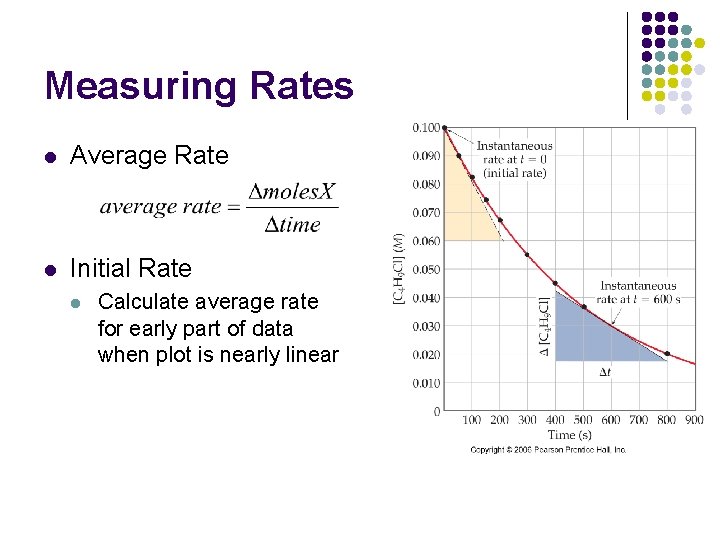
Measuring Rates l Average Rate l Initial Rate l Calculate average rate for early part of data when plot is nearly linear

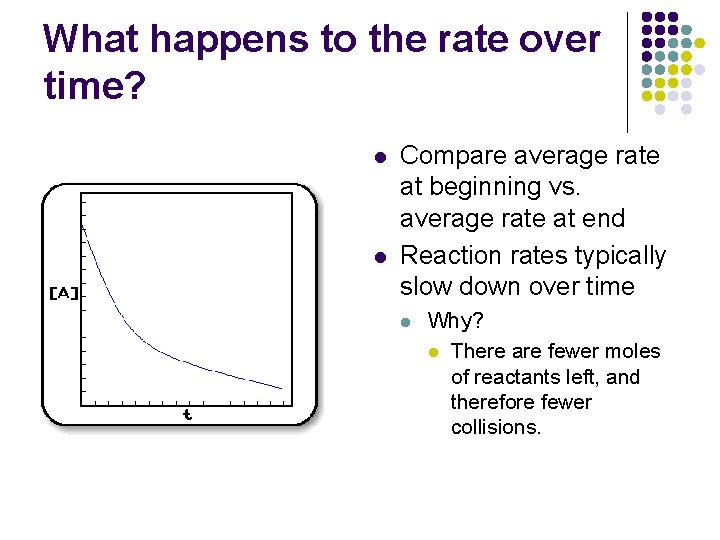
What happens to the rate over time? l l Compare average rate at beginning vs. average rate at end Reaction rates typically slow down over time l Why? l There are fewer moles of reactants left, and therefore fewer collisions.

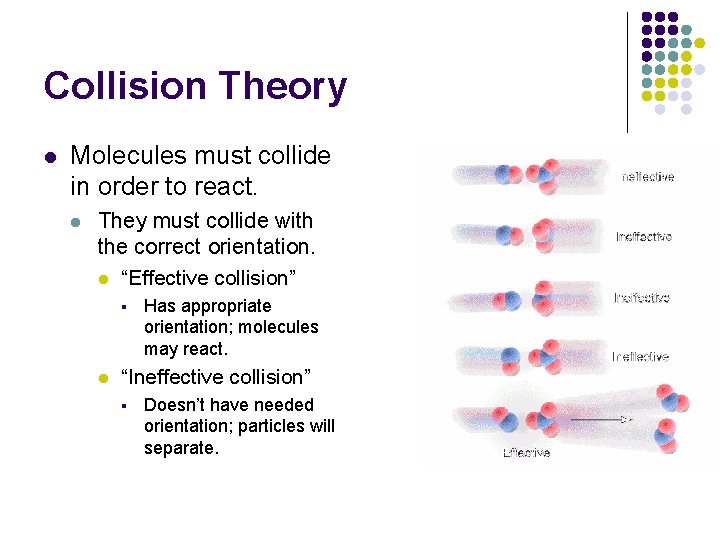
Collision Theory l Molecules must collide in order to react. l They must collide with the correct orientation. l “Effective collision” § l Has appropriate orientation; molecules may react. “Ineffective collision” § Doesn’t have needed orientation; particles will separate.

Collision Theory, cont. l Molecules must collide in order to react. l They must have enough energy to react. l Activation Energy, Ea § The minimum energy that reactants must have for the reaction to occur

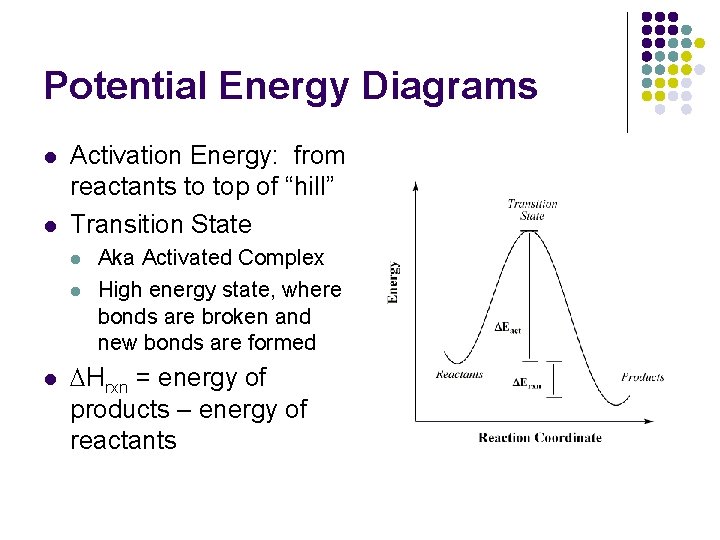
Potential Energy Diagrams l l Activation Energy: from reactants to top of “hill” Transition State l l l Aka Activated Complex High energy state, where bonds are broken and new bonds are formed DHrxn = energy of products – energy of reactants

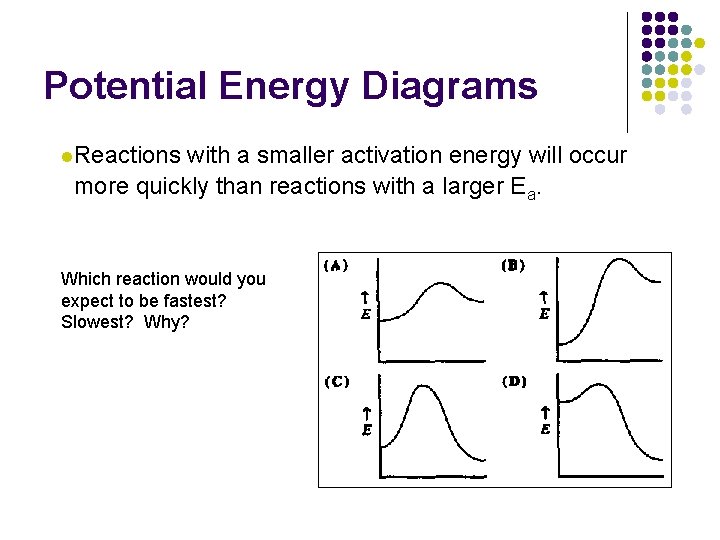
Potential Energy Diagrams l. Reactions with a smaller activation energy will occur more quickly than reactions with a larger Ea. Which reaction would you expect to be fastest? Slowest? Why?

Collision Theory l Basic premise: l l More collisions = faster reaction rate More collisions = greater likelihood for effective collisions

How can we speed up the rate of a reaction? l Increase temperature l l l Increase concentration l l Particles move more quickly, so more possible collisions More particles are likely to have enough energy to overcome activation energy barrier More particles, so more possible collisions Increase surface area l More particles are exposed, so more collisions are possible

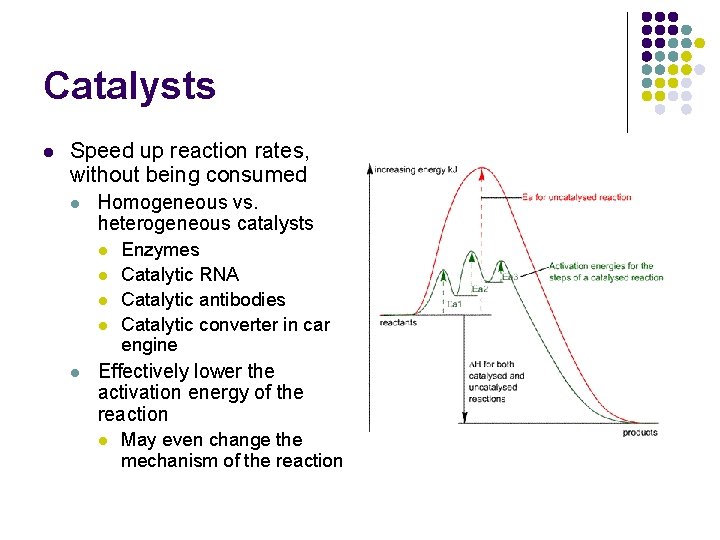
Catalysts l Speed up reaction rates, without being consumed l Homogeneous vs. heterogeneous catalysts l l l Enzymes Catalytic RNA Catalytic antibodies Catalytic converter in car engine Effectively lower the activation energy of the reaction l May even change the mechanism of the reaction