COLLISION THEORY OF REACTION RATES Class xii COLLISION













- Slides: 16

COLLISION THEORY OF REACTION RATES Class xii

COLLISION THEORY OF REACTION RATES �Reaction occur when � contact with each other or collide each other. �The theory / concepts regarding this is called collision theory.

According to collision theory : �the basic condition for a chemical reaction is the collision among the reacting species. �Collision frequency ( Z AB ) : the number of collisions per second per unit volume of the reaction mixture. �

�Collisions occur at a very fast rate and Z values are very high in many gaseous reactions. �For a binary collisions among gaseous reacting molecules under ordinary reaction condition of temperature (298 K) and pressure of 1 atm � Z values of order 1025 to 1028

�Z values 1025 to 1028 ……. Means reactions should be very fast or rather instantaneous. �But it is not so… �Many reactions under such conditions are very slow or do not occur at all.

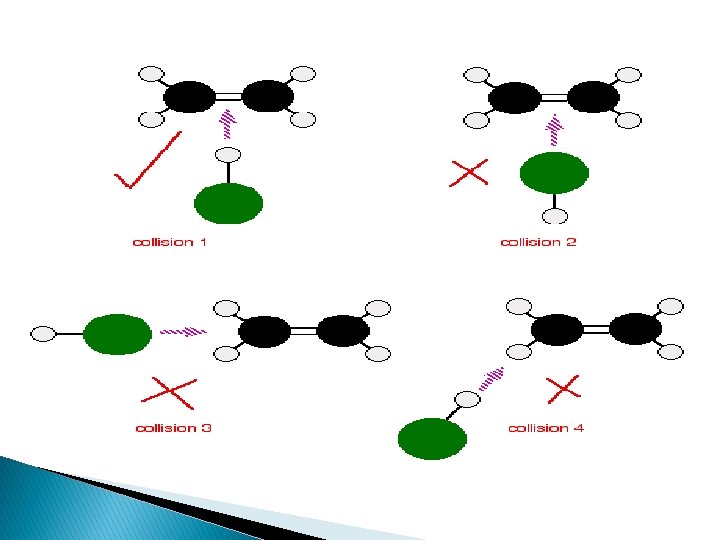
�This means it is not necessary that all collisions lead to the products. �Effective collisions : “The collisions among reacting species which results in the formation of products “

EFFECTIVE COLLISIONS AND THE BARRIERS � Energy barrier � Orientation Barrier

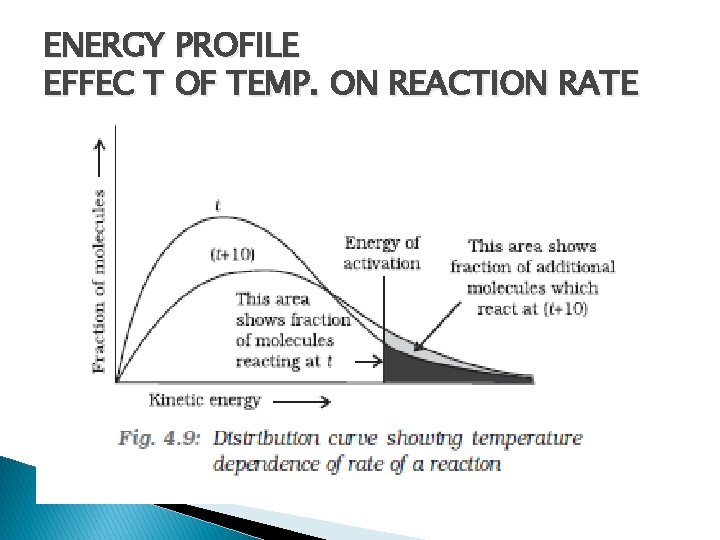
EFFECTIVE COLLISIONS-ENERGY BARRIER � Reacting species must have certain minimum energy called Threshold Energy E 0 � (Threshold Energy =Activation Energy + Energy possessed by reacting species) � � =

� �Is ZAB is collision Frequency the fraction of molecules having energy either equal or greater than activation energy � Arrhenius factor ( A) is related to collision Frequency (ZAB)

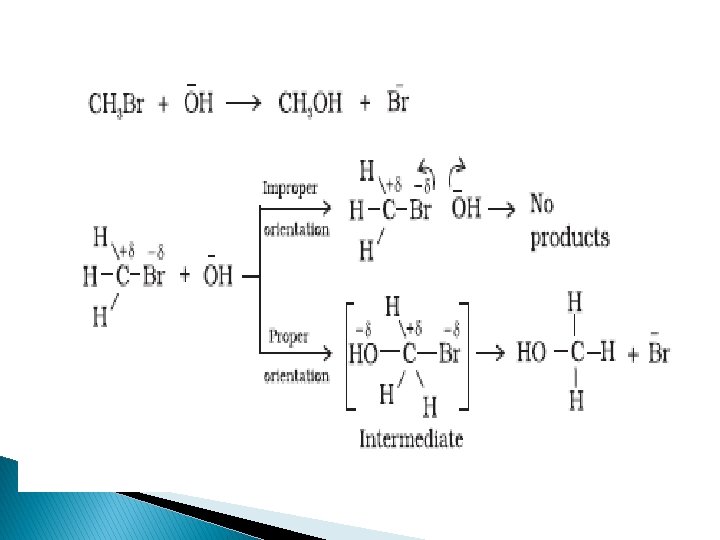
EFFECTIVE COLLISIONSORIENTATION BARRIER



�To account for effective collisions another factor P, The probability factor or steric factor is introduced.

ENERGY PROFILE EFFEC T OF TEMP. ON REACTION RATE

Conclusion : � �In collision theory Activation energy and Proper Orientation of the molecules together determine criteria for effective collisions and hence the rate of chemical reactions.

Assignments : Define Collision frequency ? 2. What do you mean by Effective collisions ? 3. Explain the factors which influence the chemical reactions ? 4. Explain how Energy barrier and Orientation barrier affect the rate of a chemical reaction ? 1.