REACTION RATES EQUILIBRIUM Rates of Reaction COLLISION THEORY

REACTION RATES & EQUILIBRIUM Rates of Reaction
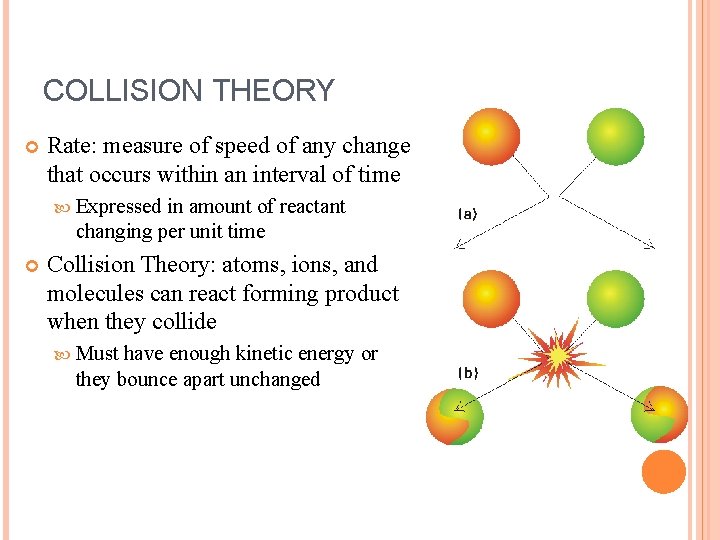
COLLISION THEORY Rate: measure of speed of any change that occurs within an interval of time Expressed in amount of reactant changing per unit time Collision Theory: atoms, ions, and molecules can react forming product when they collide Must have enough kinetic energy or they bounce apart unchanged
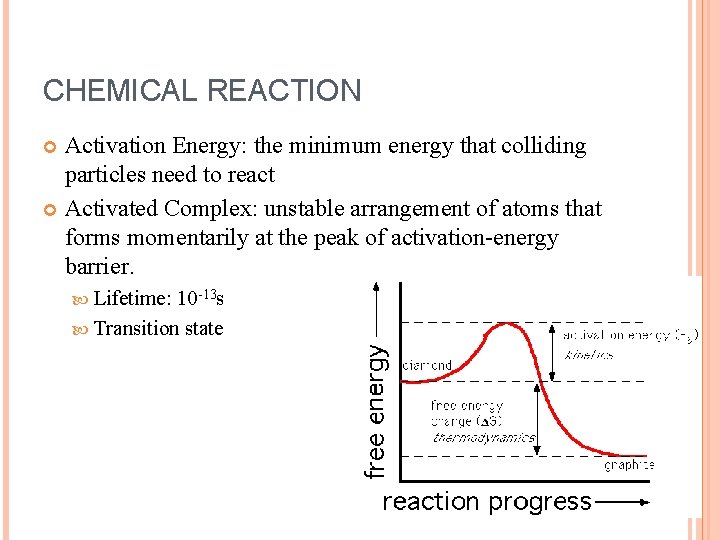
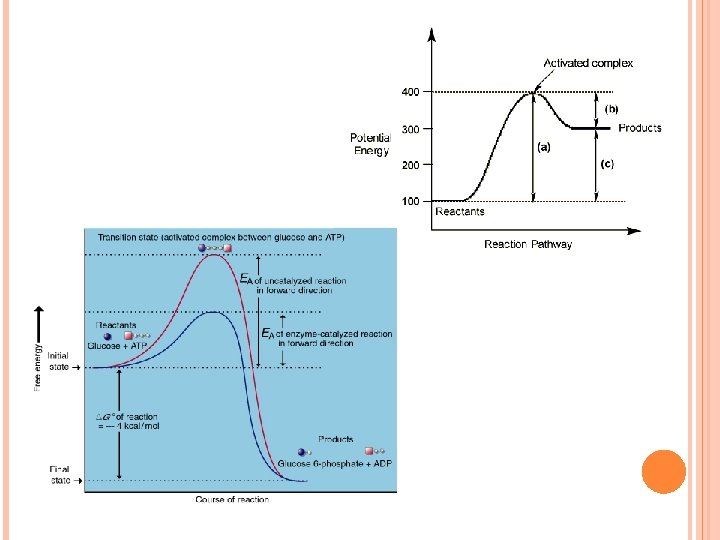
CHEMICAL REACTION Activation Energy: the minimum energy that colliding particles need to react Activated Complex: unstable arrangement of atoms that forms momentarily at the peak of activation-energy barrier. Lifetime: 10 -13 s Transition state

FACTORS AFFECTING RATION RATE Temperature Hi temp >KE more collisions inc. rxn. rate Concentration [Hi] more collision inc. rxn. rate Particle Size Smaller particle size greater surface area Greater surface area, increase exposure to collision, rate Catalyst: increases a reaction without being changed Inhibitor: and inc. rxn. interferes with a catalyst Reaction slows or even stops


REVERSIBLE REACTIONS AND EQUILIBRIUM
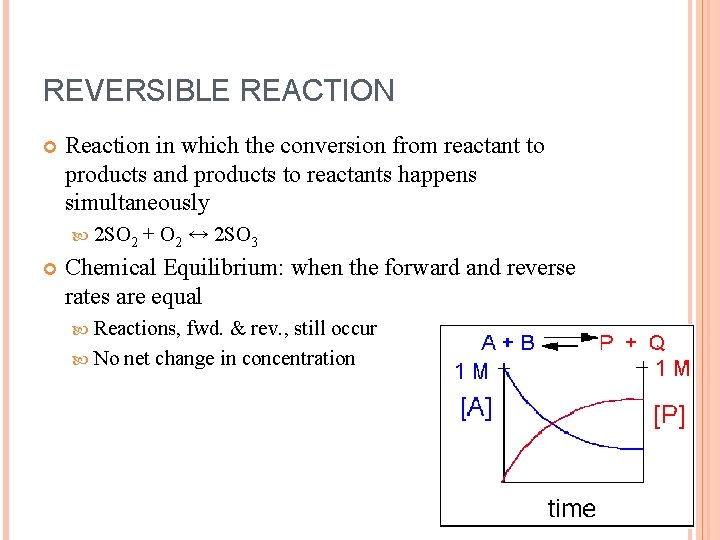
REVERSIBLE REACTION Reaction in which the conversion from reactant to products and products to reactants happens simultaneously 2 SO 2 + O 2 ↔ 2 SO 3 Chemical Equilibrium: when the forward and reverse rates are equal Reactions, fwd. & rev. , still occur No net change in concentration
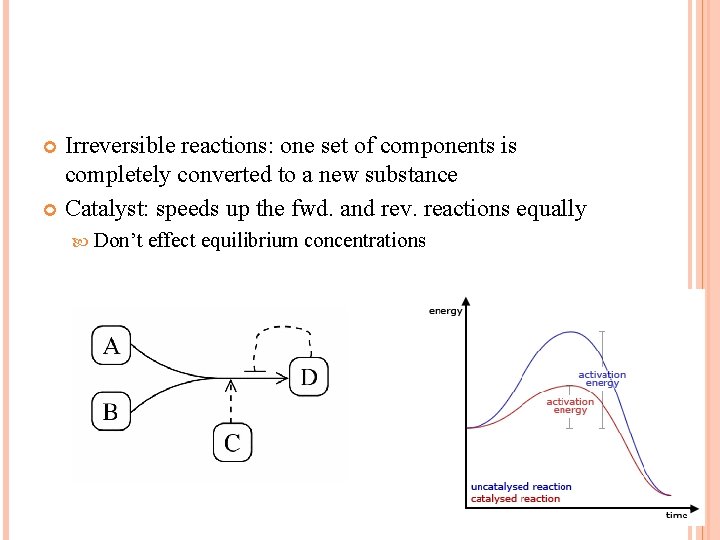
Irreversible reactions: one set of components is completely converted to a new substance Catalyst: speeds up the fwd. and rev. reactions equally Don’t effect equilibrium concentrations

LE CH TELIER’S PRINCIPLE When stress is applied to a system, the system will responds to relieve the stress Shift equilibrium position Stresses: Concentration Temperature Pressure
LE CH TELIER’S PRINCIPLE Concentration H 2 CO 3 ↔ CO 2 + H 2 O Add CO 2 Shifts Remove CO 2 Shifts left right Add H 2 O Shifts left
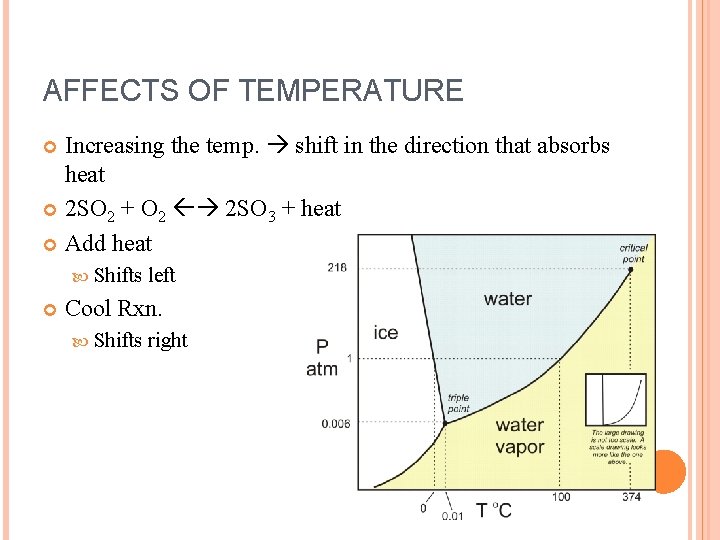
AFFECTS OF TEMPERATURE Increasing the temp. shift in the direction that absorbs heat 2 SO 2 + O 2 2 SO 3 + heat Add heat Shifts left Cool Rxn. Shifts right

AFFECTS OF PRESSURE Pressure changes affect gaseous systems that have unequal # of moles of reactants and products Want to optimize room N 2(g) + 3 H 2 (g) 2 NH 3(g) Increase pressure Shifts right Decrease pressure Shifts left

EQUILIBRIUM CONSTANT Keq: ratio of product concentration to reactant concentration at equilibrium raised to the power = # of moles Only the concentration of gases and substances in solution are shown Concentration for pure liquids and solids are not shown

EQUILIBRIUM CONSTANT a. A Keq + b. B c. C + d. D >1: favors products Keq <1: favors reactants

EXAMPLE PROBLEMS A liter of gas mixture at equilibrium contains 0. 0045 mol of N 2 O 4 and 0. 030 mol of NO 2. Write the Keq expression and determine its value. N 2 O 4 2 NO 2

CALCULATING AN EQUILIBRIUM CONSTANT HI placed in a sealed container and comes to equilibrium; equilibrium reaction is: 2 HI(g) H 2(g) + I 2(g) Equilibrium concentrations: [HI] = 0. 54 M [H 2] = 1. 72 M [I 2] = 1. 72 M Substitute Keq = [H 2] [I 2] [HI]2 concentrations: Keq= [1. 72] = 2. 96 [0. 54]2 0. 29 = 10. 1 or 1. 0 x 101 2 significant figures
- Slides: 17