Reaction Mechanisms The sequence of events that describes

Reaction Mechanisms The sequence of events that describes the actual process by which reactants become products is called the reaction mechanism. • Reactions may occur all at once or through several discrete steps. • Each of these processes is known as an elementary reaction • The overall reaction cannot occur faster than this slowest, rate-determining step. also called the RDS

Mechanisms • Intermediate – A species that is created in one step and consumed in the other • Catalyst – A species that is present originally then reforms later on during the reaction – It is not written in the overall equation, but you may see it noted above the reaction arrow.
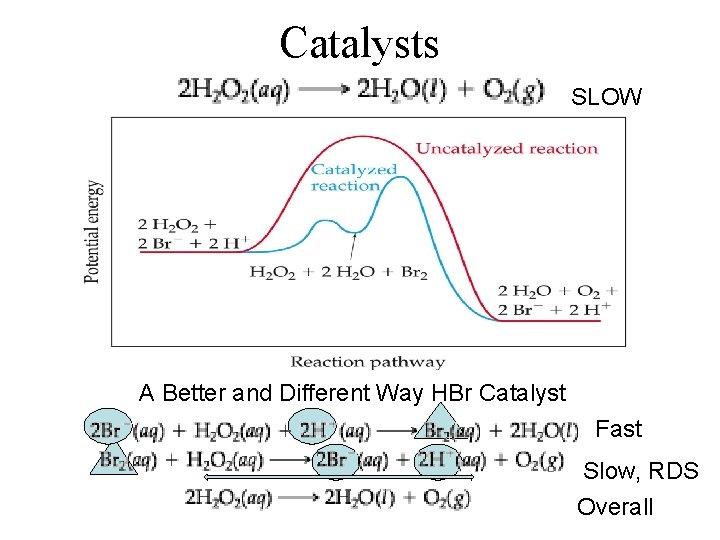
Catalysts SLOW A Better and Different Way HBr Catalyst Fast Slow, RDS Overall
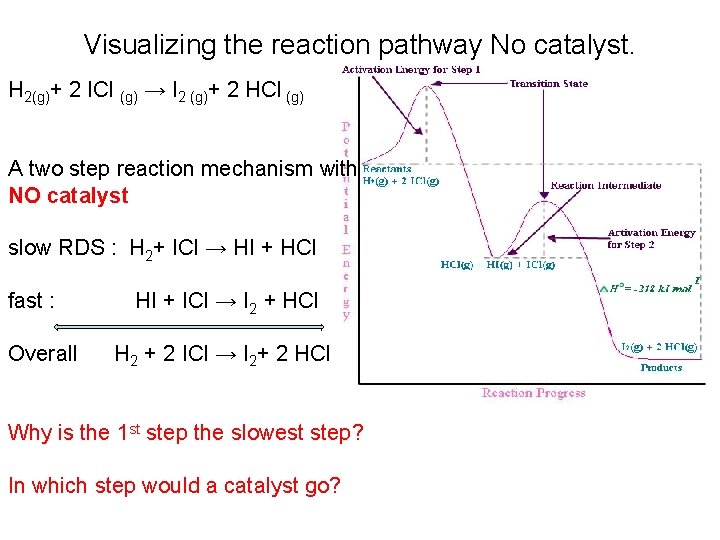
Visualizing the reaction pathway No catalyst. H 2(g)+ 2 ICl (g) → I 2 (g)+ 2 HCl (g) A two step reaction mechanism with NO catalyst slow RDS : H 2+ ICl → HI + HCl fast : Overall HI + ICl → I 2 + HCl H 2 + 2 ICl → I 2+ 2 HCl Why is the 1 st step the slowest step? In which step would a catalyst go?

Reaction Mechanisms • • The molecularity of a process tells how many molecules are involved in the process. The rate law for an elementary step CAN be written directly from that step. Ter steps is NOT likely, or highly improbable The rate law for the rate-determining step becomes the rate law for the overall reaction.

A Mechanism With A Slow Step Gives Rate ORDER Slow step: Fast step: H 2 O 2 + I- H 2 O + OIH 2 O 2 + OI- H 2 O + O 2 + I- ______________________________ Net equation: 2 H 2 O 2 2 H 2 O + O 2 The catalyst? IThe intermediate? OIThe slow step is the rate-determining step, so from it the Rate of reaction is? Rate = rate of slow step = k[H 2 O 2][I-]

Determining Molecularity and Rate Laws for Elementary Steps PROBLEM: The following elementary steps are proposed for a reaction mechanism: (1) NO 2 Cl(g) → NO 2(g) + Cl(g) (slow) (2) NO 2 Cl(g) + Cl(g) → NO 2(g) + Cl 2(g) (fast) Sample Problem (a) (b) (c) Write the overall balanced equation. Determine the molecularity of each step. Write the rate law for each step. PLAN: We find the overall equation from the sum of the elementary steps. The molecularity of each step equals the total number of reactant particles. We write the rate law for each step using the molecularities as reaction orders.

Sample Problem SOLUTION: (a) Writing the overall balanced equation: (1) NO 2 Cl(g) → NO 2(g) + Cl(g) (2) NO 2 Cl(g) + Cl(g) → NO 2(g) + Cl 2(g) 2 NO 2 Cl(g) → 2 NO 2(g) + Cl 2(g) (b) Step(1) is unimolecular, it is the slowest step (simplest). Step(2) is bimolecular. (c) rate 1 = k 1[NO 2 Cl] rate 2 = k 2[NO 2 Cl][Cl] The rate law for the ratedetermining step becomes the rate law for the overall reaction. Overall rate of reaction is rate 1 = k 1[NO 2 Cl]

Mechanisms with a Slow Initial Step The overall reaction 2 NO 2(g) + F 2(g) → 2 NO 2 F(g) has an experimental rate law Rate = k[NO 2][F 2]. The accepted mechanism is (1) NO 2(g) + F 2(g) → NO 2 F(g) + F(g) (2) NO 2(g) + F(g) → NO 2 F(g) [slow; rate determining] [fast] 2 NO 2(g) + F 2(g) → 2 NO 2 F(g) rate 1 = k 1[NO 2[F 2] Step 1 is the slow step, and rate 1 correlates with the observed rate law. The mechanism is therefore reasonable.

One more: Mechanisms with a Fast Initial Step The overall reaction 2 NO (g) + O 2(g) → 2 NO 2(g) has an experimental rate law Rate = k[NO]2[O 2]. A proposed mechanism is (1) NO(g) + O 2(g) D NO 3(g) (2) NO 3(g) + NO(g) → 2 NO 2(g) 2 NO (g) + O 2(g) → 2 NO 2(g) rateoverall = k 2[NO 3][NO] [fast] [slow; rate determining] RDS
- Slides: 10