Collision Theory and Reaction Rates Collision Theory Chemical














- Slides: 19

Collision Theory and Reaction Rates

Collision Theory • Chemical reactions only occur when atoms, ions, or molecules collide. • The species must make contact for a reaction to occur. • Collisions must have proper orientation and sufficient energy to be successful.

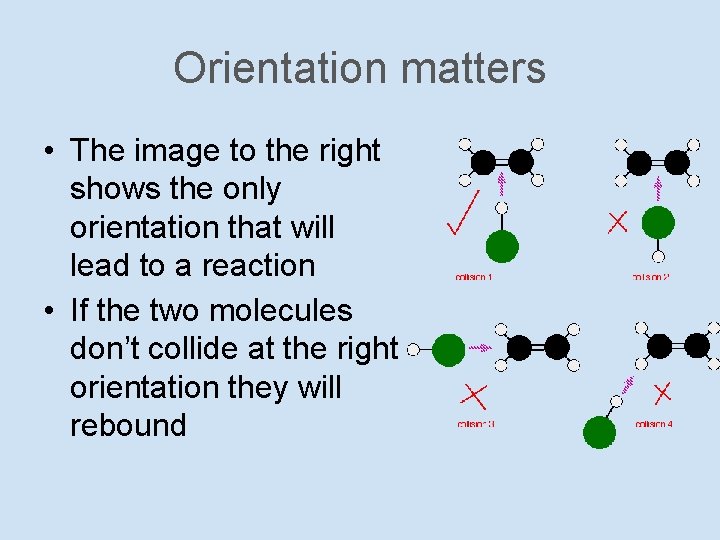
Orientation matters • The image to the right shows the only orientation that will lead to a reaction • If the two molecules don’t collide at the right orientation they will rebound

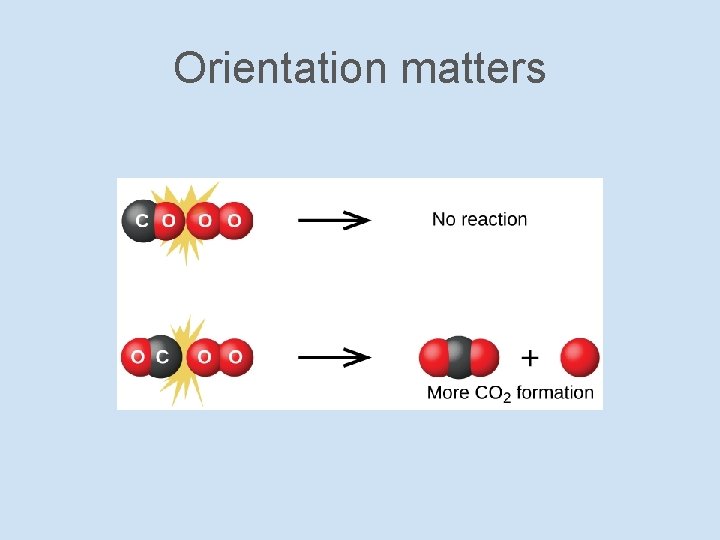
Orientation matters

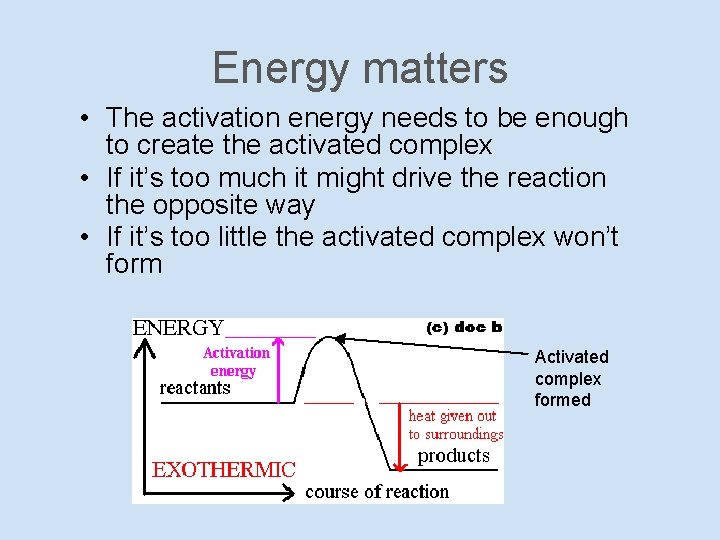
Energy matters • The activation energy needs to be enough to create the activated complex • If it’s too much it might drive the reaction the opposite way • If it’s too little the activated complex won’t form Activated complex formed

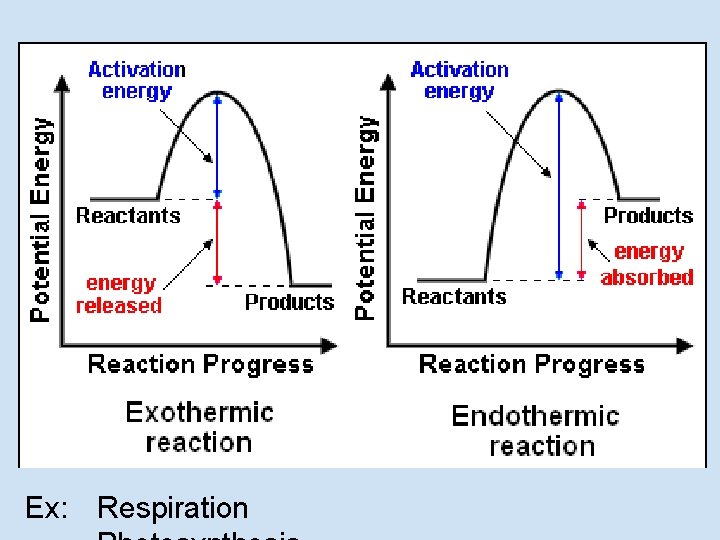
Ex: Respiration

Other connections • The graph you saw for the activation energy helps you to identify if a reaction is exothermic or endothermic • The activation energy is connected to the overall energy of the reaction • It takes energy to break bonds (Activation energy) and when bonds are reformed some energy is released.

Quiz check! True or false: • As long as there is enough energy the reaction will go forward • A poor orientation can be overcome with more energy • When a product is being formed and is in a high energy intermediate step we call that a. b. c. d. Homogenous reaction Heterogeneous reaction Activated complex Rebounded actuality

Reaction Rates ➢ The rate of a chemical reaction is affected by many factors, including: ● Concentration ● Pressure ● Temperature ● Surface area ● Catalyst

Concentration of reactants as concentration of reactants increases the rate of reaction generally increases Remember - Particles (molecules & atoms/ions) have to collide in order for a reaction to occur. Ex: If you're driving a car, which freeway would you have a HIGHER chance of getting into a car accident?

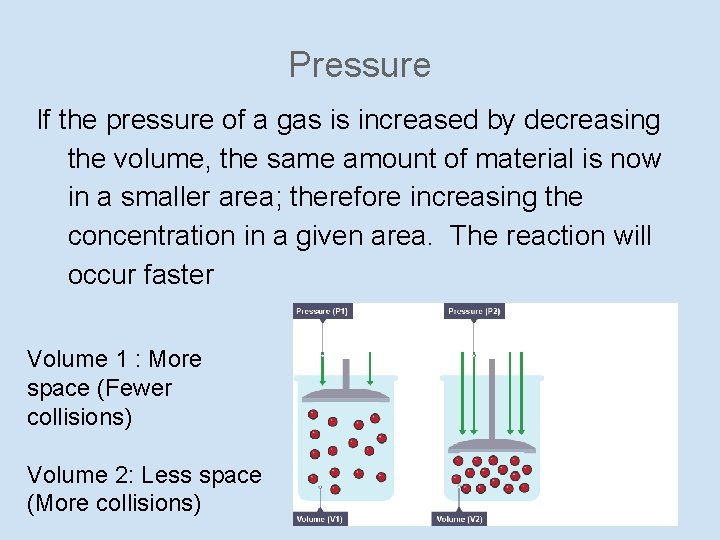
Pressure If the pressure of a gas is increased by decreasing the volume, the same amount of material is now in a smaller area; therefore increasing the concentration in a given area. The reaction will occur faster Volume 1 : More space (Fewer collisions) Volume 2: Less space (More collisions)

Temperature of the reaction Increasing temperature increases the speed of the particles increasing the rate of a reaction Decreasing the temperature decrease the speed of the particles decreasing the rate of a reaction food spoils more quickly at room temperature than in a refrigerator bacteria grow faster at RT than at lower temperatures

Presence of a catalyst a substance that increases the rate of a reaction without being consumed in the reaction • Enzymes ● biological catalysts ● proteins that increase the rate of biochemical reactions • Catalytic converters ● Expensive metals (platinum) allow harmful gases to be broken down into H 2 O, CO 2, N 2

Surface Area as surface area increases the rate of reaction generally increases Ex: A chunk of iron rusting releases heat VERY SLOWLY. Small amount of heat Iron rusting in a hand warmer releases heat VERY QUICKLY LARGE AMOUNT OF HEAT

Question ➢ What is not something that affects reaction rate? : A. Orientation B. Presence of a catalyst C. Temperature D. Concentration of reactants

Reaction Rates ➢ The speed of an object or event is the change that occurs in a given time interval. ● Speed of a car = change in distance change in time = Δd Δt Remember, the term change (Δ) always refers to final value minus initial value.

Reaction Rates ➢ Similarly, the rate (or speed) of a reaction can be determined: Rate = change in concentration (or moles) of product change in time Rate = Δ (concentration or moles) Δt

Question ➢ How is reaction rate measured? a. Speed/Distance b. Distance/time c. Mols of product/mols of reactant d. Concentration/time

Comprehension check 1. What 2 factors of collision theory influence a reaction? 1. Pick one of the variables that impacts rate of a reaction. Explain how changing that variable impacts the rate of a reaction