Chemical Reaction Energy Reaction Rates Chemical Equilibrium Chapter

Chemical Reaction: • Energy • Reaction Rates • Chemical Equilibrium

Chapter Outline • Energy change in chemical reactions • Reaction rate: o Concentration o Orientation o Activation energy o Catalyst • Chemical equilibrium: Le Châtelier’s principle: o Concentration o Pressure/Volume o Temperature 2

Energy do not have mass and volume (<-> Matter) • Energy is anything that has the capacity to do work • Chemistry is the study of matter, Chemical process involve energy change • it can cause physical and/or chemical changes in matter 3

Matter Possesses Energy • when a piece of matter possesses energy, it can give some or all of it to another object • all chemical and physical changes result in the matter changing energy 4

Kinds of Energy Kinetic and Potential • Kinetic Energy is energy of motion, or energy that is being transferred from one object to another (running water) • Potential Energy is energy that is stored (water above the dam) 5

Some Forms of Energy • Electrical ü kinetic energy associated with the flow of electrical charge • Heat or Thermal Energy ü kinetic energy associated with molecular motion • Light or Radiant Energy ü kinetic energy associated with energy transitions in an atom • Nuclear ü potential energy in the nucleus of atoms • Chemical ü potential energy in the attachment of atoms or because of their position (example: Battery) 6

Units of Energy • calorie (cal) is the amount of energy needed to raise one gram of water by 1°C ükcal = energy needed to raise 1000 g of water 1°C üfood Calories = kcals Energy Conversion Factors 1 calorie (cal) 1 Calorie (Cal) 1 kilowatt-hour (k. Wh) = = = 4. 184 joules (J) 1000 calories (cal) 3. 60 x 106 joules (J) 7
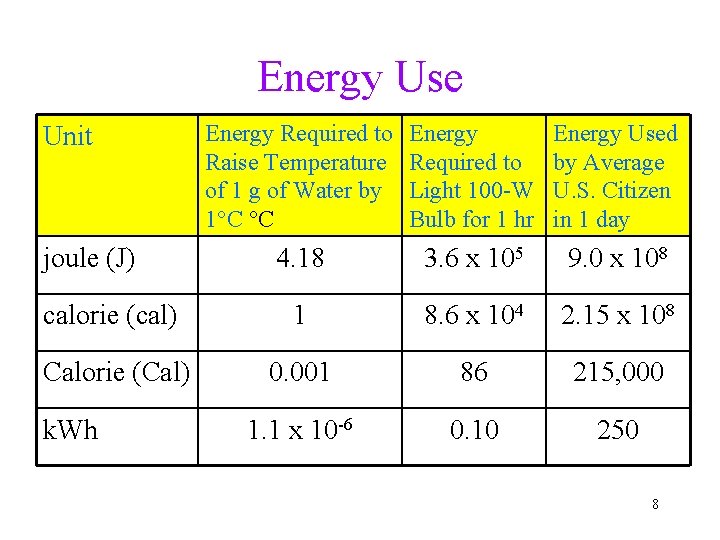
Energy Use Energy Required to Raise Temperature of 1 g of Water by 1°C °C Energy Required to Light 100 -W Bulb for 1 hr Energy Used by Average U. S. Citizen in 1 day 4. 18 3. 6 x 105 9. 0 x 108 calorie (cal) 1 8. 6 x 104 2. 15 x 108 Calorie (Cal) 0. 001 86 215, 000 1. 1 x 10 -6 0. 10 250 Unit joule (J) k. Wh 8

Energy in Chemical Changes Exothermic reaction: Reaction releases heat to the environment. • Product contains less energy than reactant. • Heat can be considered as PRODUCT. Endothermic reaction: Reaction absorbs heat from the environment. • Product contains more energy. • Heat can be considered as REACTANT. 9

Examples of Exothermic Reaction All Combustion reactions: Hydrogen gas burns in the air: O 2 + 2 H 2 O + heat Acid-Base Neutralization Reaction: Na. OH + HCl Na. Cl + 2 H 2 O + heat All explosive reactions 10

Examples of Endothermic Reaction • Mercury(II) oxide decomposes under heat 2 Hg. O(s) + heat O 2(g) + 2 Hg(l) • Soda drink loses taste at room temperature: H 2 CO 3 + heat CO 2 + H 2 O 11

Bond Dissociation Energy Breaking a stable chemical bond requires energy input: Endothermic reaction Bond Dissociation Energy: energy to equally breaking a covalent bond. Higher bond dissociation energy means stronger bond. 12

Calculate the Energy involved in Chemical Reaction For a certain chemical reaction, the energy absorbed or released depends on the amount of product, or the limiting reactant. The more product is generated, the more heat is involved. The energy involved (kcal/mol or k. J/mol) can be considered as conversion factor. 13

Determine how much energy is released when 100. g of propane is completely reacted with oxygen. 1. 20 x 103 kcal Problem solving plan: Mass propane mol propane energy released 14

How Does Chemical Reaction Take Place? • In order for a reaction to occur, the reacting molecules must collide with each other. Also two other factors: 1. Whether the collision has enough energy to “start to break the bonds holding reactant molecules together. " 2. Whether the reacting molecules collide in the proper orientation for new bonds to form. 15

Effective Collisions: Collisions with sufficient energy and proper orientation (and therefore the reaction occurs). • The higher the frequency of effective collisions, the faster the reaction rate. • There is a minimum energy needed for a collision to be effective: Activation energy. 16

Effective Collisions: Kinetic Energy Factor For a collision to lead to overcoming the energy barrier, the reacting molecules must have sufficient kinetic energy 17
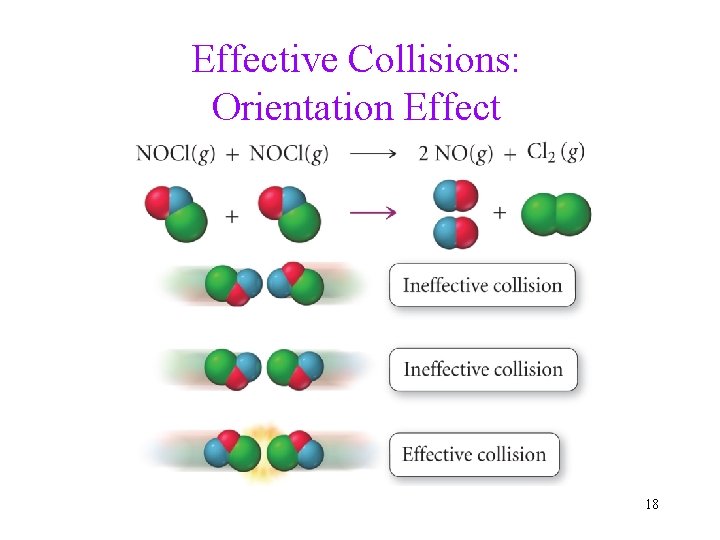
Effective Collisions: Orientation Effect 18

Activation Energy • Reactants are often stable, requiring energy to reto dissociate and form products. • Activation Energy: The energy barrier that prevents any collision between molecules from being an effective collision • The larger the activation energy of a reaction, the slower it will be. Comparable to the Hurdles: The taller hurdle or slower runner, the harder for one to run overpass, fewer people will be able to clear. 19

Many Chemical Reactions Need Initial “Push” to Start Rubbing a match head against a rough surface provides the activation energy needed for the match to ignite. 20
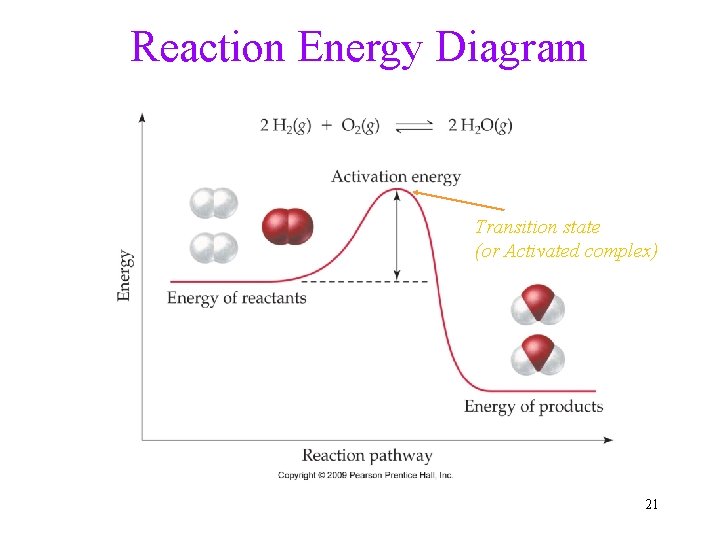
Reaction Energy Diagram Transition state (or Activated complex) 21
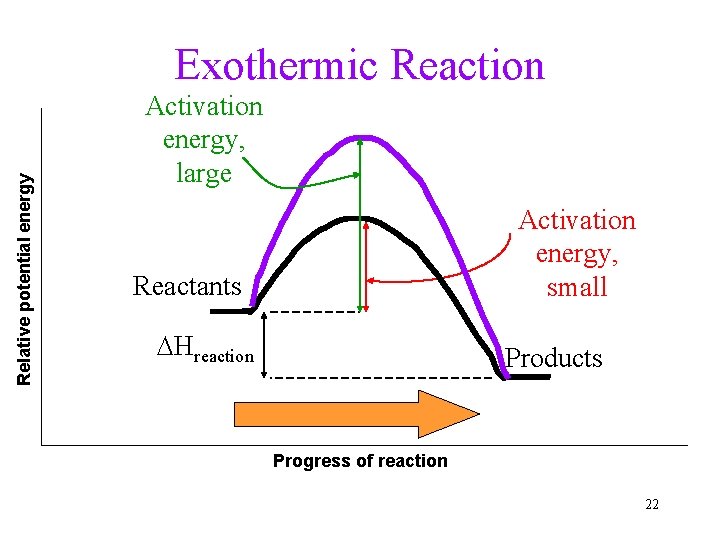
Relative potential energy Exothermic Reaction Activation energy, large Activation energy, small Reactants DHreaction Products Progress of reaction 22
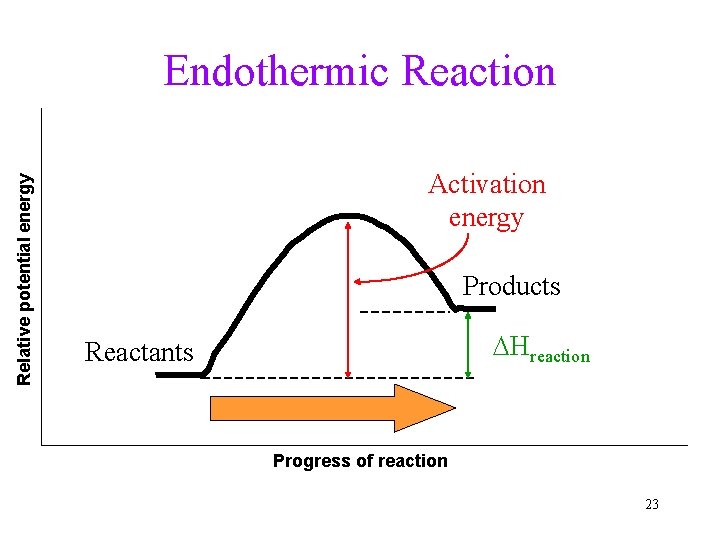
Relative potential energy Endothermic Reaction Activation energy Products DHreaction Reactants Progress of reaction 23

Reaction Rates • Some chemical reactions proceed rapidly. üLike the precipitation and acid-base reactions • Other reactions proceed slowly. üLike the discoloration of a sofa undersunlight. • The rate of a reaction is measured in the amount of reactant that changes into product in a given period of time. • Chemists study ways of controlling reaction rates. 24

Factors Effecting Reaction Rate: Reactant Concentration • The higher the concentration of reactant molecules, the faster the reaction will generally go. Why? üIncreases the frequency of reactant molecule collisions Examples: üHigh concentration (full strength) of CLORAX might overbleach or even damage fiber due to high reaction rate üWhen using Drano (Na. OH-based solution), avoid running water (water dilutes Na. OH) 25

Factors Effecting Reaction Rate: Temperature • Increasing the temperature, more molecules in the sample with enough energy to collide and overcome the activation energy. • Increasing the temperature also increases the frequency of collisions. • Example: Warm temperature quicken reaction such as CLORAX or DRANO in cleaning. 26
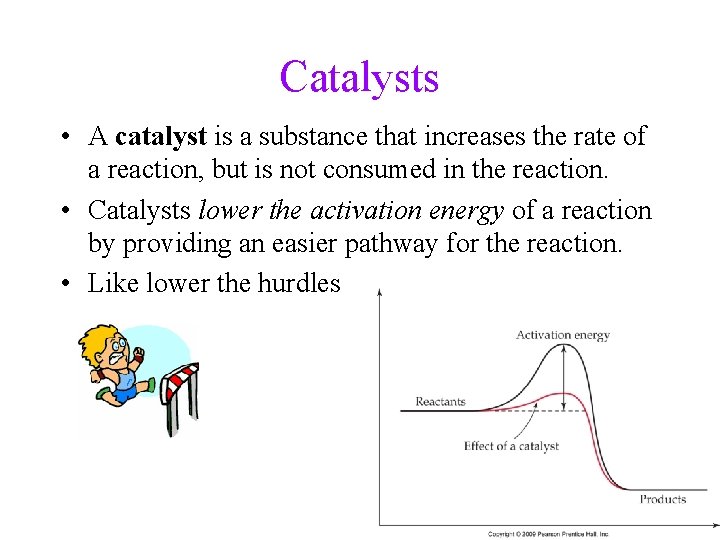
Catalysts • A catalyst is a substance that increases the rate of a reaction, but is not consumed in the reaction. • Catalysts lower the activation energy of a reaction by providing an easier pathway for the reaction. • Like lower the hurdles 27

Enzymes: Proteins as Biochemical Catalysts Enzymes are protein molecules produced by living organisms that catalyze chemical reactions. • The enzyme molecules have an active site to which organic molecules bind. When the organic molecule is bound to the active site, certain bonds are weakened. Weakening bond makes chemical change easier. ü i. e. , the activation energy is lowered. 28
Amylase: An Enzyme Breaking down starch molecules 29

Overview of Factors Affecting Reaction Rates 30

Many Chemical Reactions are Reversible • If the products of a reaction are removed from the system as they are made, then a chemical reaction will proceed until the limiting reactants are used up. • However, if the products are allowed to accumulate, they will start reacting together to form the original reactants. Product Reactant: Reverse Reaction. • Reactions that can proceed in both the forward and reverse directions are called Reversible Reactions. 31

Examples of Reversible Reactions • Photochromic sunglass: Transparent + Light Dark • Temperature dependent coloration: N 2 O 4 + heat 2 NO 2 • Rechargeable battery: Charged Depleted + electricity 32

Reaction Dynamics, Continued • The forward reaction _______ as the amounts of reactants decreases. • At the same time, the reverse reaction ____ as the concentration of the products increases. • Eventually, the forward reaction is using reactants and making products as fast as the reverse reaction is using products and making reactants. This is called Chemical Equilibrium. ü Dynamic equilibrium is reached when the rates of two opposite processes are the same. 33

Equilibrium Equal • of forward rxn = of reverse rxn. • Concentrations of reactants and products are not always equal. • Some equilibria occur reactant ⇌ product position of equilibrium favors the products. • Other equilibria occur reactant ⇌ product position of equilibrium favors the reactants. 34

Reaching Equilibrium: Population Changes When country A feel overcrowded, some will emigrate to country B. The population of country B is so low that no one emigrates to counry A. As time passes, emigration will occur in both directions at the same rate, leading to populations in country A and country B are constant, though not necessarily equal. 35

Equilibrium Constant • Even though the concentrations of reactants and products are not equal at equilibrium, there is a relationship between them. • For the reaction H 2(g) + I 2(g) 2 HI(g) at equilibrium, the ratio of the concentrations in [ ] raised to the power of their coefficients is constant. 36

Equilibrium Constant, Continued • For the general equation a. A + b. B c. C + d. D, the relationship is given below: üThe lowercase letters represent the coefficients of the balanced chemical equation. üAlways products over reactants. • The constant is called the equilibrium constant, Keq. 37

What Does the Value of Keq Imply? • When the value of Keq > > 1, we know that when the reaction reaches equilibrium, there will be many more product molecules present than reactant molecules. ü The position of equilibrium favors products. • When the value of Keq < < 1, we know that when the reaction reaches equilibrium, there will be many more reactant molecules present than product molecules. ü The position of equilibrium favors reactants. 38
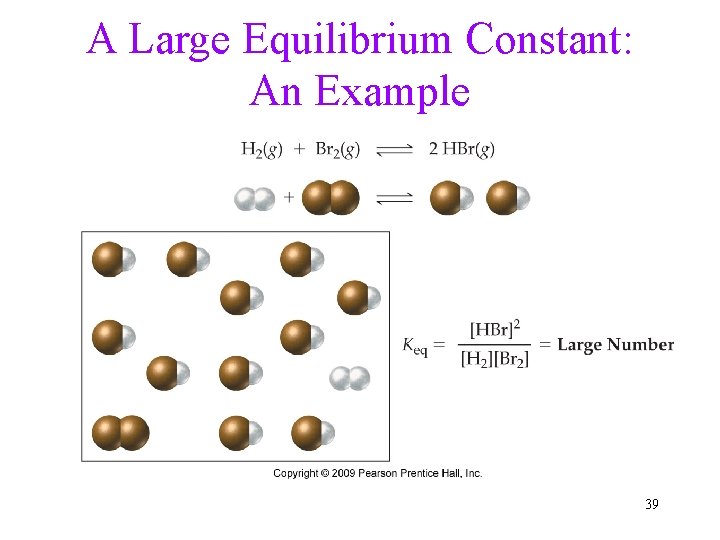
A Large Equilibrium Constant: An Example 39

Disturbing and Re-Establishing Equilibrium • Once a reaction is at equilibrium, the concentrations of all the reactants and products remain constant. • However, if the conditions are changed, the concentrations of all the chemicals will change (not equilibrium) until equilibrium is reestablished. • The new concentrations will be different, but the equilibrium constant will be the same. ü Unless you change the temperature. 40

Le Châtelier’s Principle • Le Châtelier’s principle guides us in predicting the effect on the position of equilibrium when conditions change. • “When a chemical system at equilibrium is disturbed, the system shifts in a direction that will minimize the disturbance. ” 41
![Le Chatelier: Concentration Effect Scenario A: [Reactant] , Other reactants , products (shift toward Le Chatelier: Concentration Effect Scenario A: [Reactant] , Other reactants , products (shift toward](http://slidetodoc.com/presentation_image_h2/8e1d4633f9bba313563275a8bba68061/image-42.jpg)
Le Chatelier: Concentration Effect Scenario A: [Reactant] , Other reactants , products (shift toward product) üThat has the same Keq. Scenario B: [Product] , Other products , reactants (shift toward reactant) üYou can keep removing product in an equilibrium to drive a reaction to completion! 42
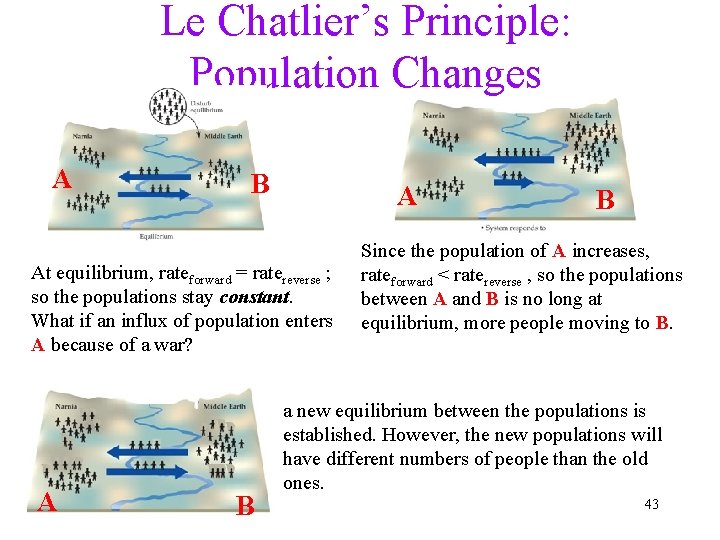
Le Chatlier’s Principle: Population Changes A B A At equilibrium, rateforward = ratereverse ; so the populations stay constant. What if an influx of population enters A because of a war? A B B Since the population of A increases, rateforward < ratereverse , so the populations between A and B is no long at equilibrium, more people moving to B. a new equilibrium between the populations is established. However, the new populations will have different numbers of people than the old ones. 43
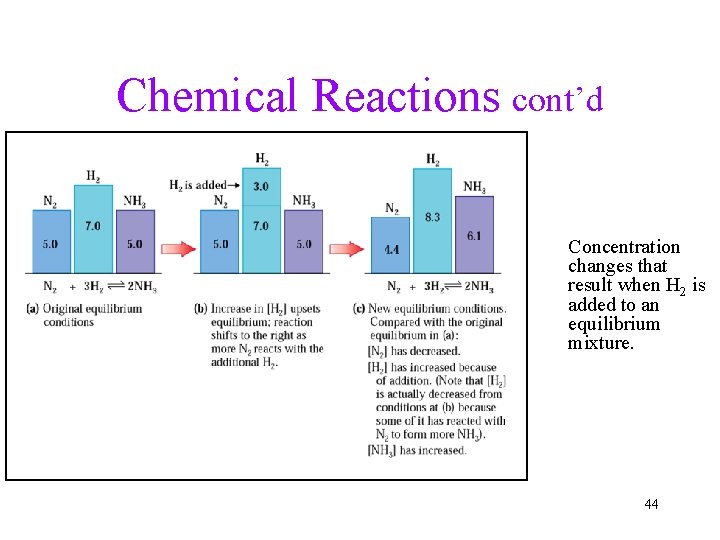
Chemical Reactions cont’d Concentration changes that result when H 2 is added to an equilibrium mixture. 44

Practice—Predict the Effect on the Equilibrium When the Underlined Substance Is Added to the Following Systems: • 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) 45

Practice—Predict the Effect on the Equilibrium When the Underlined Substance Is Removed from the Following Systems: • 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) 46

Effect of Volume Change on Equilibrium • For solids, liquids, or solutions, changing the size of the container has no effect on the concentration. • Changing the volume of a container changes the concentration of a gas. üSame number of moles, but different number of liters, resulting in a different molarity. 47

Effect of Volume Change for Gas on Equilibrium • Decreasing the size of the container increases the concentration of all the gases in the container. • According to Le Châtelier’s principle, the equilibrium should shift to remove that pressure. • The way to reduce the pressure is to reduce the number of molecules in the container. • When the volume decreases, the equilibrium shifts to the side with fewer molecules. 48

The Effect of Volume Change on Equilibrium, Continued When Sincethe there pressure are more is decreased gas molecules by increasing on the reactants volume, the side position of theof reaction, equilibrium whenshifts the toward pressure the is side increased with thethe greater position number of of equilibrium molecules—the shifts toward reactant the products. side. 49

Volume Changes on Equilibrium Volume decrease: dark brown color fades away (2 NO 2→N 2 O 4) Volume increase: brown color turns darker (N 2 O 4→ 2 NO 2)50

Practice—Predict the Effect on the Equilibrium When the Volume Is Reduced. • 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) 51

The Effect of Temperature Changes on Equilibrium • Exothermic reactions release Heat: Reactant Product + Heat • Endothermic reactions absorb Heat: Reactant + Heat Product • Increase temperature = Add Heat • Decrease temperature = Remove Heat 52
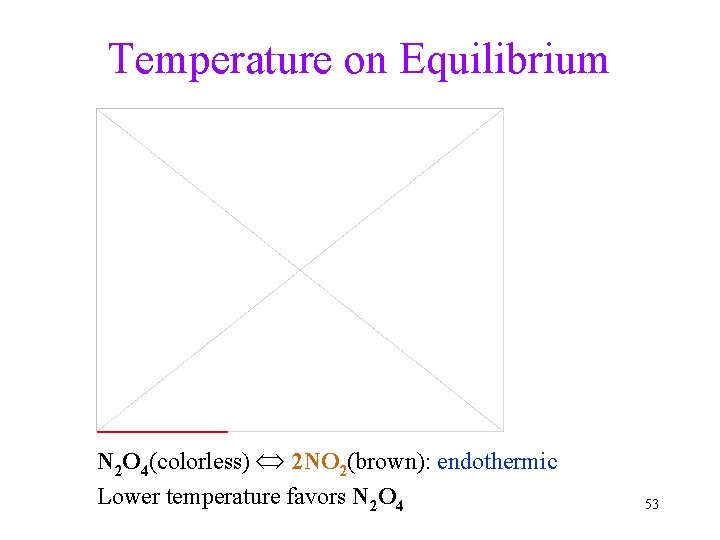
Temperature on Equilibrium Online video: N 2 O 4(colorless) 2 NO 2(brown): endothermic Lower temperature favors N 2 O 4 53

Temperature affects the Equilibrium Co. Cl 64 - + 6 H 2 O Co(H 2 O)62+ + 6 Cl- : exothermic Lower temperature favors Co(H 2 O)62+ 54

Practice—Predict the Effect on the Equilibrium When the Underlined Substance Is Added to the Following Systems, Continued: • 2 CO 2(g) 2 CO(g) + O 2(g) Shift right, removing some of the added CO 2 and increasing the concentrations of CO and O 2. • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) Shift left, removing some of the added Ba 2+ and reducing the concentration of SO 42 -. • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) Shift right, removing some of the added CO 2 and decreasing the O 2, 55 while increasing the concentration of CO 2.

Practice—Predict the Effect on the Equilibrium When the Volume Is Reduced, Continued. • 2 CO 2(g) 2 CO(g) + O 2(g) Shift left because there are fewer gas molecules on the reactant side than on the product side. • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) No effect because none of the substances are gases. • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) Shift right because there are fewer gas molecules 56 on the product side than on the reactant side.
- Slides: 56