Chemical Kinetics Reaction Rates Reaction Rate How fast




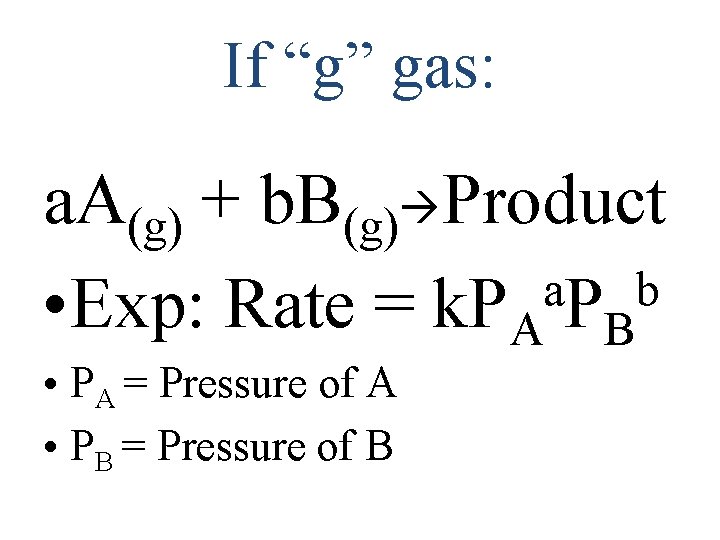



![Think about it. . Rate = [A][B]2 If the initial concentration of B is Think about it. . Rate = [A][B]2 If the initial concentration of B is](https://slidetodoc.com/presentation_image/0374c710da8d37ea1350387dba0a3663/image-10.jpg)


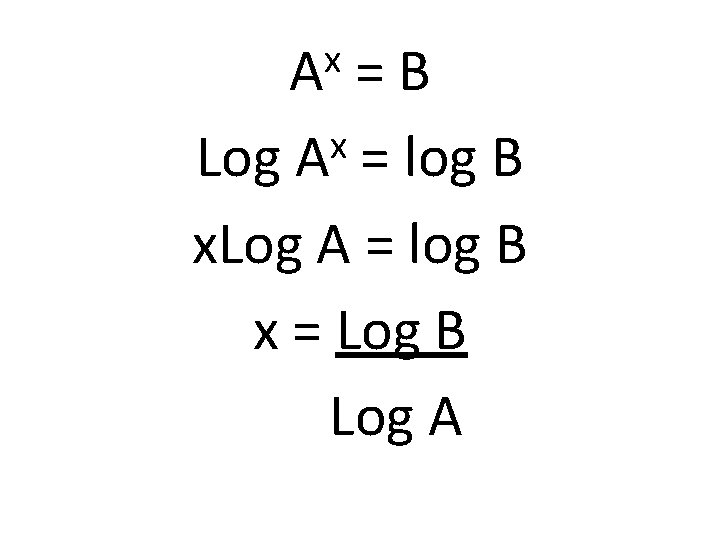

![Experiment [Br. O 3 -] (M) [Br-] (M) [H+] (M) Initial Rate (M/s) 1 Experiment [Br. O 3 -] (M) [Br-] (M) [H+] (M) Initial Rate (M/s) 1](https://slidetodoc.com/presentation_image/0374c710da8d37ea1350387dba0a3663/image-16.jpg)








- Slides: 26

Chemical Kinetics/ Reaction Rates

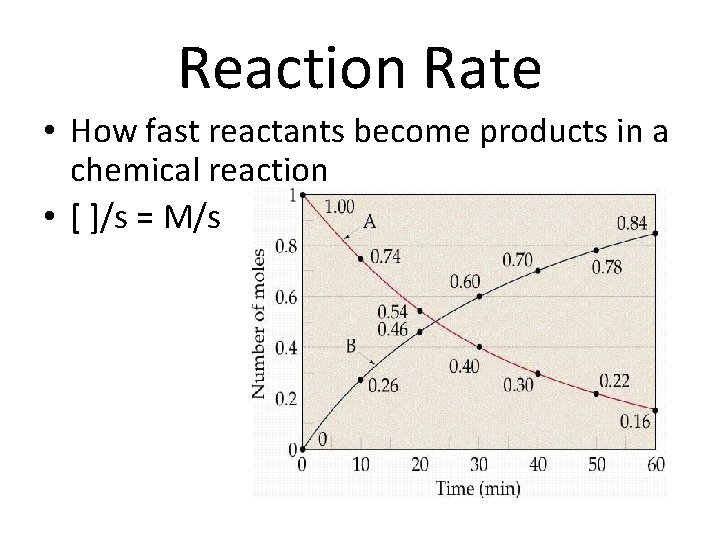
Reaction Rate • How fast reactants become products in a chemical reaction • [ ]/s = M/s

Kinetic Theory 1. 2. 3. All matter is made up of tiny particles are in constant motion All collisions are elastic (no energy change) *In order for particles to react, they must collide! How fast they collide determines reaction rate.

Rate Expressions • Reactions do not necessarily occur in one step Ex: 2 NO 2(g)→ 2 NO(g)+O 2(g) Actually occurs in TWO steps: 2 NO 2→NO 3+NO (fast) NO 3→NO+O 2 (Slow) *The SLOW step is the rate determining step*

Writing Rate Expressions for Rate Determining Step If “aq” Aqueous: a. A(aq) + b. B(aq) P Rate = k[A]a[B]b • [A] = molarity of A • Coefficients exponents • k=rate constant
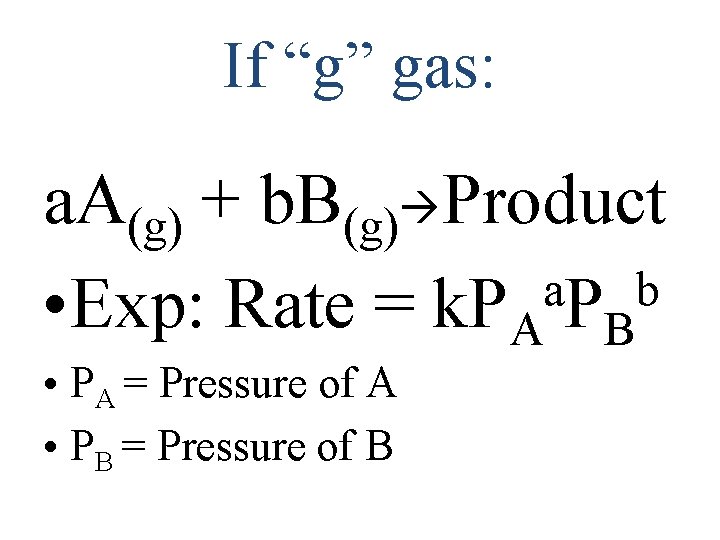
If “g” gas: a. A(g) + b. B(g) Product a b • Exp: Rate = k. PA PB • PA = Pressure of A • PB = Pressure of B

If it’s a combination of (g) and (aq), just pick 1 1 Cl 2(g) + 1 HOOH (aq) 2 HOCl(aq) Rate = k(PCl 2) OR Rate = k[HOOH] Solids don’t contribute! (concentration =1) 2 A(aq)+ B(aq)+ 3 C(s) P Rate = k[A]2[B]

Reaction Order • Reaction Order = total exponents in rate expression (don’t forget 1!) Ex. Rate = k[A][B]2 Reaction Order = 3

Rate Determining Step: 2 A(aq)+ B(aq)+ 3 C(s) P Write the rate law expression, calculate the rate and determine the order. [A] = 0. 40 M [B] = 0. 20 M k = 2. 0 x 102 M-2 s-1
![Think about it Rate AB2 If the initial concentration of B is Think about it. . Rate = [A][B]2 If the initial concentration of B is](https://slidetodoc.com/presentation_image/0374c710da8d37ea1350387dba0a3663/image-10.jpg)
Think about it. . Rate = [A][B]2 If the initial concentration of B is increased from 0. 1 M to 0. 3 M, by what factor will the rate increase?

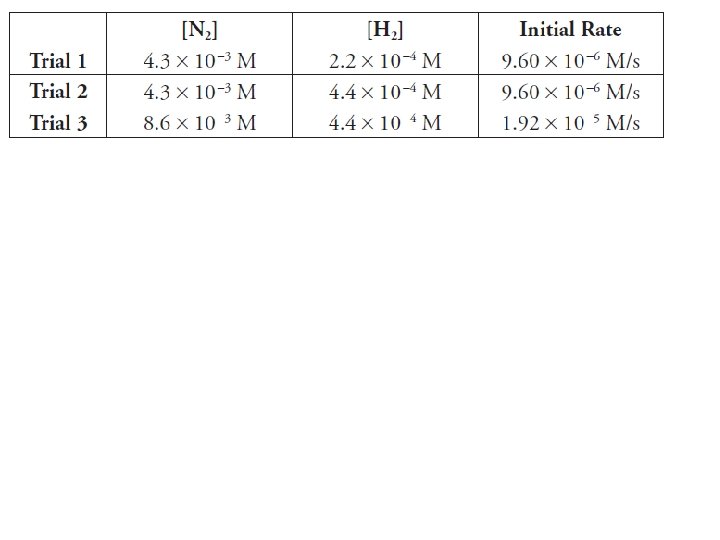
Determining Rate Law from Experimental Results Rate 2 Rate 1 = a b k[A 2] [B 2] a b k[A 1] [B 1] Solve for a & b

How do you solve for x if Ax=B?
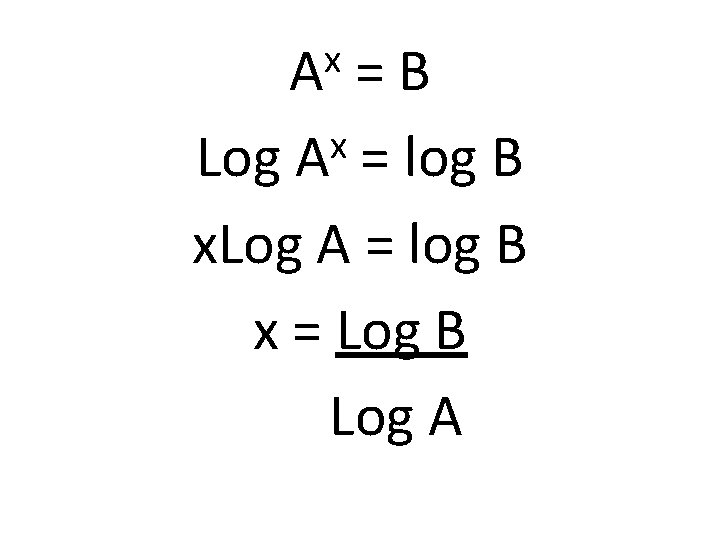
x A =B x Log A = log B x = Log B Log A

Once you have determined rate law, you can use one of the trials to solve for k!

![Experiment Br O 3 M Br M H M Initial Rate Ms 1 Experiment [Br. O 3 -] (M) [Br-] (M) [H+] (M) Initial Rate (M/s) 1](https://slidetodoc.com/presentation_image/0374c710da8d37ea1350387dba0a3663/image-16.jpg)
Experiment [Br. O 3 -] (M) [Br-] (M) [H+] (M) Initial Rate (M/s) 1 0. 10 8. 0 x 10 -4 2 3 4 0. 20 0. 10 0. 20 1. 6 x 10 -3 3. 2 x 10 -3

VIDEO • https: //www. youtube. com/watch? v=Ott. RV 5 yk. P 7 A

The Collision Model Molecules must collide to react!

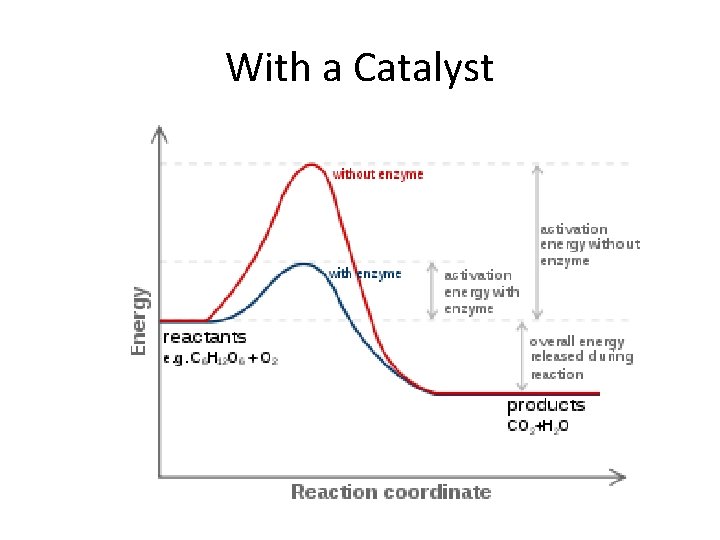
Factors That Affect Reaction Rates • Concentration of Reactants – As the concentration of reactants increases, so does the likelihood that reactant molecules will collide (more particles in a smaller space to run into each other). • Temperature – At higher temperatures, reactants have more KE, move faster, and collide more often. • Catalysts – Speeds up rxn by lowering activation energy. • Surface Area – The larger the surface area, more particles available to collide, the faster the reaction

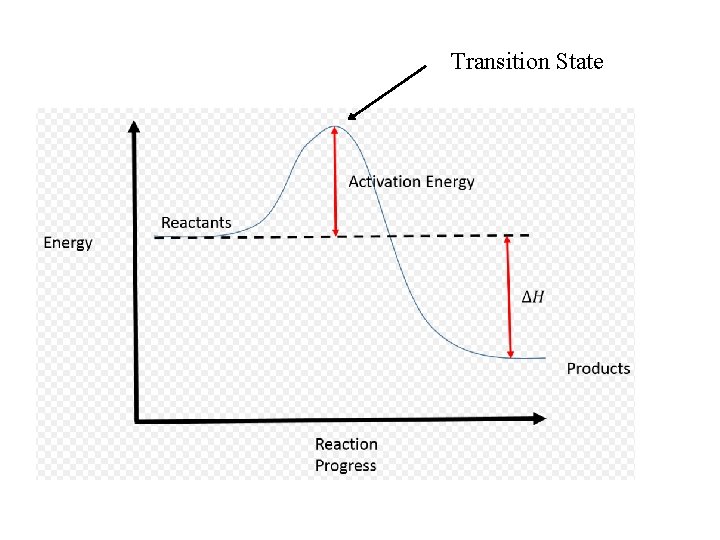
Activation Energy (Ea) • a minimum amount of energy required for reaction to take place *picture ball rolling up a hill

Transition State

With a Catalyst

Rate Constants • Rate constants (k) are DIFFERENT at DIFFERENT TEMPERATURES • Can use the different rate constants to determine activation energy

Clausius-Claperon Eq For: Activation Energy (T 2)(T 1) k 2 Ea= R (T – T ) ln k 2 1 1 R= 8. 314 J/mol K Temperatures MUST be in K

Calculate the activation energy of a reaction whose rate constant is 2. 0 x 103 M-2 S-1 at 27 o. C and is 2. 0 x 106 M-2 S-1 at 77 o. C:

Examples