Chemical Kinetics Factors Affecting Reaction Rate Collision Theory









- Slides: 12

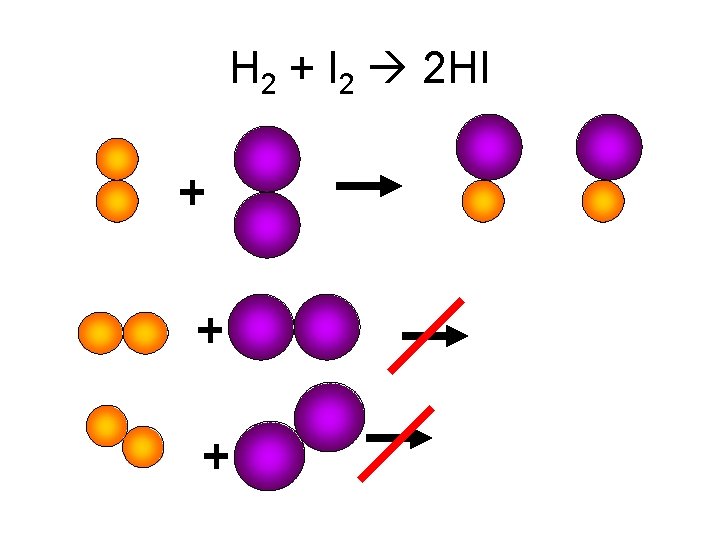
Chemical Kinetics Factors Affecting Reaction Rate Collision Theory • In order for a reaction to occur, reactant particles must collide with each other. • In order for a reaction to occur, the collisions must be effective. • Effective collisions = sufficient energy and correct orientation.

H 2 + I 2 2 HI

Factors Affecting Rate of Reaction 1. Nature of Reactants 2. Surface Area 3. Temperature 4. Concentration 5. Pressure 6. Catalyst Increasing frequency of effective collisions increases rate of reaction. http: //glencoe. com/sec/science/cgi-bin/splitwindow. cgi? top=http: //www. glencoe. com/sec/science/top 2. html&link=http: //www. educationusingpowerpoint. org. uk/Animations/rates%20 of%20 reaction. html http: //phet. colorado. edu/new/simulations/sims. php? sim=Reactions_and_Rates

1. Nature of Reactants • _______________ bonded substances are slower to react than _______________ bonded substances.

How does increasing surface area affect reaction rate? Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) Large Piece of Mg Small Pieces of Mg http: //www. crocodile-clips. com/absorb/AC 4/sample/LR 1502. html

2. Surface Area Draw a curve (graph) showing the relationship between surface area and reaction rate: Reaction Rate As surface area increases, reaction rate ________. Surface Area Relate this relationship to collision theory:

3. Temperature Draw a curve (graph) showing the relationship between temperature and reaction rate: Reaction Rate As temperature increases, reaction rate _________. Temperature Relate this relationship to collision theory:

Concentration HC 2 H 3 O 2 + Acetic Acid H 2 CO 3 Carbonic Acid Na. HCO 3 Sodium Bicarbonate H 2 O Water + Na. C 2 H 3 O 2 Sodium Acetate + H 2 CO 3 Carbonic Acid CO 2 Carbon Dioxide http: //www. crocodile-clips. com/absorb/AC 4/sample/LR 1503. html

4. Concentration Draw a curve (graph) showing the relationship between concentration and reaction rate: Reaction Rate As concentration increases, reaction rate ________. Concentration Relate this relationship to collision theory:

5. Pressure Draw a curve (graph) showing the relationship between pressure and reaction rate: Reaction Rate As pressure increases, reaction rate ________. Pressure Relate this relationship to collision theory: http: //www. crocodile-clips. com/absorb/AC 4/sample/LR 1503. html

6. Catalyst: The addition of a catalyst ______ reaction rate. Definition of a catalyst: http: //www. eepybird. com/dcm 1. html

Homework • Collision Theory: Orange Book: Pg. 110, Q 1 -10 *Pg. 121, Q 1, 2, 4, 5 *Pg. 123, Q 18, 19, 21, 22, 23 * Must do. Other HW questions are optional.