ISOTOPES WHAT IS AN ISOTOPE Atoms of the


























- Slides: 28

ISOTOPES

WHAT IS AN ISOTOPE? � Atoms of the same element with different numbers of neutrons. � What has changed if there is a different number of neutrons. HINT: look at your periodic tables. Something must have changed.

WHAT IS AN ISOTOPE � If you have a different number of neutrons, that means your atomic mass has changed. � Remember that atomic mass = neutrons + protons � THE NUMBER OF PROTONS NEVER CHANGES. � Why doesn’t this number ever change?

WHAT IS AN ISOTOPE? � If you change the number of protons you now have a different element. � EX: If you have an atom of Ne (Neon) with 11 protons now. . You actually would have an atom of Na (Sodium)

CALCULATE THE FOLLOWING IN SODIUM � Protons � Neutrons � Electrons

CALCULATE THE FOLLOWING IN SODIUM � Protons 11 � Neutrons 12 � Electrons 11 � Draw out the model

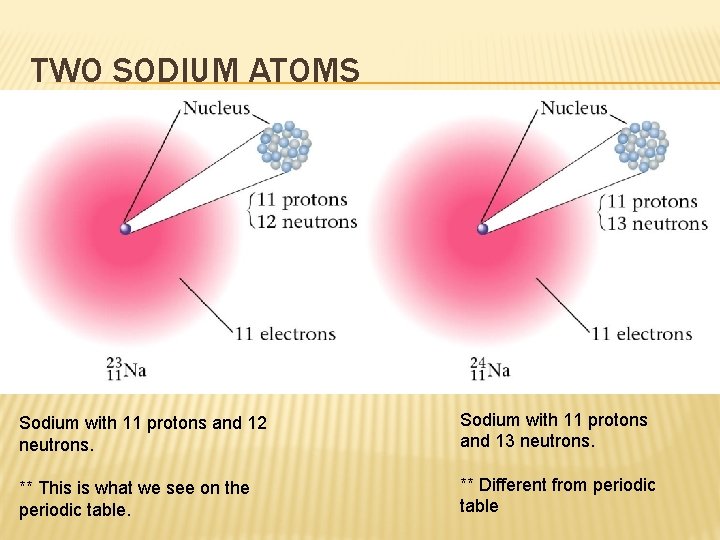
TWO SODIUM ATOMS Sodium with 11 protons and 12 neutrons. Sodium with 11 protons and 13 neutrons. ** This is what we see on the periodic table. ** Different from periodic table

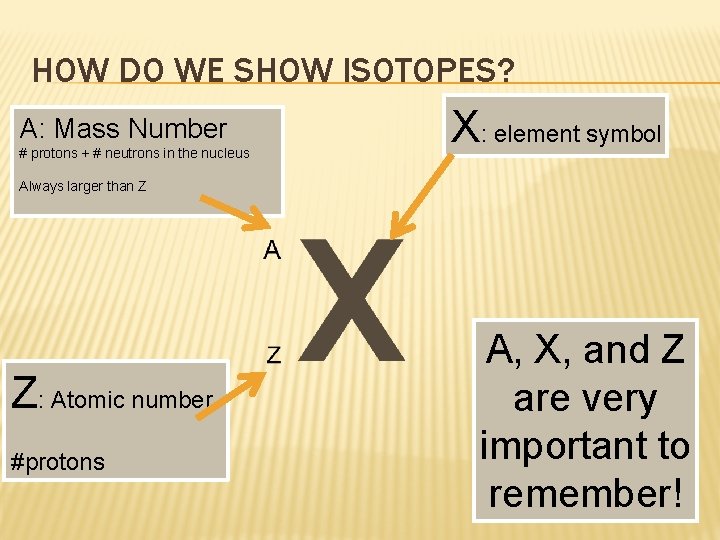
HOW DO WE SHOW ISOTOPES? A: Mass Number # protons + # neutrons in the nucleus X: element symbol Always larger than Z Z: Atomic number #protons A, X, and Z are very important to remember!

TRY IT IN YOUR NOTES � What is the element? � How many protons are in the element? � What is the elements atomic mass? N 23 11

TRY IT IN YOUR NOTES � What is the element? � Sodium N 23 � How many protons are in the element? � 11 � What is the elements atomic mass? � 23 11

TRY IT IN YOUR NOTES � What is the element? � How many protons are in the element? � What is the elements atomic mass? Cl 33 17

TRY IT IN YOUR NOTES � What is the element? � � How many protons are in the element? � � Chlorine 17 What is the elements atomic mass? � 33 Cl 33 17

TRY IT IN YOUR NOTES � An atom of Nitrogen � 7 protons � Mass of 14 number

TRY IT IN YOUR NOTES � An atom of Nitrogen N 14 � 7 protons � Atomic 14 mass of 7

HOW ELSE DO WE SHOW ISOTOPES? Mass Number Element Name Sodium-23 ** This is what we see on the periodic table

Questions, Comments, Concerns?

� Please take out your whiteboards.

� Write the isotope symbol for the chemical element that has 6 protons and 8 neutrons (write the symbol both ways)

� Write the isotope symbol for the chemical element that has 6 protons and 8 neutrons (write the symbol both ways) Carbon – 14 14 6 C

� Write the isotope symbol for the chemical element that has 17 protons and 16 neutrons

� Write the isotope symbol for the chemical element that has 17 protons and 16 neutrons 33 17 Cl or Chlorine-33

� How many protons are in the isotope 54 Cr? (Chromium-54)

� How many protons are in the isotope 54 Cr? (Chromium-54) � 24 just like CHANGE all Chromium Protons NEVER

TRUE/FALSE � Isotopes of an element always have the same number of neutrons. � A. False � B. True

TRUE/FALSE � Isotopes of an element always have the same number of neutrons. � A. False

TRUE/FALSE � Isotopes of an element always have the same number of protons. � A. False � B. True

TRUE/FALSE � Isotopes of an element always have the same number of protons. � B. True

�Z =9 � A = 29 � Draw the symbol. � How many protons? � How many neutrons?