Warm Up Rubidium has two common isotopes 85

Warm Up: Rubidium has two common isotopes, 85 Rb and 87 Rb. If the abundance of 85 Rb is 72. 2% and the abundance of 87 Rb is 27. 8%, what is the average atomic mass of rubidium? Today: Orbital Diagram and Electron Configuration Notes and Practice Homework: Finish Practice
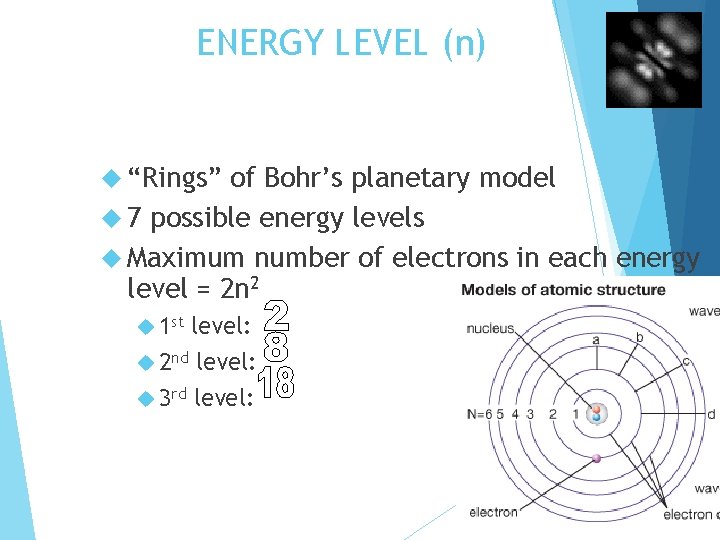
ENERGY LEVEL (n) “Rings” of Bohr’s planetary model 7 possible energy levels Maximum number of electrons in each energy level = 2 n 2 1 st level: 2 nd level: 3 rd level:
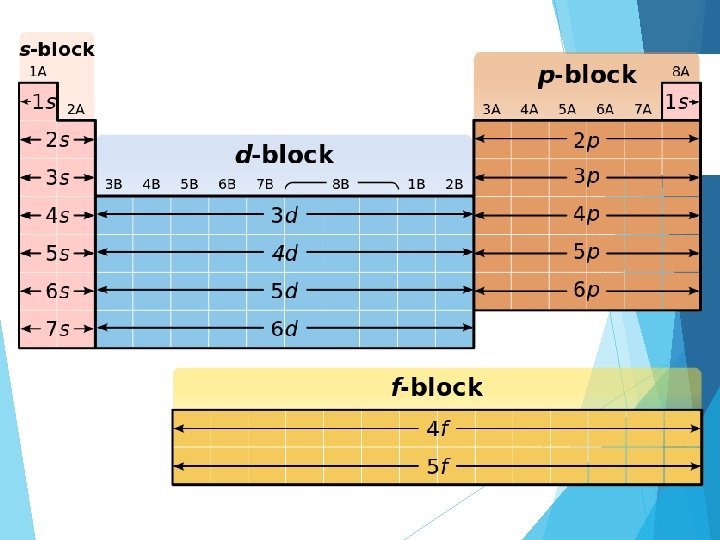
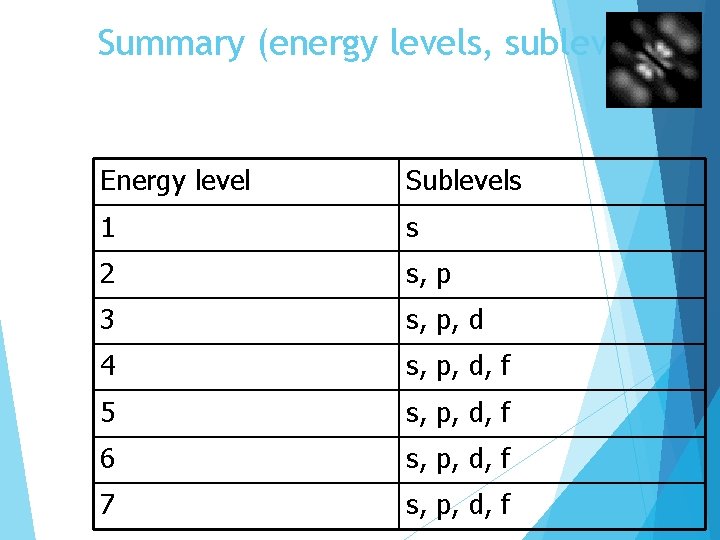
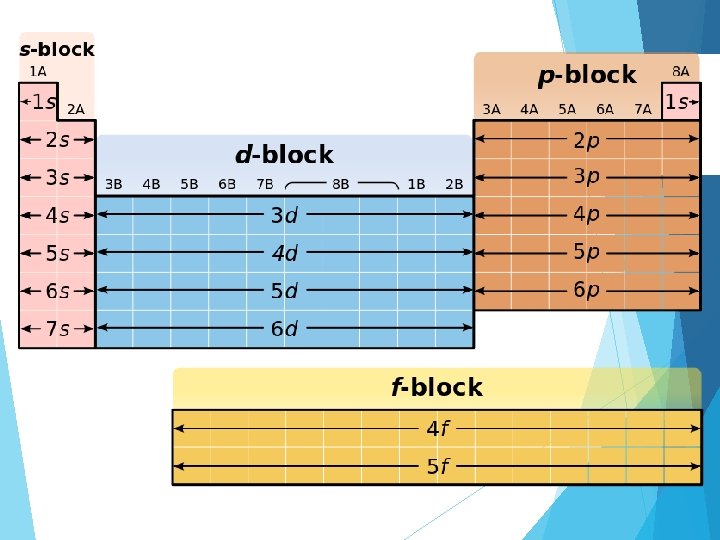
Summary (energy levels, sublevels) Energy level Sublevels 1 s 2 s, p 3 s, p, d 4 s, p, d, f 5 s, p, d, f 6 s, p, d, f 7 s, p, d, f
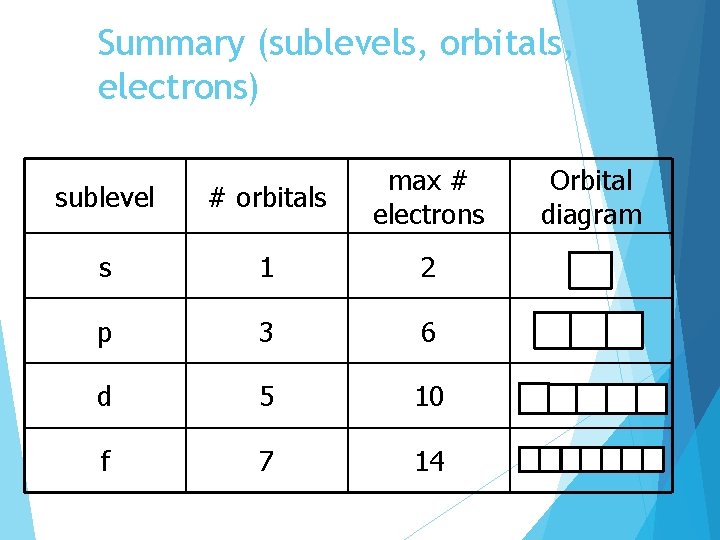
Summary (sublevels, orbitals, electrons) sublevel # orbitals max # electrons s 1 2 p 3 6 d 5 10 f 7 14 Orbital diagram

MAGNETIC SPIN “Spin up” “Spin down”

RULES FOR ELECTRONS An orbital can hold a maximum of TWO electrons Pauli exclusion principle Aufbau principle Hund’s rule

Pauli Exclusion Principle no two electrons can have the same “address” Energy level Sublevel Orbital Spin

Aufbau Principle Electrons will fill the orbitals with the lowest amount of energy before filling in orbitals that have more energy Order is: 1 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5 s, 4 d, 5 p, 6 s, 4 f, 5 d, 6 p, 7 s, 5 f, 6 d, 7 p

Hund’s Rule Electrons will not share an orbital of the same energy if there is an empty orbital with that energy is available
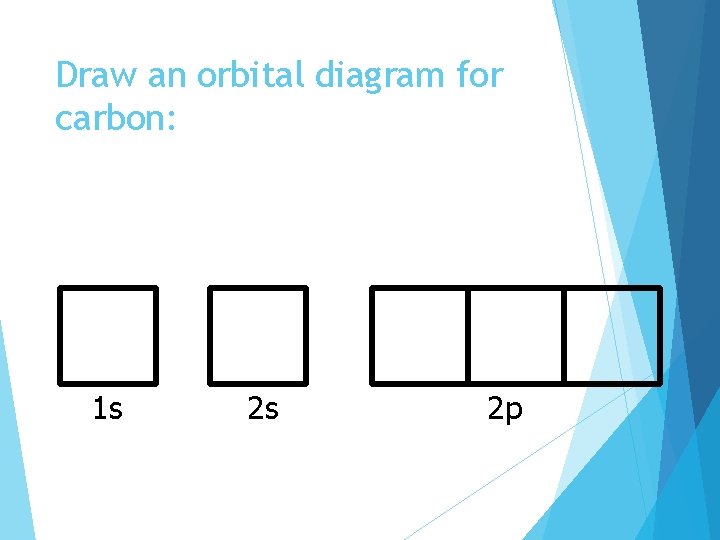
Draw an orbital diagram for carbon: 1 s 2 s 2 p

How to draw orbital diagrams Use periodic table to figure out where to end (energy level and sublevel of last electron) Draw boxes (orbitals) for all energy levels and sublevels up to the ending point 1 box for s, 3 boxes for p, 5 boxes for d, 7 boxes for f Fill boxes with electrons (2 per orbital) with opposite spins Follow Hund’s rule when in the p, d, or f sublevels

Electron configurations Based on orbital diagrams Use numbers and superscripts along with s, p, d and f to show location of electrons in atoms Coefficient = energy level Letter = sublevel Superscript = # of electrons in sublevel
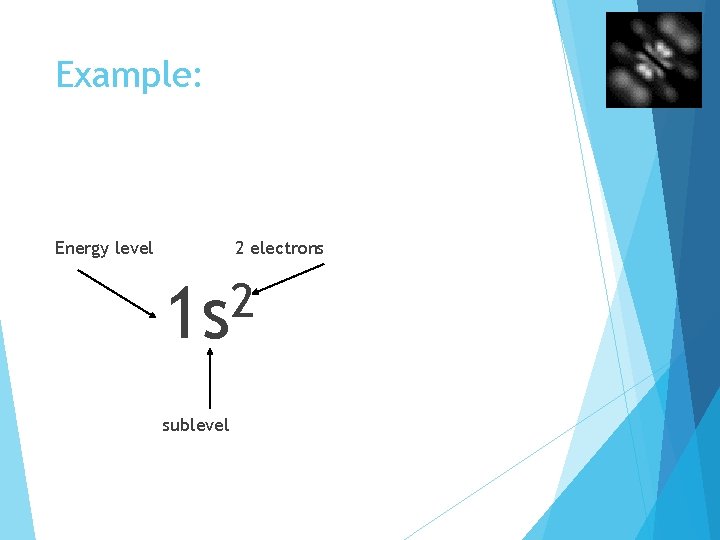
Example: Energy level 2 electrons 2 1 s sublevel
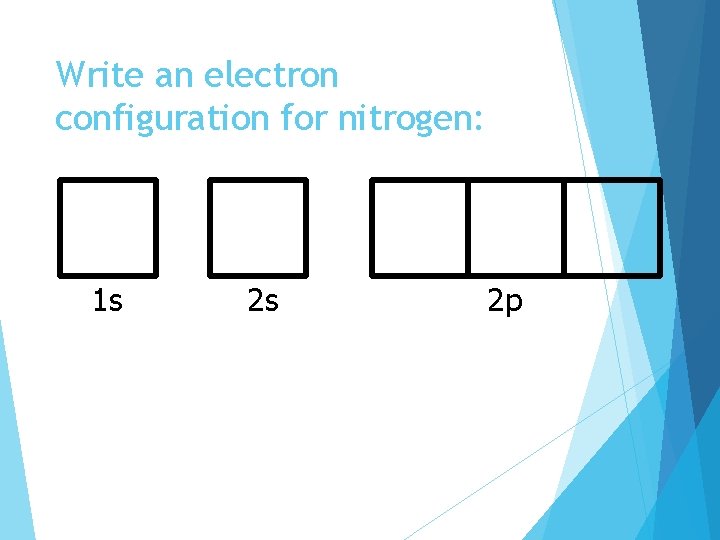
Write an electron configuration for nitrogen: 1 s 2 s 2 p

Practice 1. Draw the orbital diagram for oxygen 1 s 2. 2 s 2 p Write the electron configuration for oxygen 1 s 22 p 4
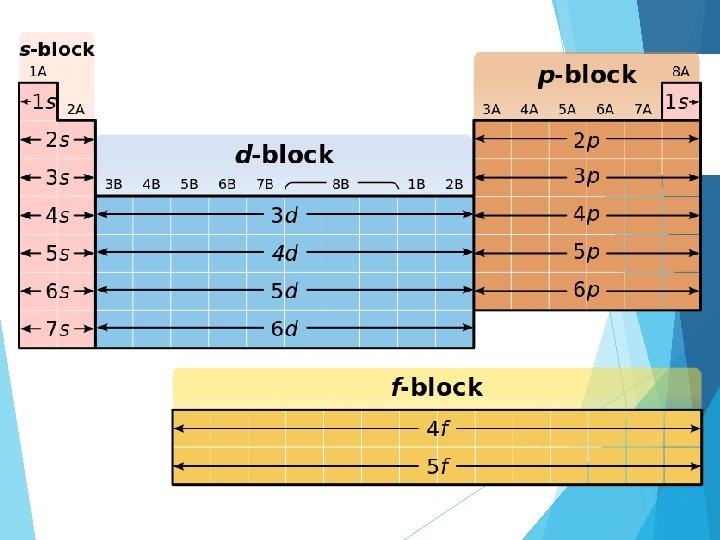

Shortcuts!! Shortcut (shortened) Notation: Use closest Noble Gas before the element For bromine: Closest Noble Gas before the element: Argon: Shortcut Notation: [Ar] 4 s 23 d 104 p 5 b

What is the electron configuration of potassium? 2 2 6 1 A) 1 s 2 s 2 p 3 s 3 p 3 d 2 2 2 1 B) 1 s 2 s 2 p 3 s 3 p 4 s C) 1 s 22 p 63 s 23 p 3 D) 1 s 22 p 63 s 23 p 64 s 1

Each period number in the periodic table corresponds to _______ A) an atomic mass B) an energy level C) an energy sublevel number D) an atomic
- Slides: 23