Isotopes and Average Atomic Mass Subtitle Isotopes Atoms

Isotopes and Average Atomic Mass Subtitle
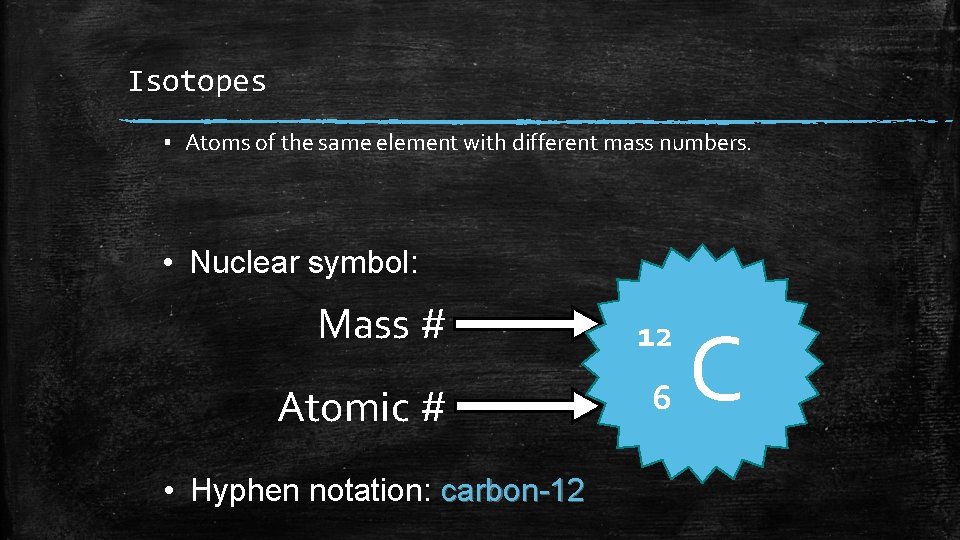
Isotopes ▪ Atoms of the same element with different mass numbers. • Nuclear symbol: Mass # 12 Atomic # 6 • Hyphen notation: carbon-12 C
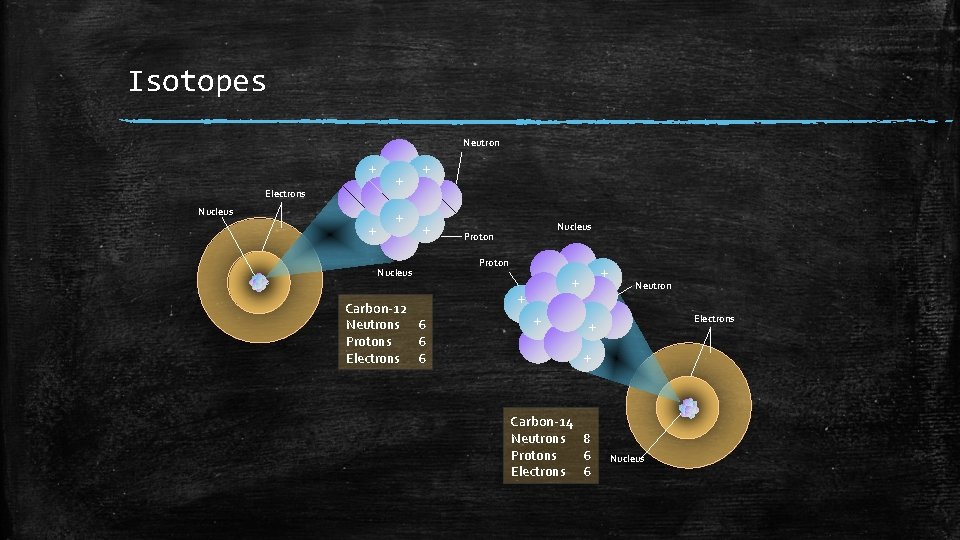
Isotopes Neutron + Electrons Nucleus + + + Nucleus Carbon-12 Neutrons 6 Protons 6 Electrons 6 Nucleus Proton + + Neutron Electrons + + Carbon-14 Neutrons 8 Protons 6 Electrons 6 Nucleus
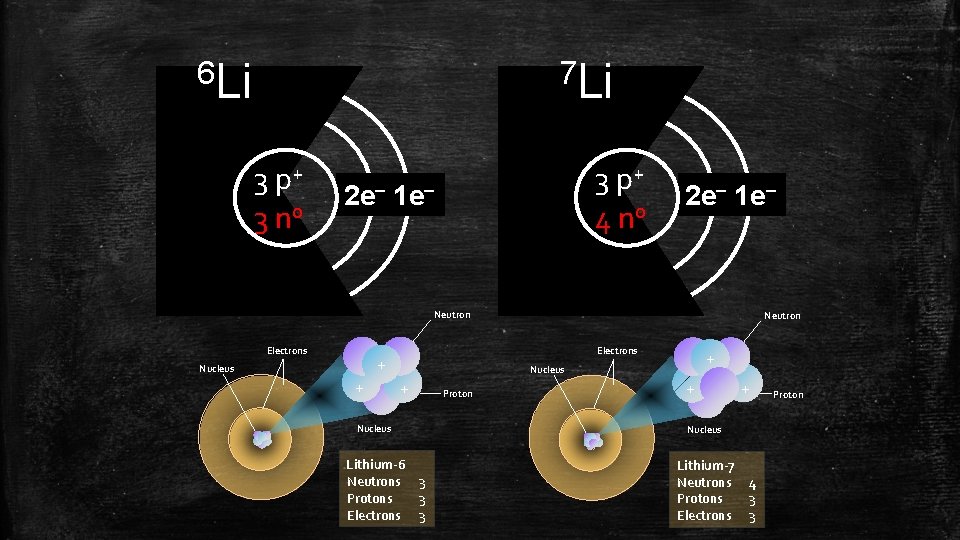
6 Li 7 Li 3 p+ 3 n 0 3 p+ 4 n 0 2 e– 1 e– Neutron Electrons + Nucleus + Neutron + Nucleus Lithium-6 Neutrons 3 Protons 3 Electrons 3 Proton + + Nucleus Lithium-7 Neutrons Protons Electrons 4 3 3 Proton

Cl Isotopes 37 ▪ Chlorine-37 – atomic #: 17 – mass #: 37 – # of protons: 17 37 – # of electrons: 17 – # of neutrons: 20 17 Cl 17

Average Atomic Mass ▪ weighted average of all isotopes ▪ on the Periodic Table ▪ round to 2 decimal places Avg. (mass)(%) + (mass)(%) Atomic = 100 Mass Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Average Atomic Mass ▪ EX: Calculate the avg. atomic mass of oxygen if its abundance in nature is 99. 76% 16 O, 0. 04% 17 O, and 0. 20% 18 O. Avg. (16)(99. 76) + (17)(0. 04) + (18)(0. 20) Atomic = 100 Mass Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem = 16. 00 amu

Isotopes vs. Ions ▪ Isotopes vary in the number of neutrons so mass is affected ▪ Ions vary in the number of electrons so charge is affected
- Slides: 9