Helsinki3 Direct Estimate of Conjugation Hyperconjugation and Aromaticity







- Slides: 50

Helsinki-3 Direct Estimate of Conjugation, Hyperconjugation and Aromaticity With an Energy Decomposition Analysis Gernot Frenking Fachbereich Chemie, Philipps-Universität Marburg

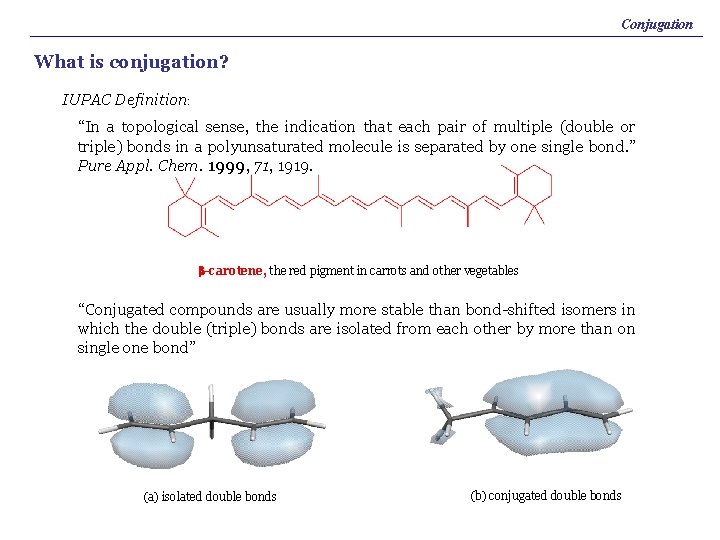
Conjugation What is conjugation? IUPAC Definition: “In a topological sense, the indication that each pair of multiple (double or triple) bonds in a polyunsaturated molecule is separated by one single bond. ” Pure Appl. Chem. 1999, 71, 1919. b-carotene, the red pigment in carrots and other vegetables “Conjugated compounds are usually more stable than bond-shifted isomers in which the double (triple) bonds are isolated from each other by more than on single one bond” (a) isolated double bonds (b) conjugated double bonds

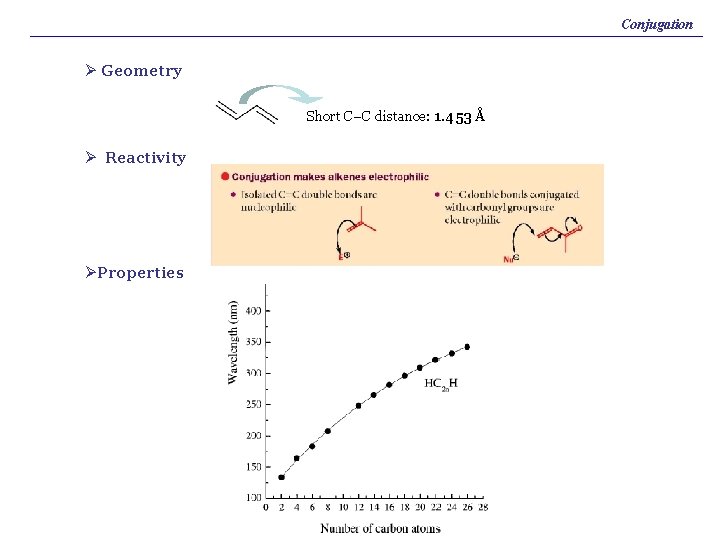
Conjugation Ø Geometry Short C–C distance: 1. 453 Å Ø Reactivity ØProperties


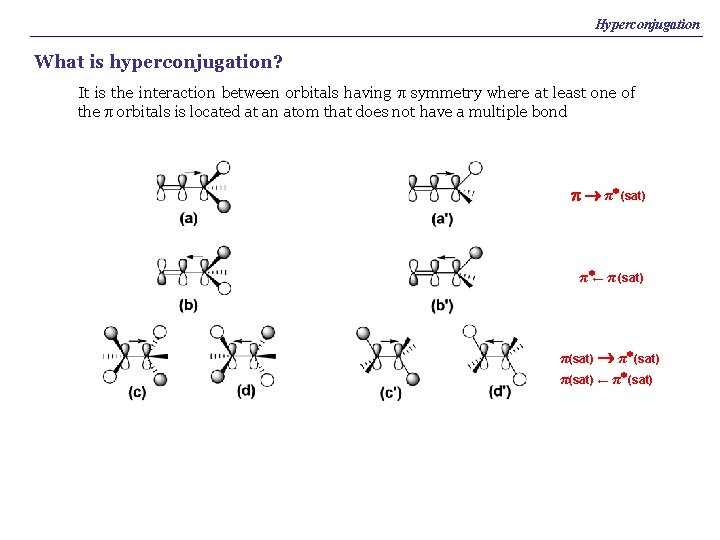
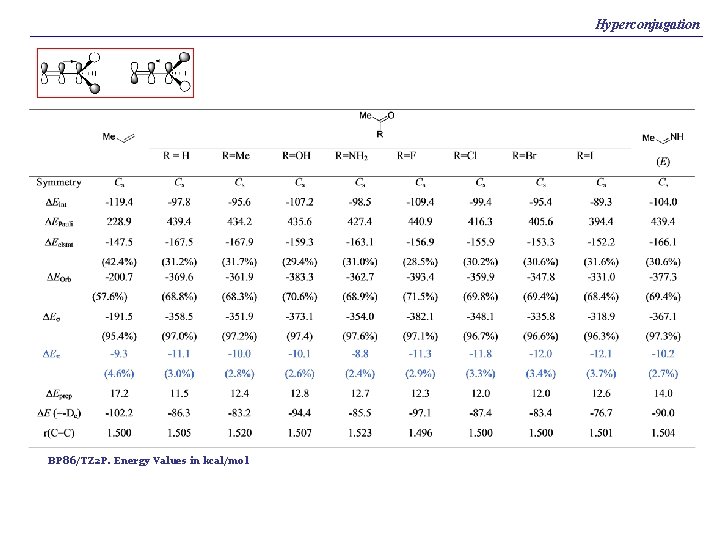
Hyperconjugation What is hyperconjugation? It is the interaction between orbitals having p symmetry where at least one of the p orbitals is located at an atom that does not have a multiple bond p π* (sat) π*← π (sat) π(sat) π* (sat) π(sat) ← π* (sat)


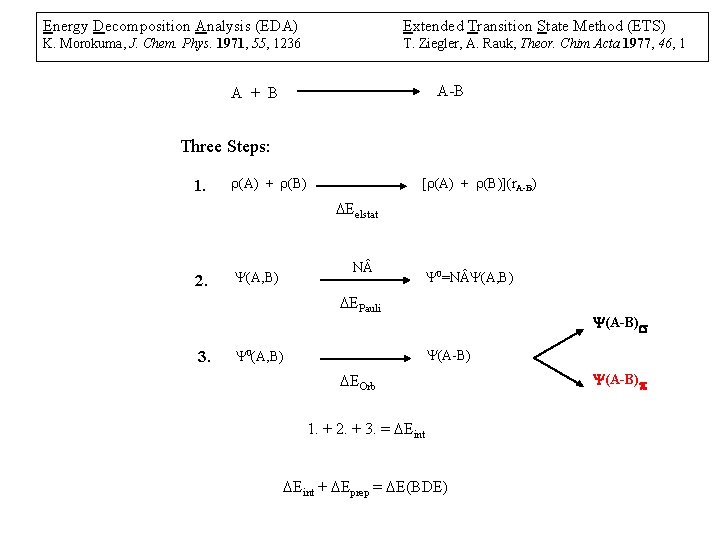
Energy Decomposition Analysis (EDA) Extended Transition State Method (ETS) K. Morokuma, J. Chem. Phys. 1971, 55, 1236 T. Ziegler, A. Rauk, Theor. Chim Acta 1977, 46, 1 A-B A + B Three Steps: 1. r(A) + r(B) [r(A) + r(B)](r. A-B) DEelstat 2. N (A, B) 0=N (A, B) DEPauli 3. (A-B)s (A-B) 0(A, B) DEOrb 1. + 2. + 3. = DEint + DEprep = DE(BDE) (A-B)p

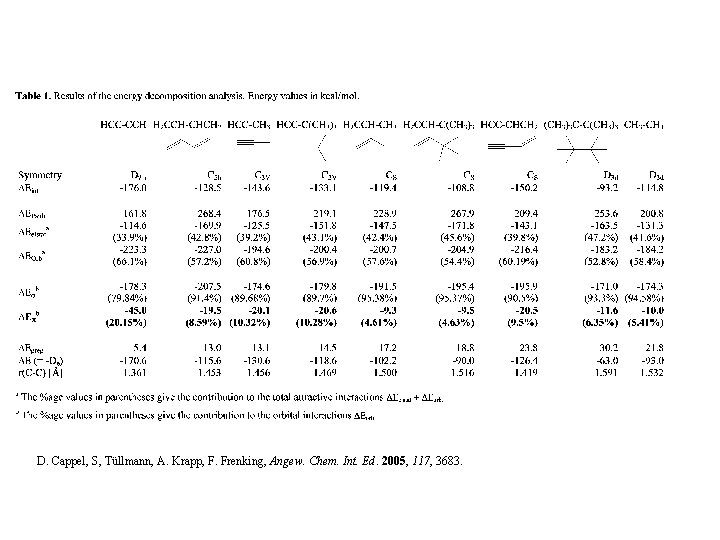
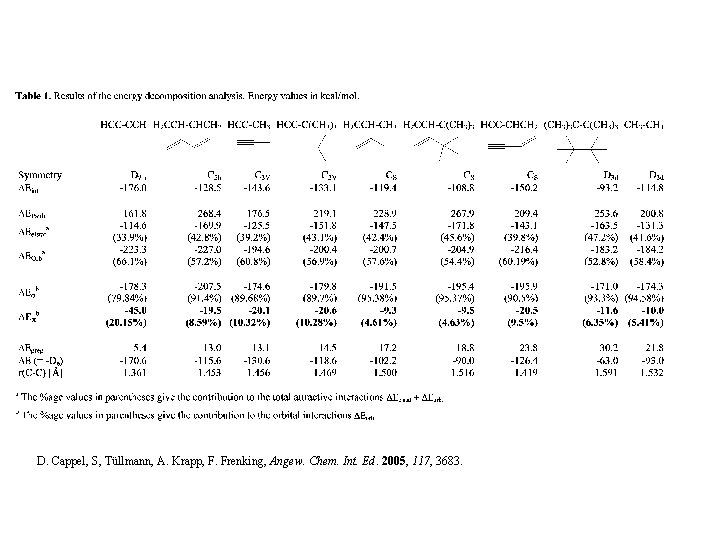
D. Cappel, S, Tüllmann, A. Krapp, F. Frenking, Angew. Chem. Int. Ed. 2005, 117, 3683.

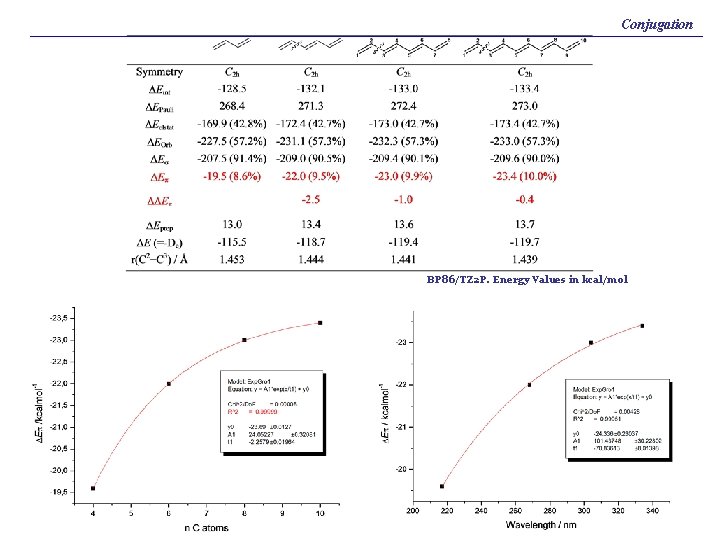
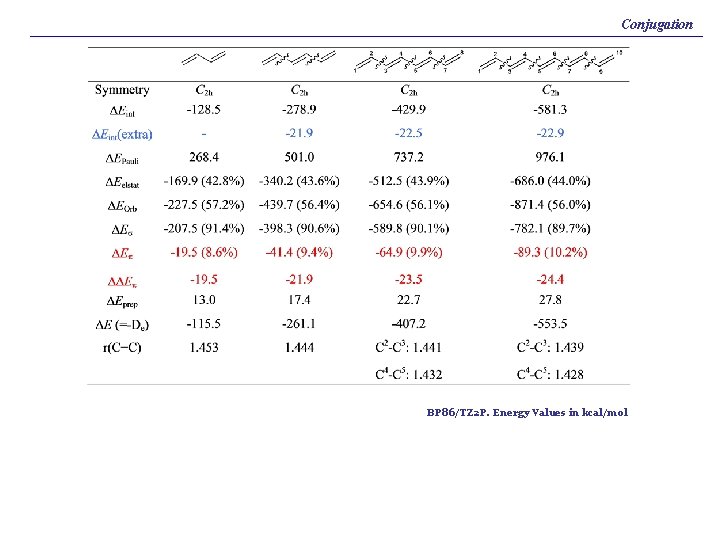
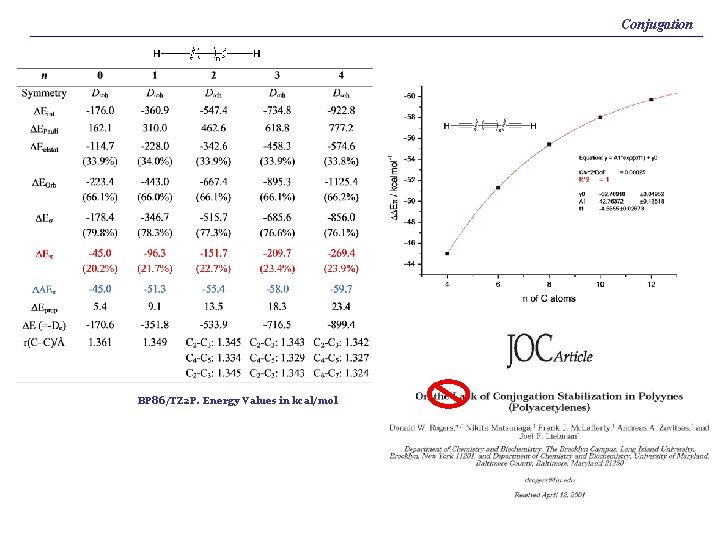
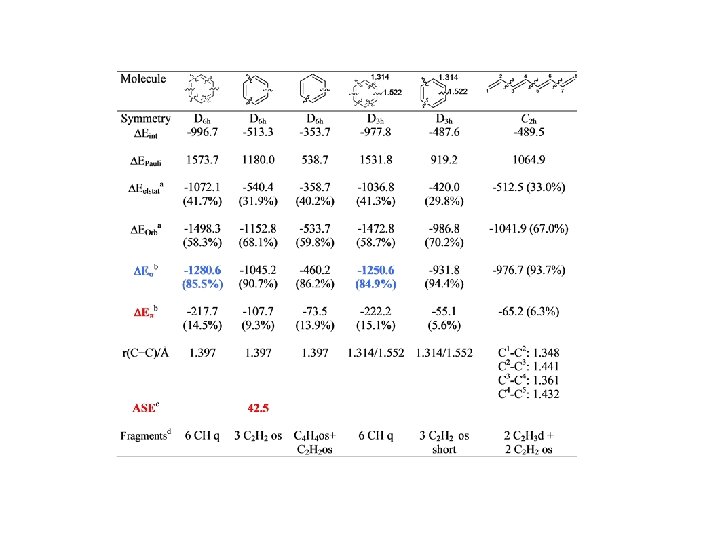
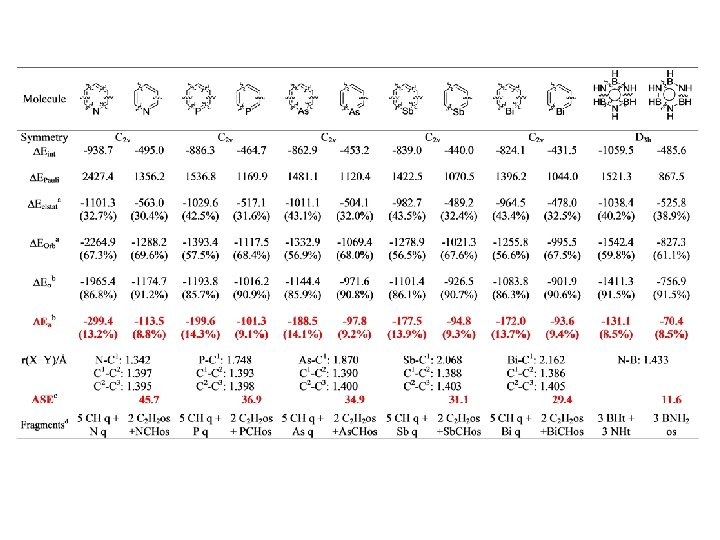
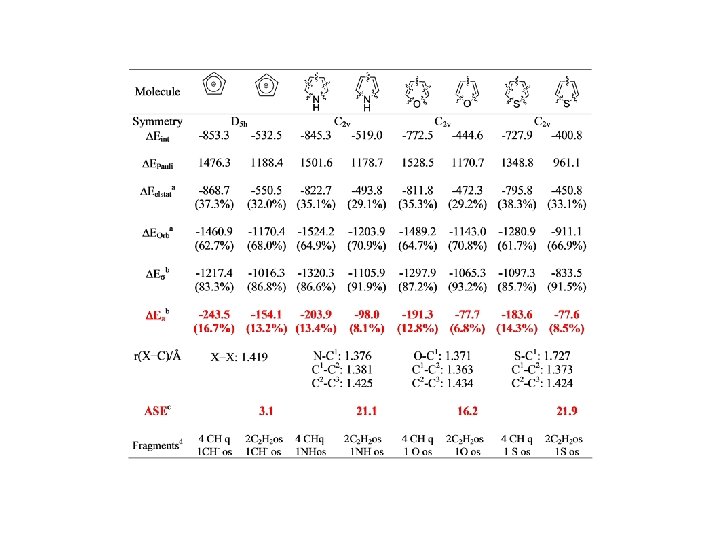
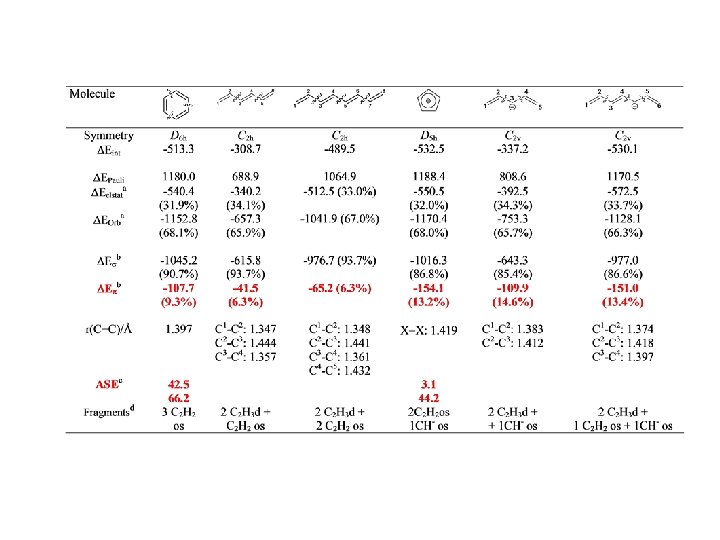
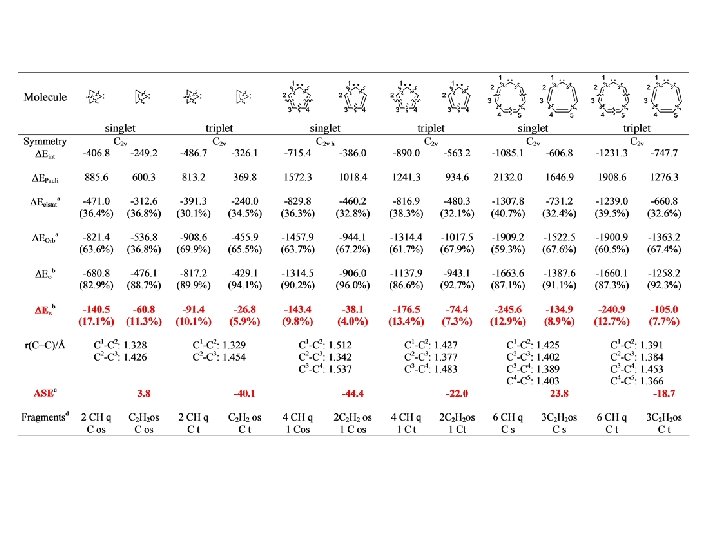
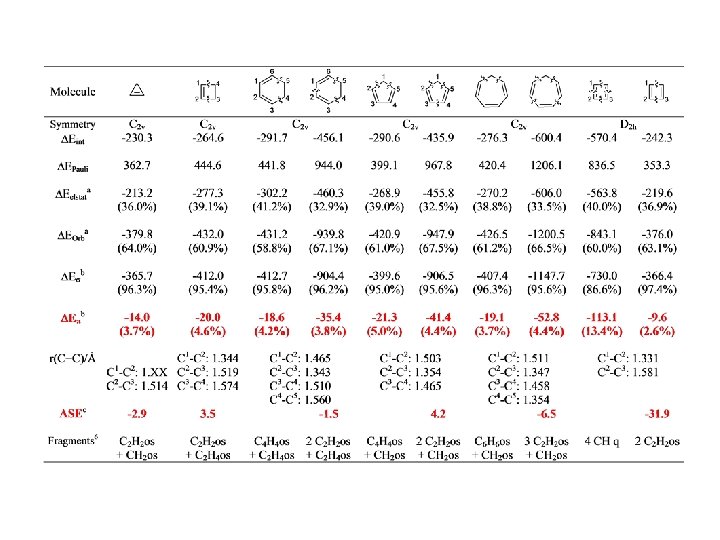
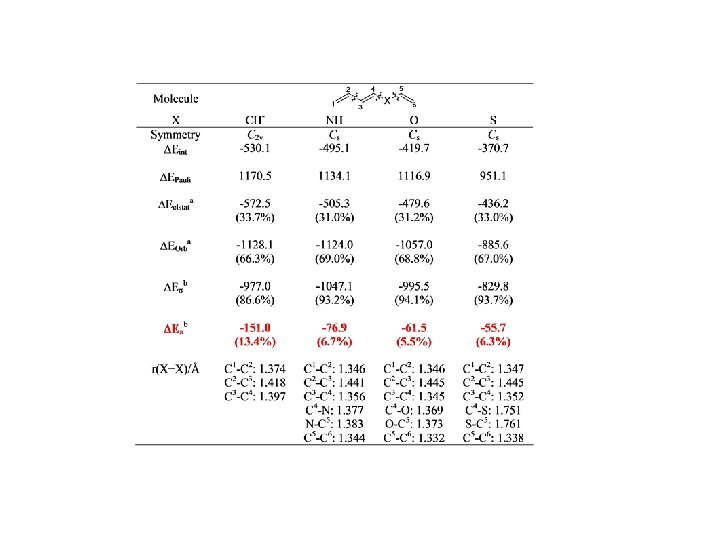
Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

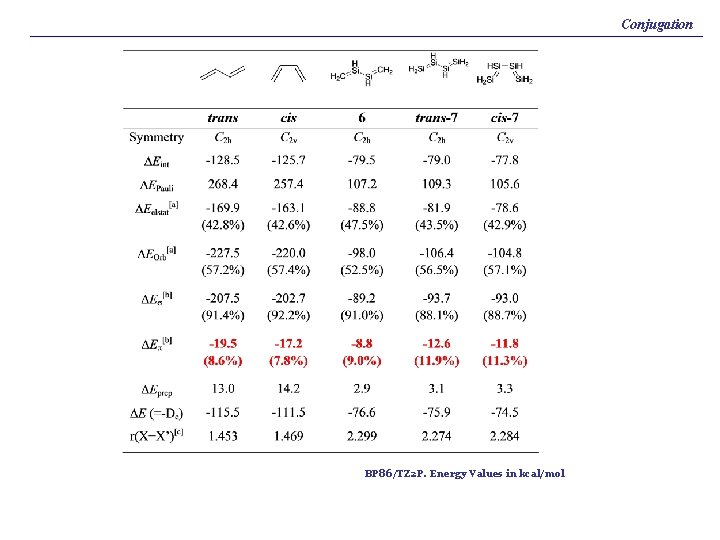
Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

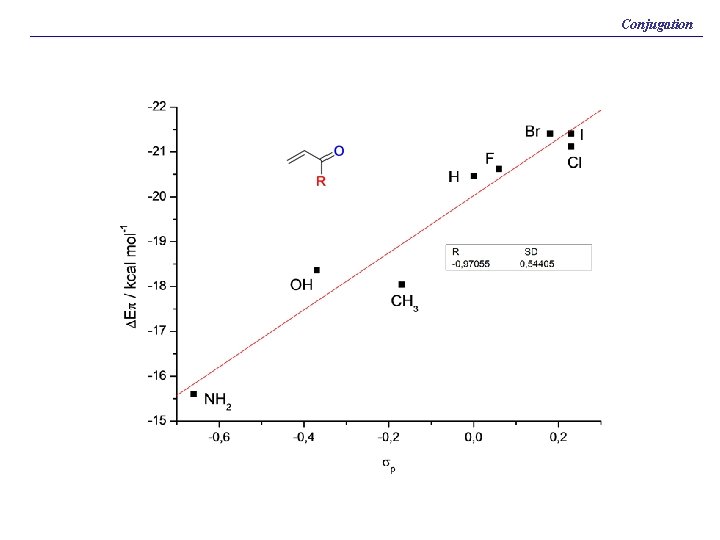
Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol

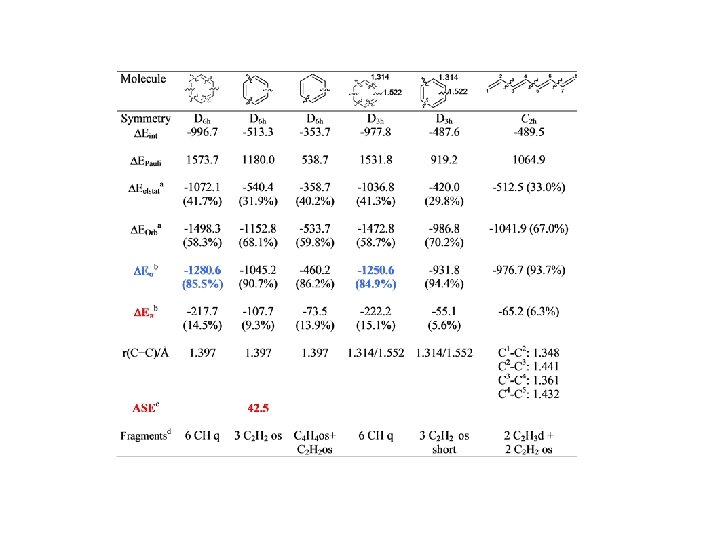
Conjugation

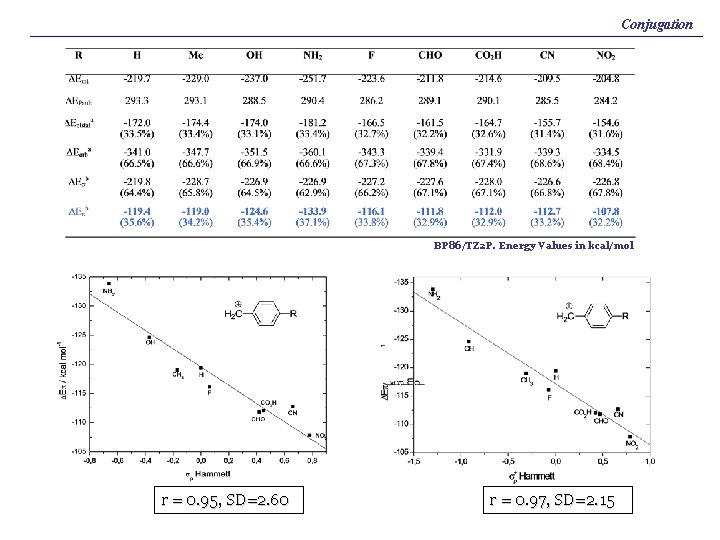
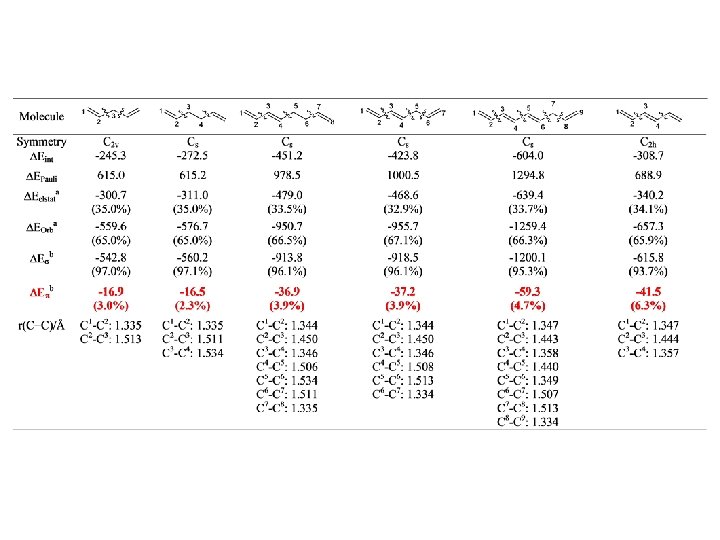
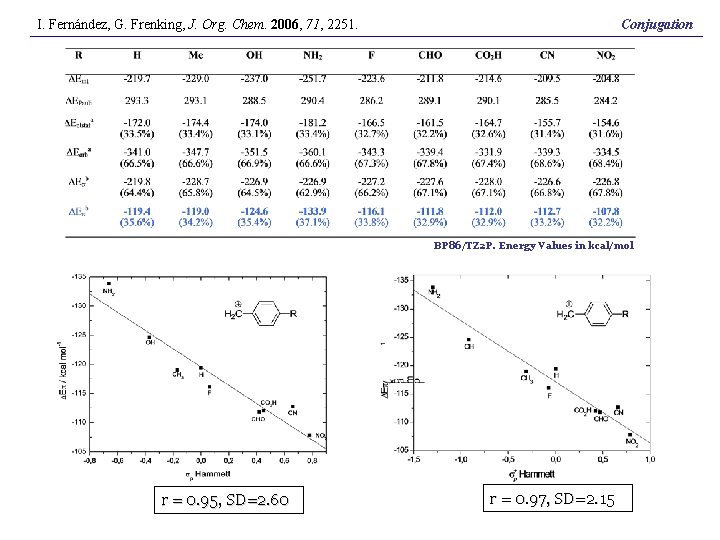
Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol r = 0. 95, SD=2. 60 r = 0. 97, SD=2. 15

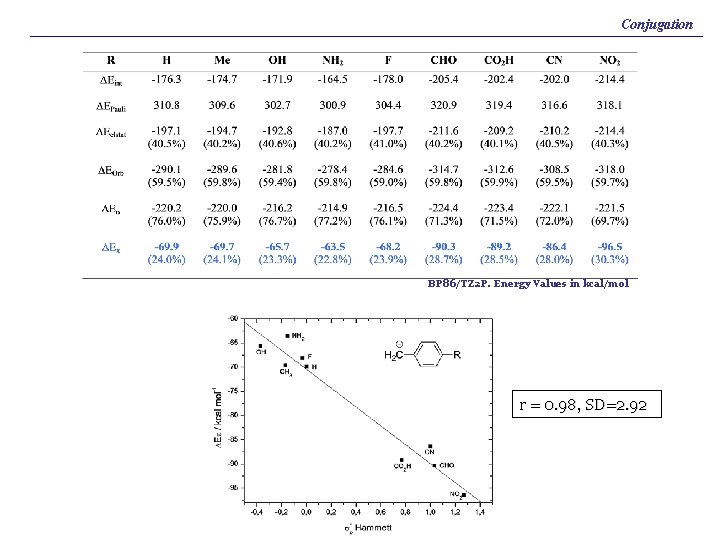
Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol r = 0. 98, SD=2. 92

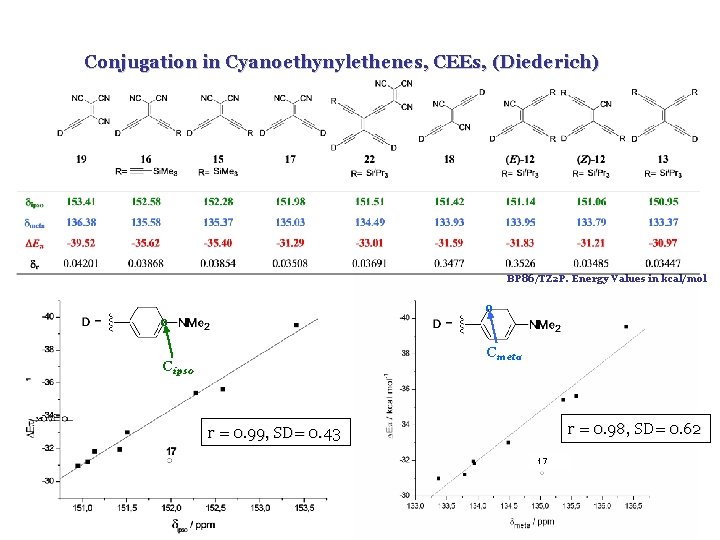
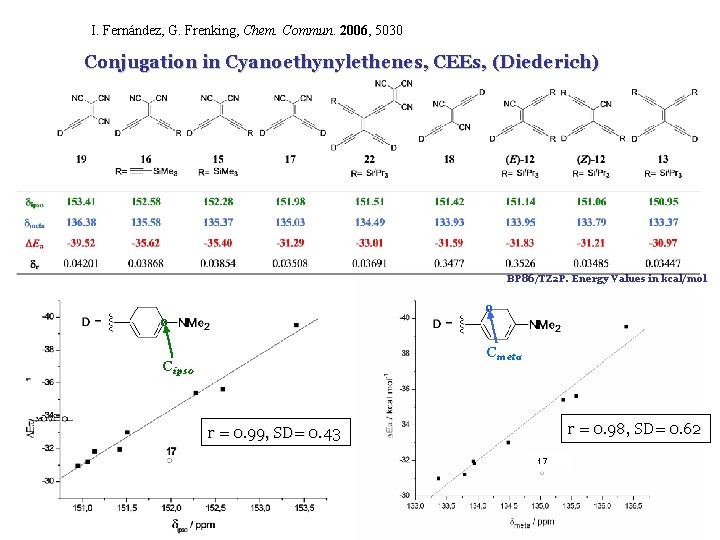
Conjugation in Cyanoethynylethenes, CEEs, (Diederich) BP 86/TZ 2 P. Energy Values in kcal/mol o o Cmeta Cipso r = 0. 98, SD= 0. 62 r = 0. 99, SD= 0. 43 17

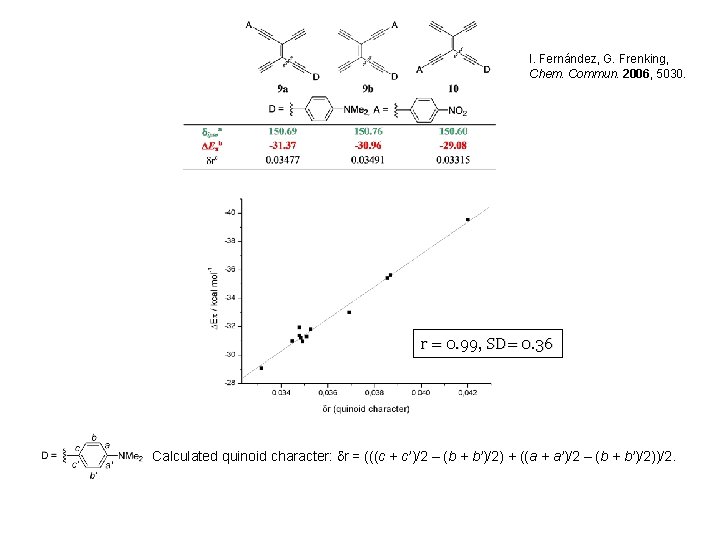
I. Fernández, G. Frenking, Chem. Commun. 2006, 5030. r = 0. 99, SD= 0. 36 Calculated quinoid character: dr = (((c + c’)/2 – (b + b’)/2) + ((a + a’)/2 – (b + b’)/2))/2.

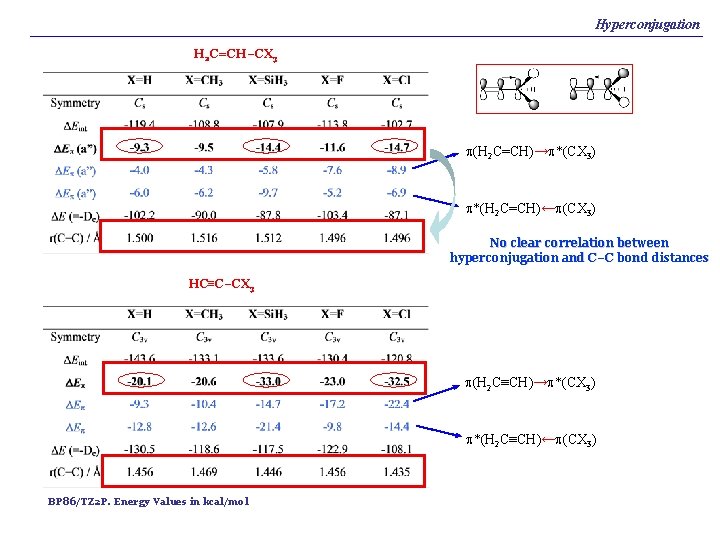
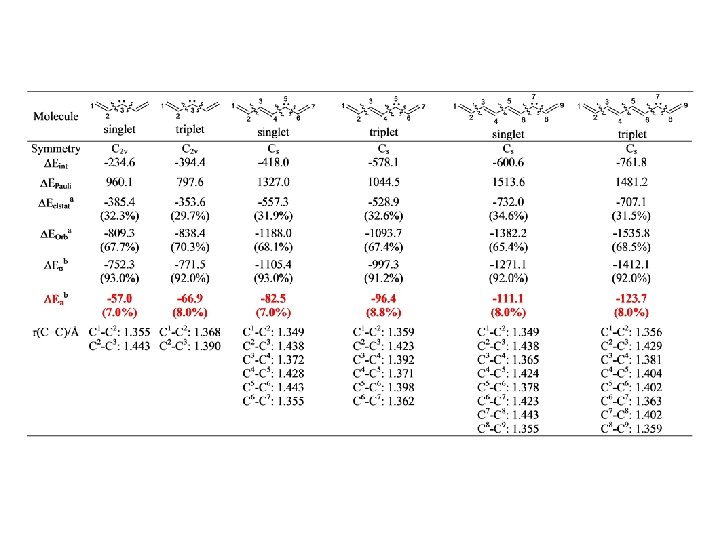
Hyperconjugation H 2 C=CH–CX 3 p(H 2 C=CH)→p*(CX 3) p*(H 2 C=CH)←p(CX 3) No clear correlation between hyperconjugation and C–C bond distances HC≡C–CX 3 p(H 2 C≡CH)→p*(CX 3) p*(H 2 C≡CH)←p(CX 3) BP 86/TZ 2 P. Energy Values in kcal/mol

Hyperconjugation BP 86/TZ 2 P. Energy Values in kcal/mol

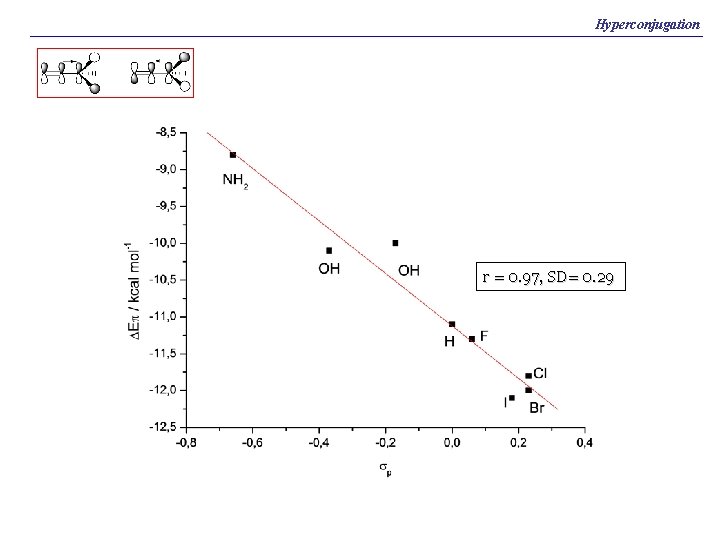
Hyperconjugation r = 0. 97, SD= 0. 29

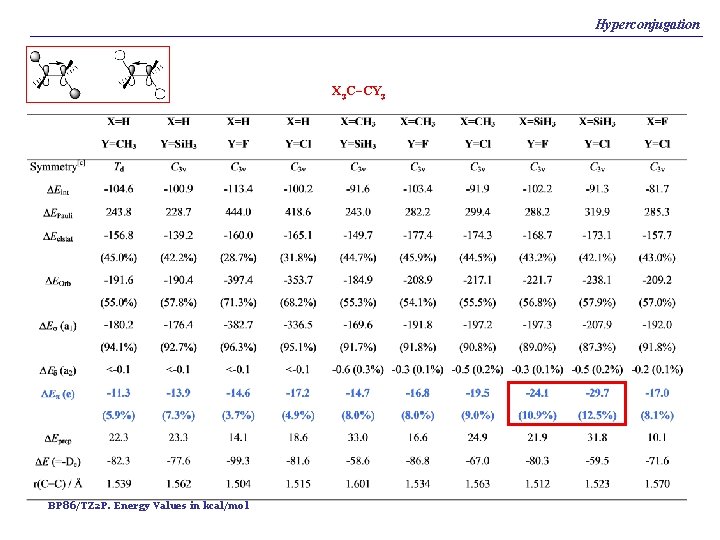
Hyperconjugation X 3 C–CY 3 BP 86/TZ 2 P. Energy Values in kcal/mol

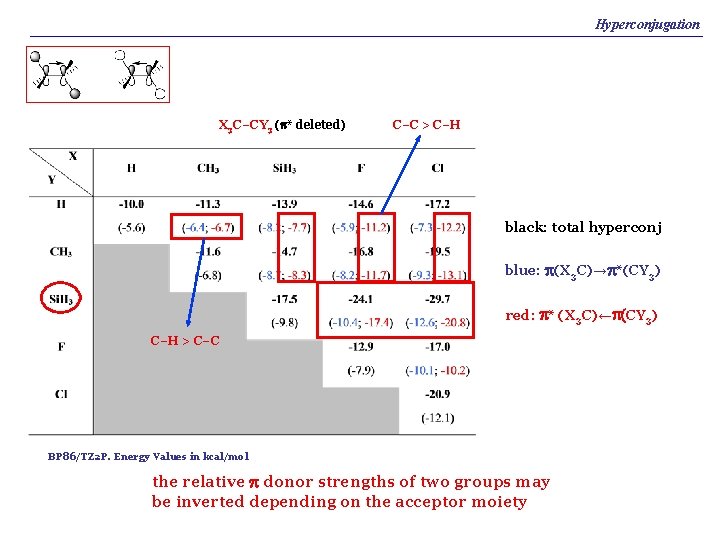
Hyperconjugation X 3 C–CY 3 (p* deleted) C–C > C–H black: total hyperconj blue: p(X 3 C)→p*(CY 3) red: p* (X 3 C)←p(CY 3) C–H > C–C BP 86/TZ 2 P. Energy Values in kcal/mol the relative p donor strengths of two groups may be inverted depending on the acceptor moiety







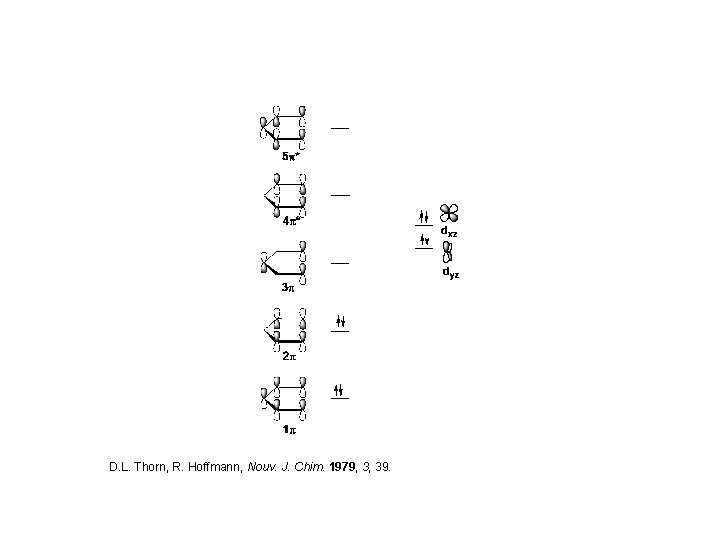
D. L. Thorn, R. Hoffmann, Nouv. J. Chim. 1979, 3, 39.


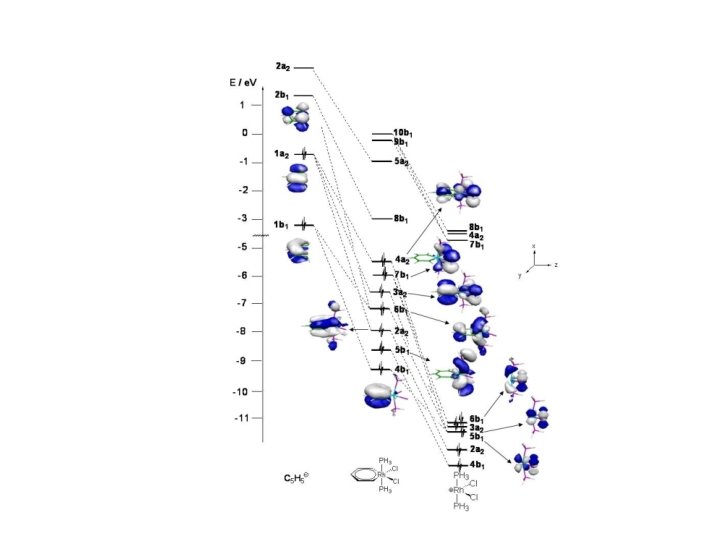
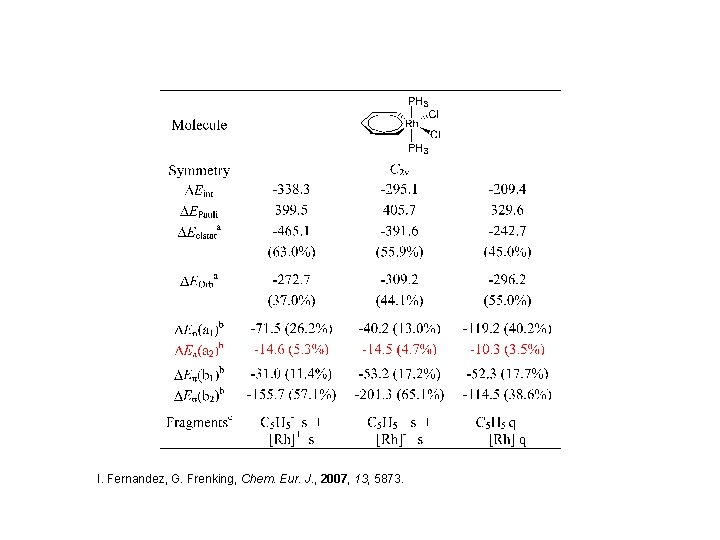
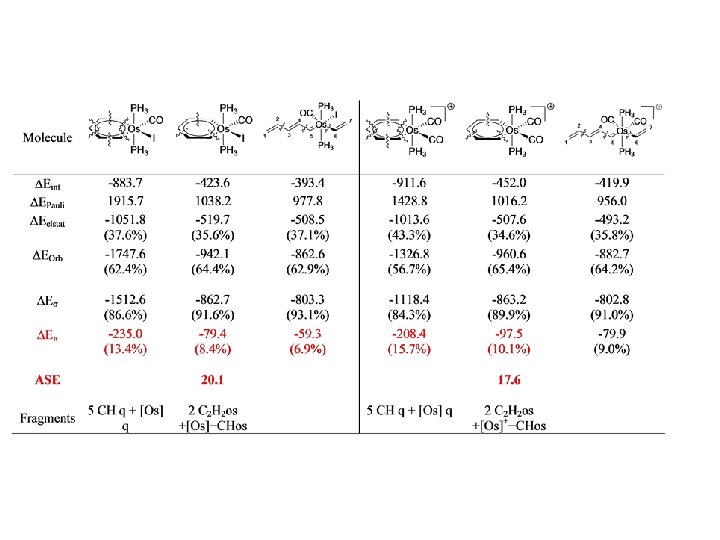
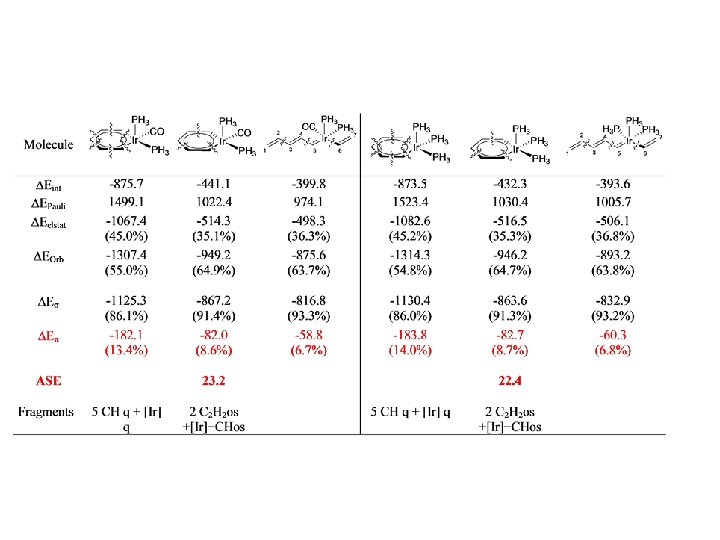
I. Fernandez, G. Frenking, Chem. Eur. J. , 2007, 13, 5873.



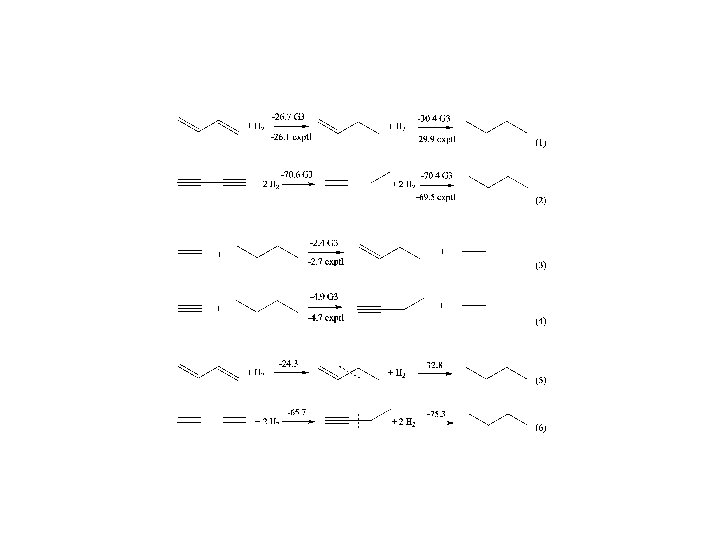
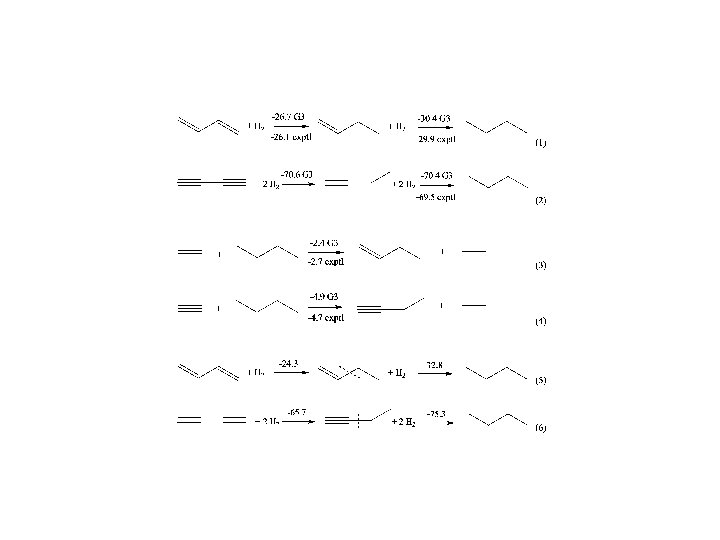
Conjugation and Hyperconjugation CONCLUSIONS Ø The calculated DEp values taken from the EDA can be used to estimate the strength of the relative contributions of p interactions that come from conjugation/hyperconjugation Ø The EDA method has a predictive value. The trend of the calculated DEp values is in very good agreement with NMR chemical shifts and experimentally derived Hammett constants. Ø The comparison of DEp values of cyclic system with a suitable reference compound makes it possible to estimate the stabilization due to aromaticity. Ø Bond energies and lengths should not be used as indicators of the strength of hyperconjugation because the effect of s interactions and electrostatic forces may compensate for the hyperconjugative effect.








D. Cappel, S, Tüllmann, A. Krapp, F. Frenking, Angew. Chem. Int. Ed. 2005, 117, 3683.

I. Fernández, G. Frenking, J. Org. Chem. 2006, 71, 2251. Conjugation BP 86/TZ 2 P. Energy Values in kcal/mol r = 0. 95, SD=2. 60 r = 0. 97, SD=2. 15

I. Fernández, G. Frenking, Chem. Commun. 2006, 5030 Conjugation in Cyanoethynylethenes, CEEs, (Diederich) BP 86/TZ 2 P. Energy Values in kcal/mol o o Cmeta Cipso r = 0. 98, SD= 0. 62 r = 0. 99, SD= 0. 43 17


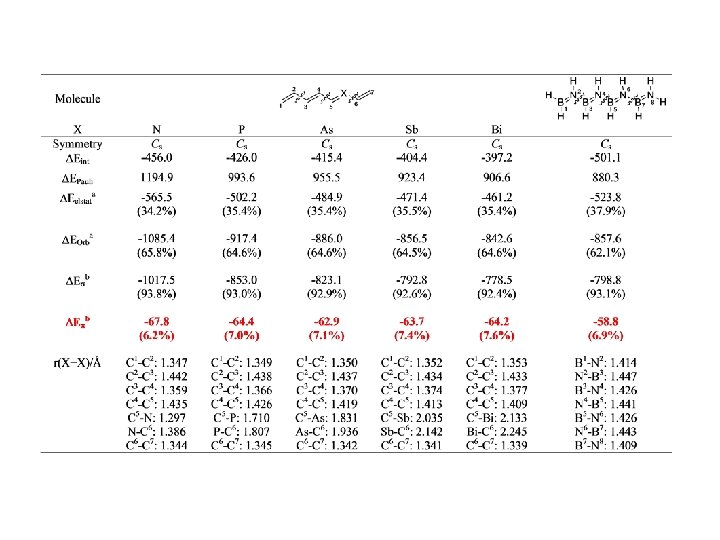
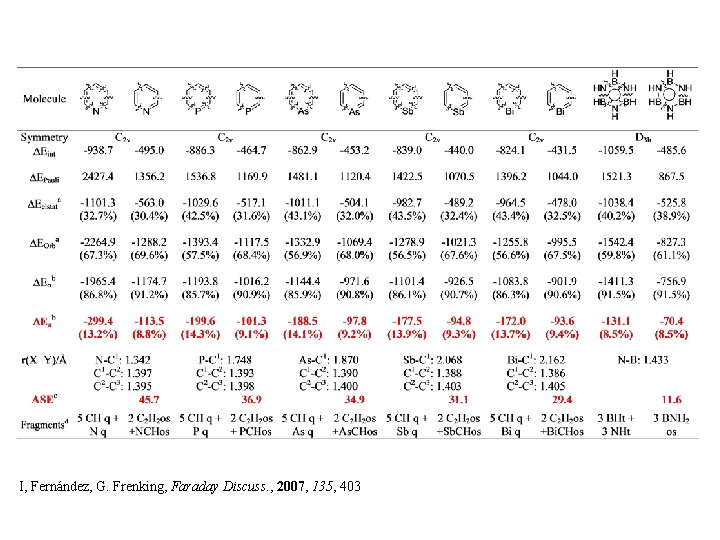
I, Fernández, G. Frenking, Faraday Discuss. , 2007, 135, 403

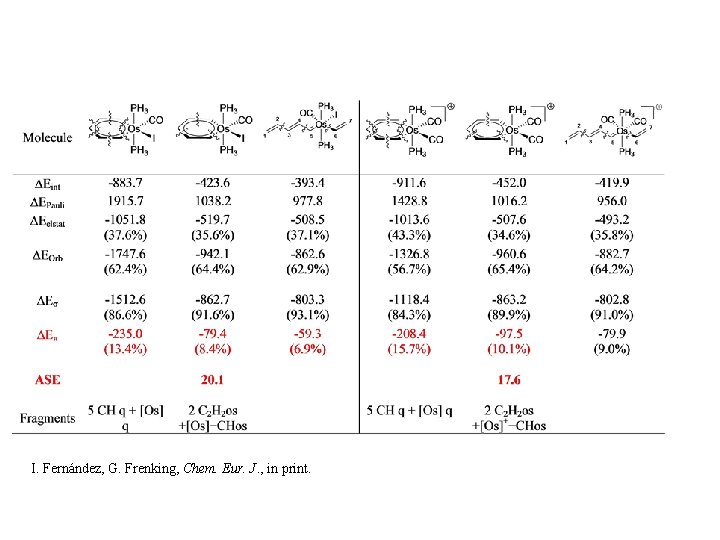
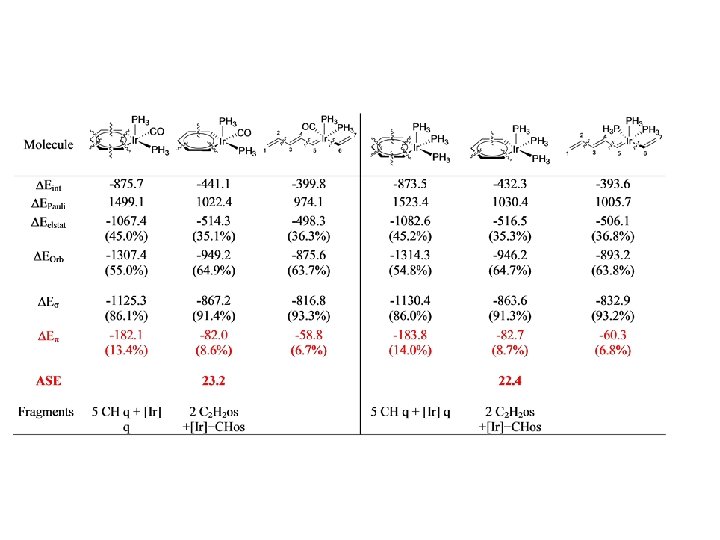
I. Fernández, G. Frenking, Chem. Eur. J. , in print.
