Empirical and Molecular Formulas Topic 20 Empirical and









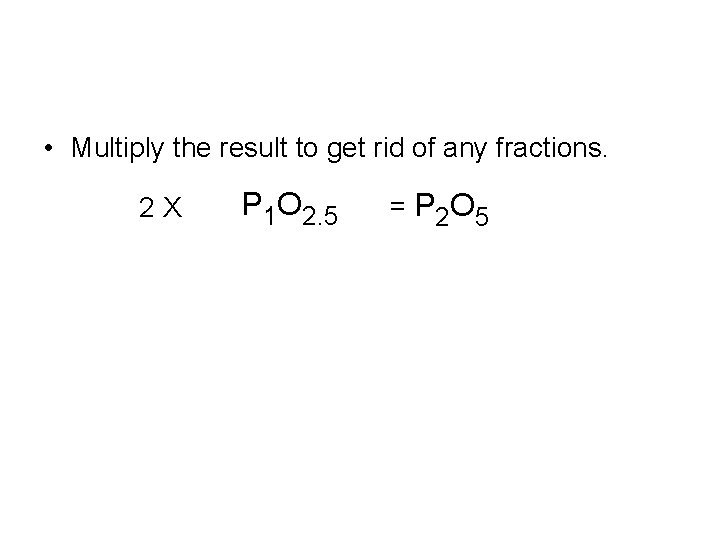




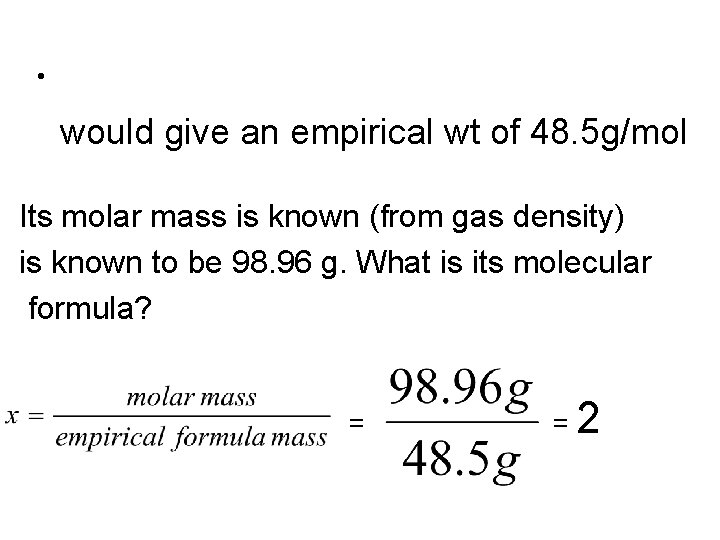
- Slides: 19

Empirical and Molecular Formulas Topic #20

Empirical and Molecular Formulas • Empirical --The lowest whole number ratio of elements in a compound. • Molecular -- the actual ratio of elements in a compound • Sometimes the two can be the same. • CH 2 empirical formula • C 2 H 4 molecular formula • C 3 H 6 molecular formula • H 2 O both

Calculating Empirical • Just find the lowest whole number ratio • C 6 H 12 O 6 • CH 4 N

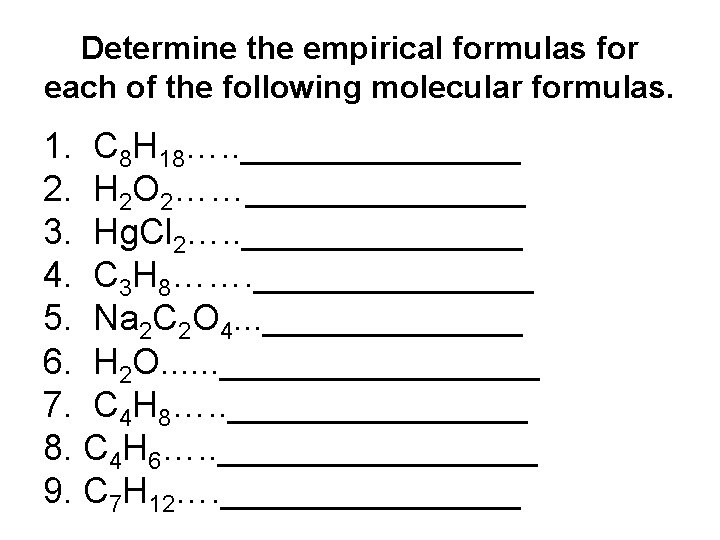
Determine the empirical formulas for each of the following molecular formulas. 1. C 8 H 18…. . _______ 2. H 2 O 2……_______ 3. Hg. Cl 2…. . _______ 4. C 3 H 8……. _______ 5. Na 2 C 2 O 4. . . _______ 6. H 2 O. . . ________ 7. C 4 H 8…. . ________ 8. C 4 H 6…. . ________ 9. C 7 H 12…. ________

You can calculate empirical formulas if you know the percent composition………….

Calculating Empirical • • • Pretend that you have a 100 gram sample of the compound. That is, change the % to grams. Convert the grams to mols for each element. Write the number of mols as a subscript in a chemical formula. Divide each number by the least number. Multiply the result to get rid of any fractions.

Example • Calculate the empirical formula of a compound composed of 38. 67 % C, 16. 22 % H, and 45. 11 %N. • Assume 100 g so • 38. 67 g C 1 mol C = 3. 220 mole C 12. 01 g. C • 16. 22 g H 1 mol H = 16. 09 mole H 1. 01 g. H • 45. 11 g N 1 mol N = 3. 219 mole N 14. 01 g. N

• 3. 220 mole C • 16. 09 mole H • 3. 219 mole N • C 3. 22 H 16. 09 N 3. 219 If we divide all of these by the smallest one It will give us the empirical formula

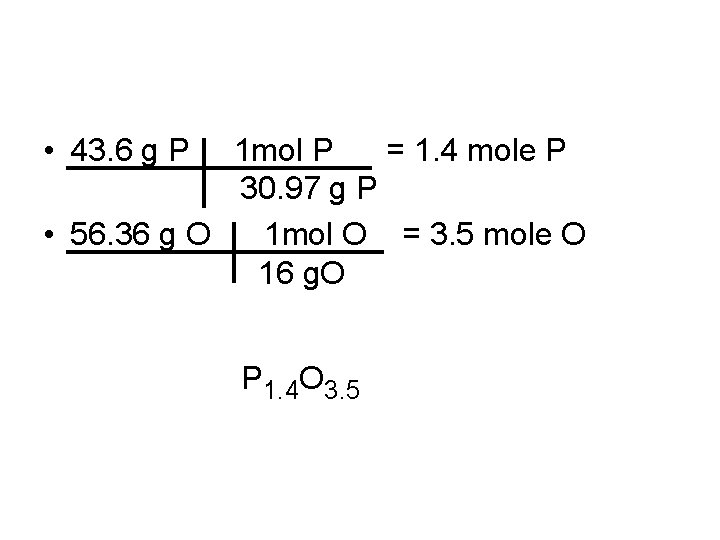
Example • The ratio is 3. 220 mol C = 1 mol C 3. 219 mol. N • The ratio is 16. 09 mol H = 5 mol H 3. 219 mol. N • C 1 H 5 N 1 is the empirical formula • A compound is 43. 64 % P and 56. 36 % O. What is the empirical formula?

• 43. 6 g P 1 mol P = 1. 4 mole P 30. 97 g P • 56. 36 g O 1 mol O = 3. 5 mole O 16 g. O P 1. 4 O 3. 5

Divide both by the lowest one P 1. 4 O 3. 5 • The ratio is 3. 5 mol O = 2. 5 mol O 1. 4 mol P P 1 O 2. 5
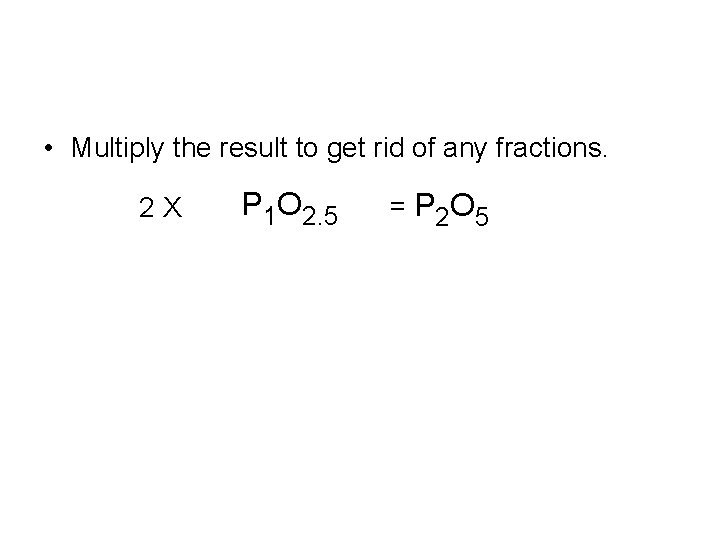
• Multiply the result to get rid of any fractions. 2 X P 1 O 2. 5 = P 2 O 5

• Caffeine is 49. 48% C, 5. 15% H, 28. 87% N and 16. 49% O. What is its empirical formula? Empirical Mass=

Empirical to molecular • Since the empirical formula is the lowest ratio the actual molecule would weigh more. • By a whole number multiple. • Divide the actual molar mass by the mass of one mole of the empirical formula.

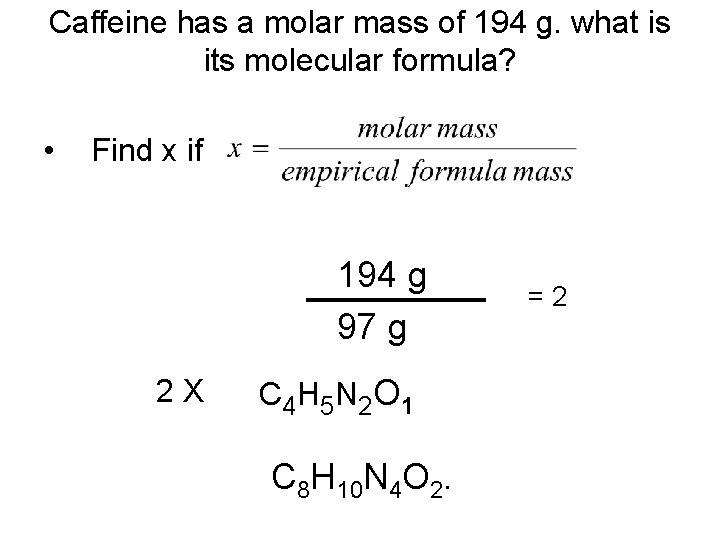
Caffeine has a molar mass of 194 g. what is its molecular formula? • Find x if 194 g 97 g 2 X C 4 H 5 N 2 O 1 C 8 H 10 N 4 O 2. =2

Example • A compound is known to be composed of 71. 65 % Cl, 24. 27% C and 4. 07% H. Its molar mass is known (from gas density) is known to be 98. 96 g. What is its molecular formula?

Example • A compound is known to be composed of 71. 65 % Cl, 24. 27% C and 4. 07% H. Its molar mass is known (from gas density) is known to be 98. 96 g. What is its molecular formula?
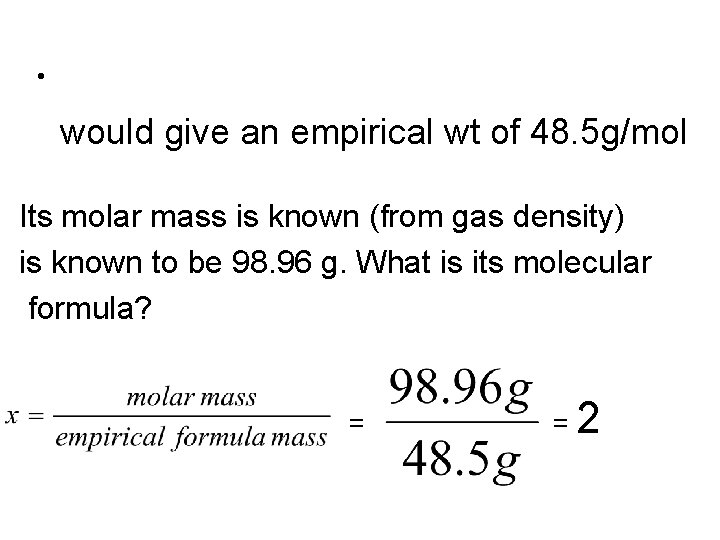
• would give an empirical wt of 48. 5 g/mol Its molar mass is known (from gas density) is known to be 98. 96 g. What is its molecular formula? = =2
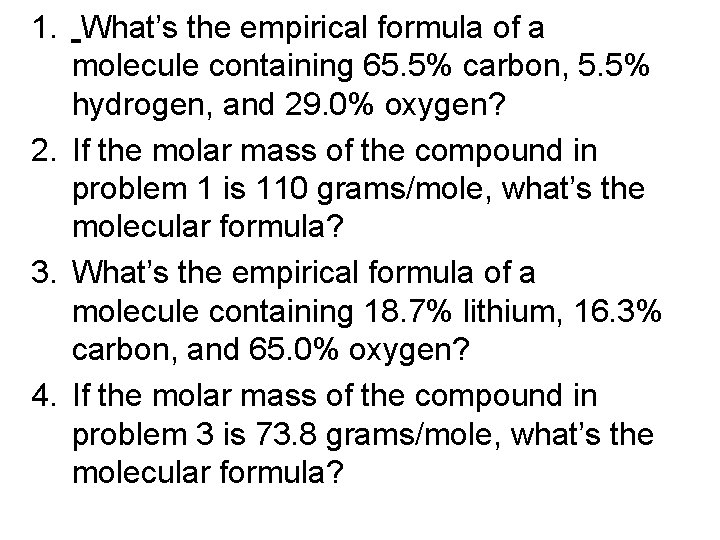
1. What’s the empirical formula of a molecule containing 65. 5% carbon, 5. 5% hydrogen, and 29. 0% oxygen? 2. If the molar mass of the compound in problem 1 is 110 grams/mole, what’s the molecular formula? 3. What’s the empirical formula of a molecule containing 18. 7% lithium, 16. 3% carbon, and 65. 0% oxygen? 4. If the molar mass of the compound in problem 3 is 73. 8 grams/mole, what’s the molecular formula?