ELECTROLYTE SOLUTIONS MILLIEQUIVALENTS MILLIMOLES AND MILLIOSMOLES SUMMER 2017


























- Slides: 28

ELECTROLYTE SOLUTIONS: MILLIEQUIVALENTS, MILLIMOLES AND MILLIOSMOLES SUMMER 2017 Augustina Kwevie Pharm. D Candidate 1

OBJECTIVES • By the end of this lecture, students should be able to: • Calculate the milliequivalent weight from an atomic or formula weight • Convert between milligrams and milliequivalents • Calculate problems involving millimoles and milliosmoles 2

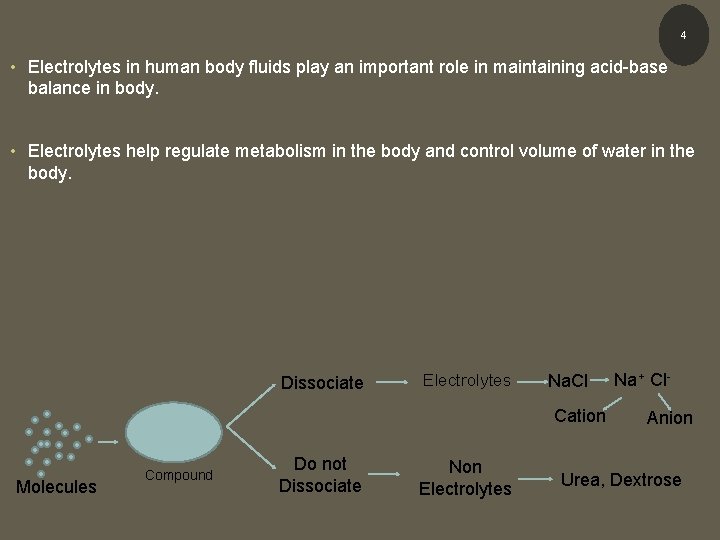
INTRODUCTION Electrolytes vs Non - electrolytes • Compounds in solution are often referred to as either electrolytes or non – electrolytes • Substances that do not dissociate are called non-electrolytes (urea, dextrose) – remain intact • Those with varying degrees of dissociation are called electrolytes (Na. Cl) • Electrolyte ions in blood plasma include cations (Na+, K+, Ca 2+, and Mg 2+) and anions (Cl-, HCO 3 -, HPO 42 -, SO 42 -) 3

4 • Electrolytes in human body fluids play an important role in maintaining acid-base balance in body. • Electrolytes help regulate metabolism in the body and control volume of water in the body. Dissociate Electrolytes Na. Cl Cation Molecules Compound Do not Dissociate Non Electrolytes Na+ Cl. Anion Urea, Dextrose

PHARMACEUTICAL APPLICATION • Electrolyte preparations are employed to treat fluid and electrolyte imbalances in the body. • Available as oral solutions, syrups, dry granules to be dissolved in water/juice, capsules, tablets and also intravenous infusions. 5

MILLIEQUIVALENTS • A chemical unit used by healthcare professionals across USA to express electrolyte concentration in solution • Internationally, molar concentrations (mmol/L or µmol/L) are employed. • A m. Eq represents the total number of ionic charges in solution, while taking into account valence of the ions. • A m. Eq expresses the chemical activity of an electrolyte 6

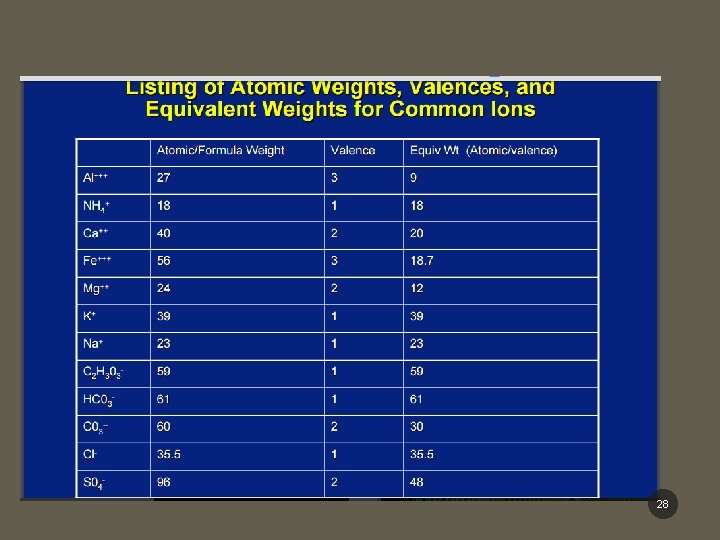
CALCULATIONS OF MILLIEQUIVALENTS • Equivalent weight (g) = atomic / molecular weight valence § Equivalent weight (Eq) = g § Milliequivalent weight (m. Eq) = mg Example: 1 equivalent KCl = 39 g K + 35. 5 g Cl = 74. 5 g /1 1 m. Eq = molecular weight (mg) valence Example: 1 m. Eq KCl = 39 mg K + 35. 5 mg Cl = 74. 5 mg/1 7

• To convert milligrams (mg) to milliequivalents (m. Eq) • m. Eq = mg × valence molecular weight • To convert m. Eq to mg • mg = m. Eq x molecular weight valence • m. Eq/m. L = mg/m. L × valence molecular weight • mg/m. L = m. Eq/m. L x molecular weight valence 8

PRACTICE PROBLEMS 9

EXAMPLE 1 • Convert 8 m. Eq of potassium to mg (MW of K+ = 39 g/mol) 10

EXAMPLE 2 • Calculate the concentration in mg/m. L, of a solution containing 3 m. Eq of KCl per m. L. (K+: 39 g/mol, Cl-: 35. 5 g/mol) 11

EXAMPLE 3 • What is the concentration in g/m. L of a solution containing 4 m. Eq of Calcium chloride (Ca. Cl 2. 2 H 2 O) per m. L? (Ca 2+: 40 g/mol, Cl-: 35. 5 g/mol, H: 1 g/mol, O: 16 g/mol) • 12

EXAMPLE 4: ESTIMATION OF % W/V FROM MEQ • What is the % w/v conc. of a 100 m. Eq/L solution of NH 4 Cl? (NH 4+: 18 g/mol, Cl-: 35. 5 g/mol) 13

EXAMPLE 5 • A solution has 10 mg / 100 m. L of K+ ions. Express the concentration in m. Eq/Lit (K+: 39 g/mol) 14

EXAMPLE 6 • A solution contains 10 mg/100 m. L of Ca 2+ ions. Express the concentration in m. Eq/Lit (Ca 2+: 40 g/mol) 15

EXAMPLE 7 • How many m. Eq of KCl is present in a 15 m. L dose of a 10% w/v KCl elixir? 16

EXAMPLE 8: CLINICAL APPLICATION • A person will receive 2 m. Eq of Na. Cl / kg of body weight. He weighs 132 lb. Calculate the ml of 0. 9% sterile Na. Cl that should be administered (Na+: 23 g/mol, Cl-: 35. 5 g/mol) 17

MILLIMOLES AND MICROMOLES Another way that drug amounts in solution can be expressed is as the number of particles in a given amount of solution • A mole is the molecular weight of substance in grams. • A millimole – one thousandth of a mole • A micromole – One millionth of a mole • SI expresses, electrolyte conc. in mmol/L § Conversions to remember: 1 mole = 1000 millimoles g/mole = mg/millimole 18

EXAMPLE 9 • How many mmol of monobasic sodium phosphate (m. w. 138 g/mol) are present in 100 g of the substance? 19

EXAMPLE 10 • Calculate the weight in mg of 1 mmol of HPO 4 - ( m. w. 96 g/mol) 20

EXAMPLE 11 • Convert plasma levels of 0. 5 µg/m. L of tobramycin (mw – 467. 52) to µmol/L? 21

OSMOLARITY • Osmotic pressure is important to biologic processes that involve the diffusion of solutes and the transfer of fluids through semi-permeable membranes • Osmotic pressure is proportional to the total number of particles in a solution • Unit of measurement is milliosmoles (m. Osmol) 22

OSMOLARITY • For non-electrolytes like dextrose, 1 mmol represents 1 mosmol • However for electrolytes, the total number of particles in solution depends on the degree of dissociation of a substance. • E. g. Assuming complete dissociation, 1 mmol Na. Cl represents 2 m. Osmol (Na+ + Cl-) of total particles • 1 mmol of Ca. Cl 2 represents 3 m. Osmol (Ca 2+ + 2 Cl-) • 1 mmol of sodium citrate (Na 3 C 6 H 5 O 7) represents 4 m. Osmol (3 Na+ + C 6 H 5 O 7 -) of total particles 23

• The milliosmolar value of the complete solution is equal to the sum of milliosmolar values of individual ions. • U. S. Pharmacopeia lists the following formula for calculation of ideal osmolar concentration: • Wt. of substance (g/L) • m. Osmol/L = --------------- × No. of Species × 1000 • Mol. Wt (g/mol) 24

EXAMPLE 12 • Calculate of ideal osmolarity for 0. 9% Sodium Chloride Solution? 25

OSMOLARITY VS. OSMOLALITY • Osmolarity -- “Milliosmoles of solute per liter of solution” • Osmolality – “Milliosmoles of solute per kilogram of solvent” • For dilute aqueous solutions – both terms are nearly identical • For more concentrated solutions – the two values are not identical • Pharmacist should make distinction between Osmolarity and Osmolality 26

EXAMPLE 13 • A solution has 5% anhydrous dextrose in water for injection. Represent the concentration in mosmol/lit? 27

28