Sour Bitter Electrolyte makes ions in solutions Perform
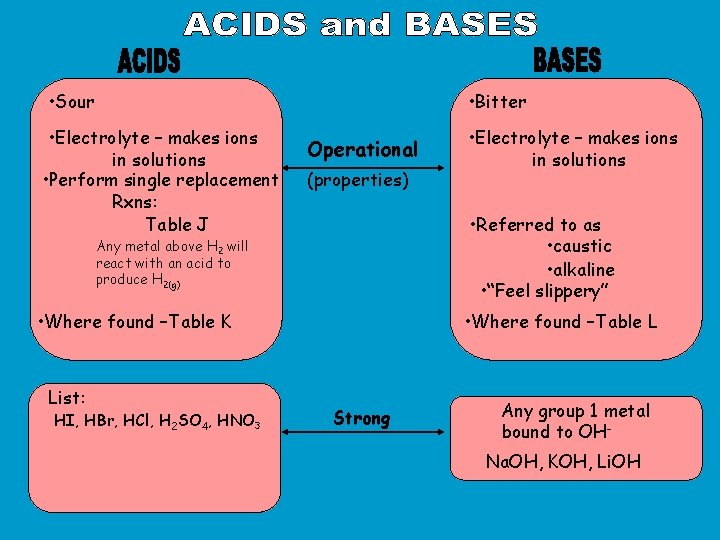
• Sour • Bitter • Electrolyte – makes ions in solutions • Perform single replacement Rxns: Table J Operational (properties) • Referred to as • caustic • alkaline • “Feel slippery” Any metal above H 2 will react with an acid to produce H 2(g) • Where found –Table K List: HI, HBr, HCl, H 2 SO 4, HNO 3 • Electrolyte – makes ions in solutions • Where found –Table L Strong Any group 1 metal bound to OHNa. OH, KOH, Li. OH
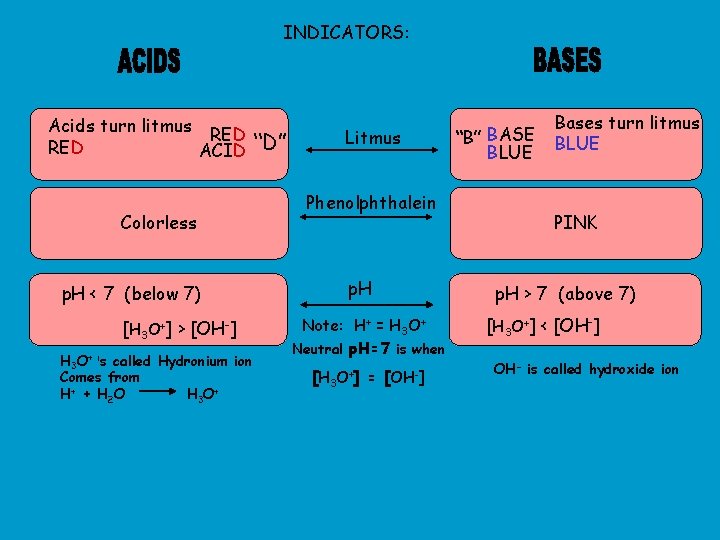
INDICATORS: Acids turn litmus RED ACID Colorless p. H < 7 (below 7) [H 3 O+] > [OH-] H 3 O+ is called Hydronium ion Comes from H+ + H 2 O H 3 O+ “D” Litmus Phenolphthalein p. H Note: H+ = H 3 O+ Neutral p. H=7 is when [H 3 O+] = [OH-] “B” BASE BLUE Bases turn litmus BLUE PINK p. H > 7 (above 7) [H 3 O+] < [OH-] OH- is called hydroxide ion
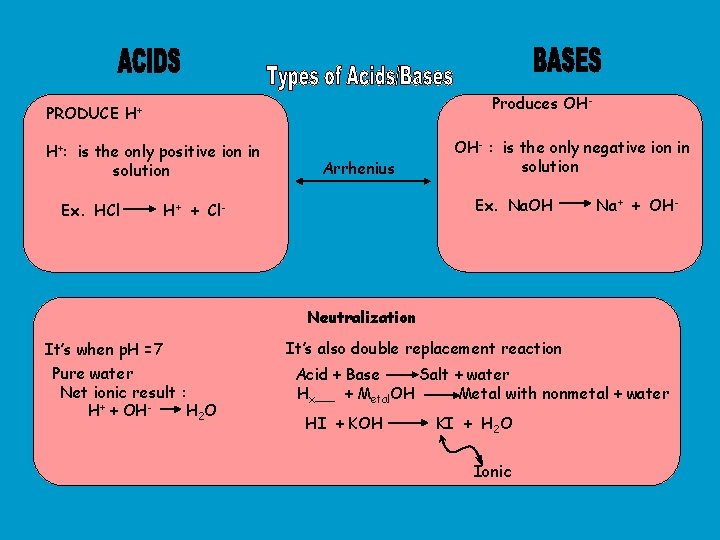
Produces OH- PRODUCE H+ H+: is the only positive ion in solution Ex. HCl Arrhenius OH- : is the only negative ion in solution Ex. Na. OH H+ + Cl- Na+ + OH- Neutralization It’s when p. H =7 Pure water Net ionic result : H+ + OHH 2 O It’s also double replacement reaction Acid + Base Salt + water Hx__ + Metal. OH Metal with nonmetal + water HI + KOH KI + H 2 O Ionic
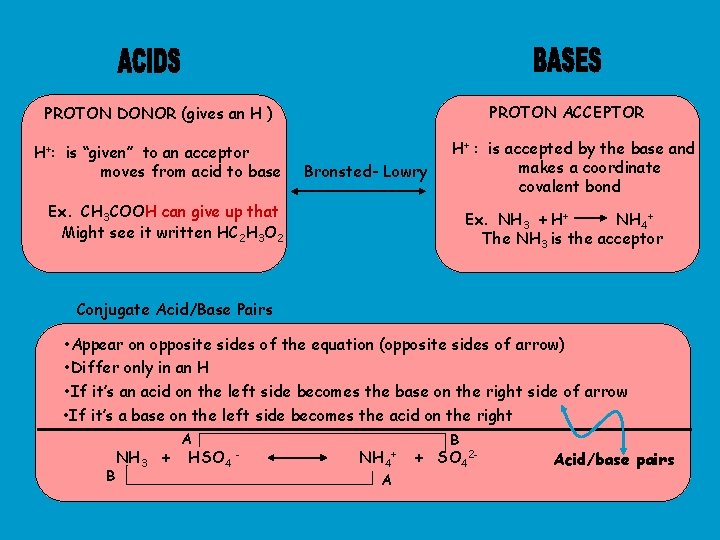
PROTON ACCEPTOR PROTON DONOR (gives an H ) H+: is “given” to an acceptor moves from acid to base Bronsted- Lowry H+ : is accepted by the base and makes a coordinate covalent bond Ex. CH 3 COOH can give up that Might see it written HC 2 H 3 O 2 Ex. NH 3 + H+ NH 4+ The NH 3 is the acceptor Conjugate Acid/Base Pairs • Appear on opposite sides of the equation (opposite sides of arrow) • Differ only in an H • If it’s an acid on the left side becomes the base on the right side of arrow • If it’s a base on the left side becomes the acid on the right B NH 3 + A HSO 4 - NH 4 A + B + SO 42 - Acid/base pairs
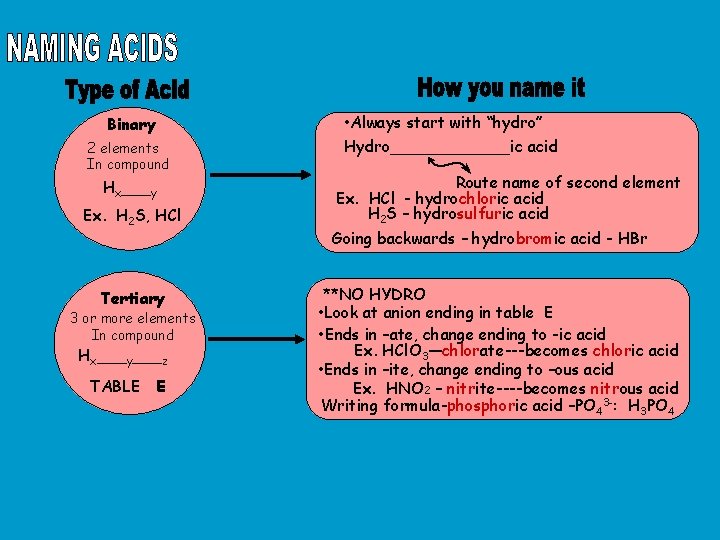
Binary 2 elements In compound Hx___y Ex. H 2 S, HCl Tertiary 3 or more elements In compound Hx___y___z TABLE E • Always start with “hydro” Hydro______ic acid Route name of second element Ex. HCl - hydrochloric acid H 2 S – hydrosulfuric acid Going backwards – hydrobromic acid - HBr **NO HYDRO • Look at anion ending in table E • Ends in –ate, change ending to -ic acid Ex. HCl. O 3—chlorate---becomes chloric acid • Ends in –ite, change ending to –ous acid Ex. HNO 2 – nitrite----becomes nitrous acid Writing formula-phosphoric acid –PO 43 -: H 3 PO 4
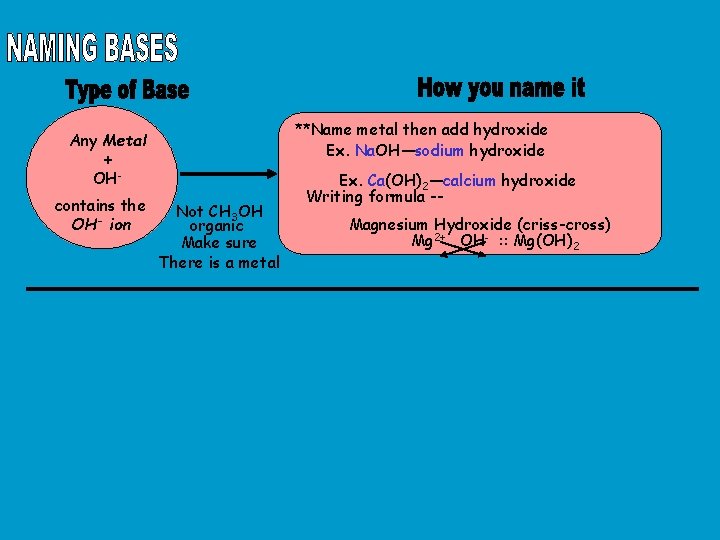
**Name metal then add hydroxide Ex. Na. OH—sodium hydroxide Any Metal + OHcontains the OH- ion Not CH 3 OH organic Make sure There is a metal Ex. Ca(OH)2—calcium hydroxide Writing formula -Magnesium Hydroxide (criss-cross) Mg 2+ OH- : : Mg(OH)2

Kw – Is the equilibrium constant for water • H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH-(aq) Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 @ 25ºC p. H + p. OH = 14 What is the hydroxide ion concentration if th hydronium ion concentration is 1. 0 x 10 -5 M? 1. 0 x 10 -5 M[OH-] = 1. 0 x 10 -14 [OH-] = Determining p. H – always use concentration of H+ ion Another way to look at it 1. 0 x 10 -14 1. 0 x 10 -5 p. H= -log[H+] = 1. 0 x 10 -9 -p. H The numerical value of exponent = value of p. H is a log scale. Each number change is a 10 fold change (factor of 10) Base Acid Neutral H+ OH 7 14 1 As you go H+ decrease, OH- increase, p. H increase As you go H+ increase, OH- decrease, p. H decreases If the p. H changes from 4 to 8, what is the change in H + concentration 4 to 5 to 6 to 7 to 8 10 x 10 x 10=10, 000 fold change DECREASE in H+ 10 10

What is the p. H of 1. 0 x 10 -3 M solution of HCl? HCl – is an acid so = [H+] = 1. 0 x 10 -3 p. H = 3 What is the p. H of 0. 00001 M solution of HCl? 0. 00001 M = 1 x 10 -5 =[H+] p. H = 5 What is the p. H of 1. 0 x 10 -3 M solution of Na. OH? Na. OH – is a base so = [OH-] Concentration of [OH-][H+]=1 x 10 -14 [H+] = 1 x 10 -11 p. H = 11 What is the p. H of 0. 001 M solution of Na. OH? Na. OH = 1 x 10 -3 Na. OH – is a base so = [OH-] Concentration of [OH-][H+]=1 x 10 -14 [H+] = 1 x 10 -11 p. H = 11
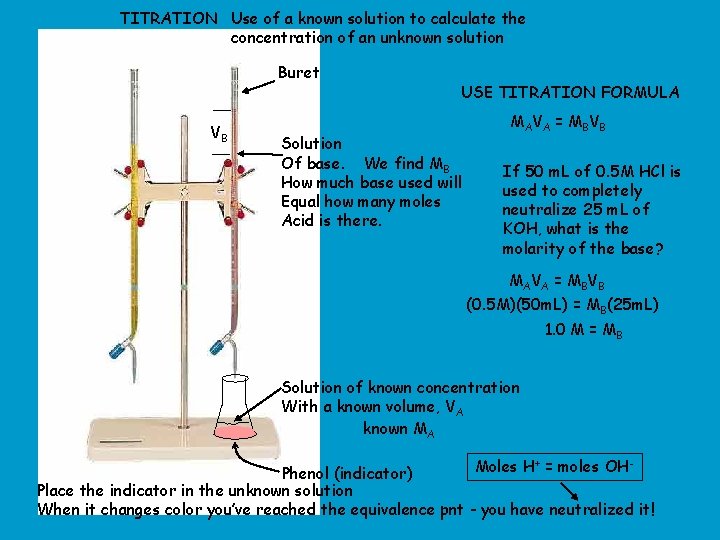
TITRATION Use of a known solution to calculate the concentration of an unknown solution Buret VB USE TITRATION FORMULA Solution Of base. We find MB How much base used will Equal how many moles Acid is there. MAVA = MBVB If 50 m. L of 0. 5 M HCl is used to completely neutralize 25 m. L of KOH, what is the molarity of the base? MAVA = MBVB (0. 5 M)(50 m. L) = MB(25 m. L) 1. 0 M = MB Solution of known concentration With a known volume, VA known MA Moles H+ = moles OHPhenol (indicator) Place the indicator in the unknown solution When it changes color you’ve reached the equivalence pnt - you have neutralized it!

OTHER Indicators – TABLE M Below the 1 st # - 1 st color listed Above the 2 nd # - 2 nd color listed Between the #’s on the chart – will be a blend of the colors Ex. A solutions p. H = 5. 8 then In methyl orange – color would be yellow In bromthymol blue – color would be yellow In litmus – color would be a reddish-purple (in middle)
- Slides: 10