24 Fluid Electrolyte and AcidBase Balance Power Point

24 Fluid, Electrolyte, and Acid-Base Balance Power. Point® Lecture Presentations prepared by Alexander G. Cheroske Mesa Community College at Red Mountain © 2011 Pearson Education, Inc.

Section 1: Fluid and Electrolyte Balance • Learning Outcomes • 24. 1 Explain what is meant by fluid balance, and discuss its importance for homeostasis. • 24. 2 Explain what is meant by mineral balance, and discuss its importance for homeostasis. • 24. 3 Summarize the relationship between sodium and water in maintaining fluid and electrolyte balance. • 24. 4 CLINICAL MODULE Explain factors that control potassium balance, and discuss hypokalemia and hyperkalemia. © 2011 Pearson Education, Inc.

Section 1: Fluid and Electrolyte Balance • Fluids constitute ~50%– 60% of total body composition • Minerals (inorganic substances) are dissolved within and form ions called electrolytes • Fluid compartments • Intracellular fluid (ICF) • Water content varies most here due to variation in: • Tissue types (muscle vs. fat) • Distinct from ECF due to plasma membrane transport • Extracellular fluid (ECF) • Interstitial fluid volume varies • Volume of blood (women < men) © 2011 Pearson Education, Inc.
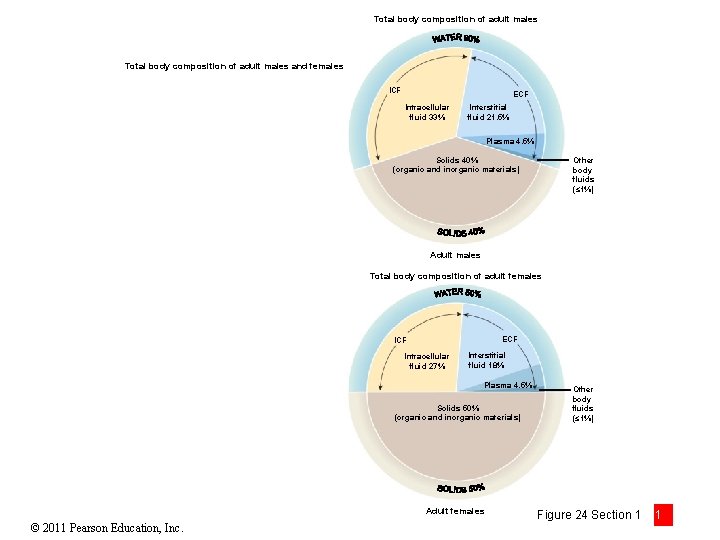
Total body composition of adult males and females ICF ECF Intracellular fluid 33% Interstitial fluid 21. 5% Plasma 4. 5% Solids 40% (organic and inorganic materials) Other body fluids (≤ 1%) Adult males Total body composition of adult females ECF Intracellular fluid 27% Interstitial fluid 18% Plasma 4. 5% Solids 50% (organic and inorganic materials) Adult females © 2011 Pearson Education, Inc. Other body fluids (≤ 1%) Figure 24 Section 1 1
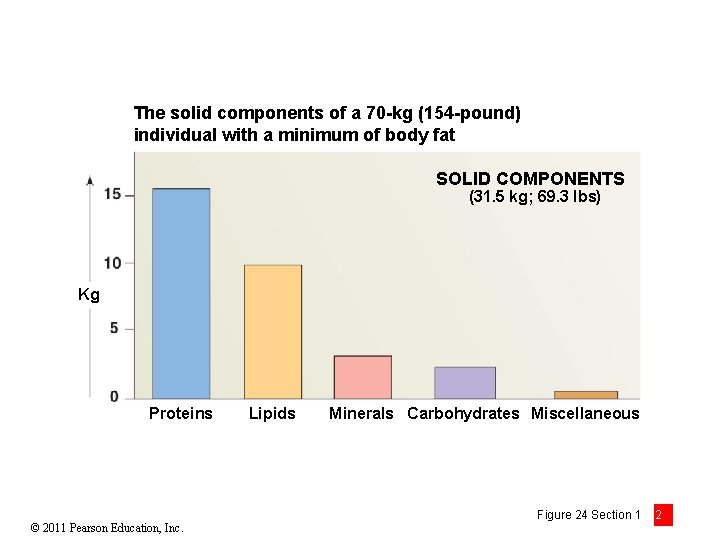
The solid components of a 70 -kg (154 -pound) individual with a minimum of body fat SOLID COMPONENTS (31. 5 kg; 69. 3 lbs) Kg Proteins © 2011 Pearson Education, Inc. Lipids Minerals Carbohydrates Miscellaneous Figure 24 Section 1 2

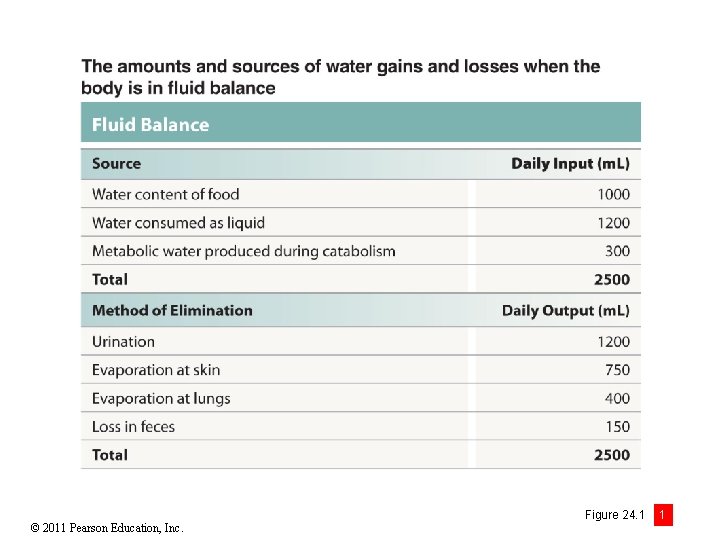
Module 24. 1: Fluid balance • Water content stable over time • Gains • Primarily absorption along digestive tract • As nutrients and ions are absorbed, osmotic gradient created causing passive absorption of water • Losses • Mainly through urination (over 50%) but other routes as well • Digestive secretions are reabsorbed similarly to ingested fluids © 2011 Pearson Education, Inc.
© 2011 Pearson Education, Inc. Figure 24. 1 1
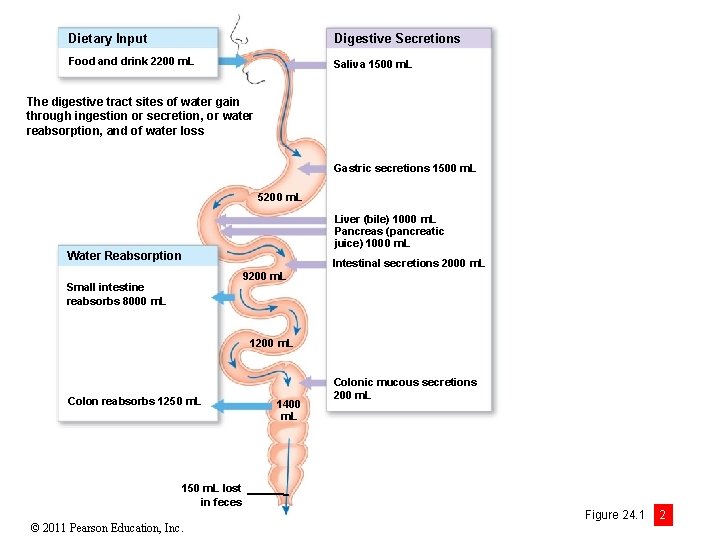
Dietary Input Digestive Secretions Food and drink 2200 m. L Saliva 1500 m. L The digestive tract sites of water gain through ingestion or secretion, or water reabsorption, and of water loss Gastric secretions 1500 m. L 5200 m. L Liver (bile) 1000 m. L Pancreas (pancreatic juice) 1000 m. L Water Reabsorption Intestinal secretions 2000 m. L 9200 m. L Small intestine reabsorbs 8000 m. L 1200 m. L Colon reabsorbs 1250 m. L 1400 m. L Colonic mucous secretions 200 m. L 150 m. L lost in feces © 2011 Pearson Education, Inc. Figure 24. 1 2

Module 24. 1: Fluid balance • ICF and ECF compartments balance • Very different composition • Are at osmotic equilibrium • Loss of water from ECF is replaced by water in ICF • = Fluid shift • Occurs in minutes to hours and restores osmotic equilibrium • Dehydration • Results in long-term transfer that cannot replace ECF water loss • Homeostatic mechanisms to increase ECF fluid volume will be employed © 2011 Pearson Education, Inc.
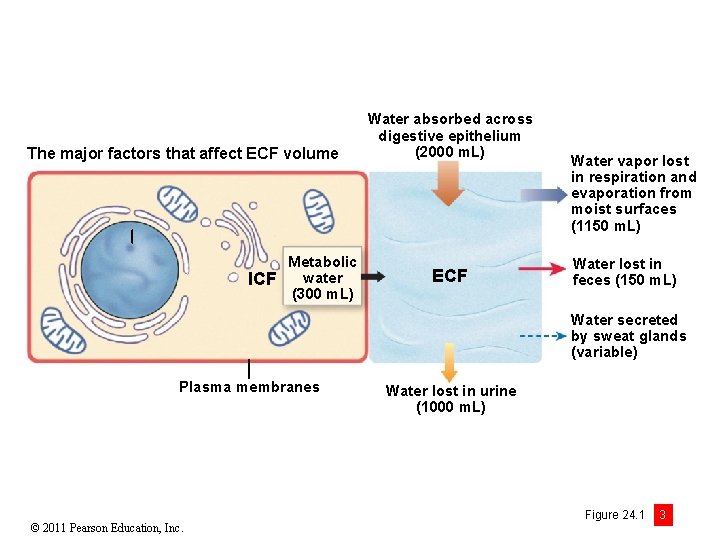
The major factors that affect ECF volume ICF Metabolic water (300 m. L) Water absorbed across digestive epithelium (2000 m. L) ECF Water vapor lost in respiration and evaporation from moist surfaces (1150 m. L) Water lost in feces (150 m. L) Water secreted by sweat glands (variable) Plasma membranes © 2011 Pearson Education, Inc. Water lost in urine (1000 m. L) Figure 24. 1 3
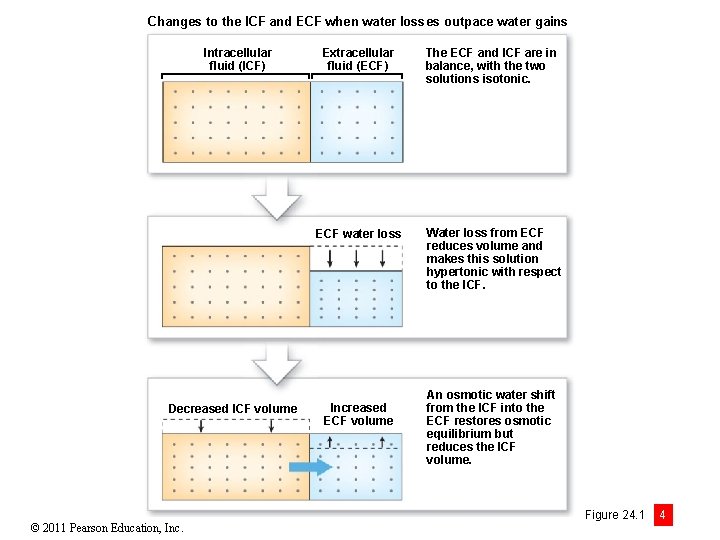
Changes to the ICF and ECF when water losses outpace water gains Intracellular fluid (ICF) Decreased ICF volume © 2011 Pearson Education, Inc. Extracellular fluid (ECF) The ECF and ICF are in balance, with the two solutions isotonic. ECF water loss Water loss from ECF reduces volume and makes this solution hypertonic with respect to the ICF. Increased ECF volume An osmotic water shift from the ICF into the ECF restores osmotic equilibrium but reduces the ICF volume. Figure 24. 1 4

Module 24. 1 Review a. Identify routes of fluid loss from the body. b. Describe a fluid shift. c. Explain dehydration and its effect on the osmotic concentration of plasma. © 2011 Pearson Education, Inc.

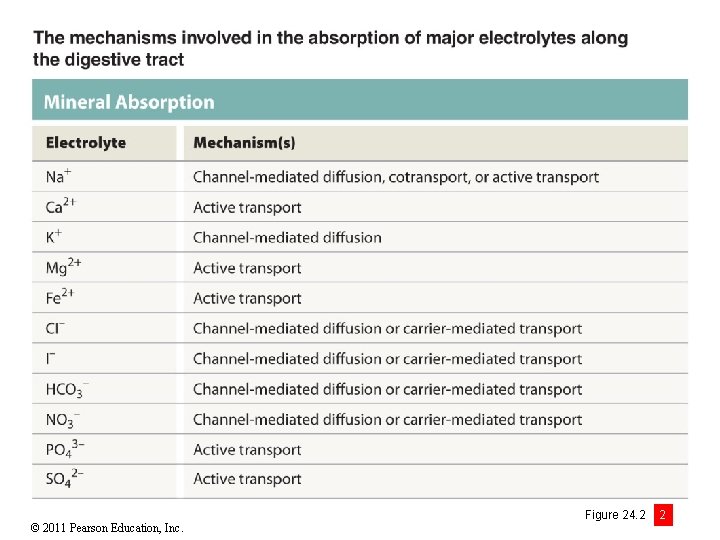
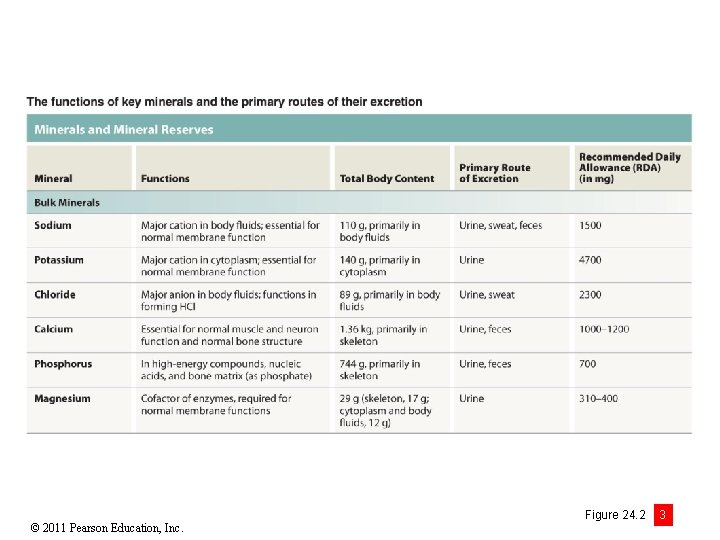
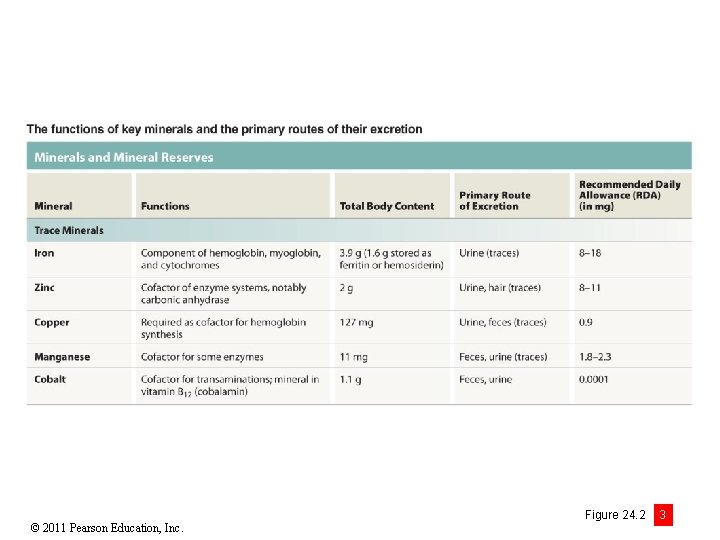
Module 24. 2: Mineral balance • Equilibrium between ion absorption and excretion • Major ion absorption through intestine and colon • Major ion excretion by kidneys • Sweat glands excrete ions and water variably • Ion reserves mainly in skeleton © 2011 Pearson Education, Inc.
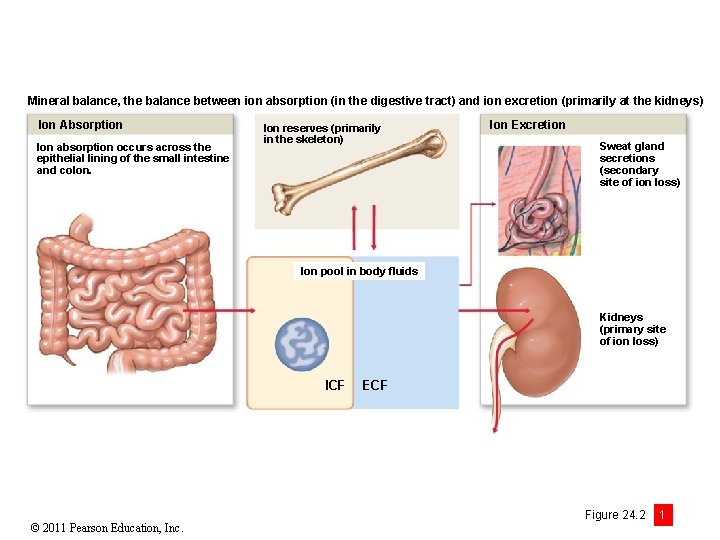
Mineral balance, the balance between ion absorption (in the digestive tract) and ion excretion (primarily at the kidneys) Ion Absorption Ion absorption occurs across the epithelial lining of the small intestine and colon. Ion reserves (primarily in the skeleton) Ion Excretion Sweat gland secretions (secondary site of ion loss) Ion pool in body fluids Kidneys (primary site of ion loss) ICF © 2011 Pearson Education, Inc. ECF Figure 24. 2 1
© 2011 Pearson Education, Inc. Figure 24. 2 2
© 2011 Pearson Education, Inc. Figure 24. 2 3
© 2011 Pearson Education, Inc. Figure 24. 2 3

Module 24. 2 Review a. Define mineral balance. b. Identify the significance of two important body minerals: sodium and calcium. c. Identify the ions absorbed by active transport. © 2011 Pearson Education, Inc.

Module 24. 3: Water and sodium balance • Sodium balance (when sodium gains equal losses) • Relatively small changes in Na+ are accommodated by changes in ECF volume • Homeostatic responses involve two parts 1. ADH control of water loss/retention by kidneys and thirst 2. Fluid exchange between ECF and ICF © 2011 Pearson Education, Inc.
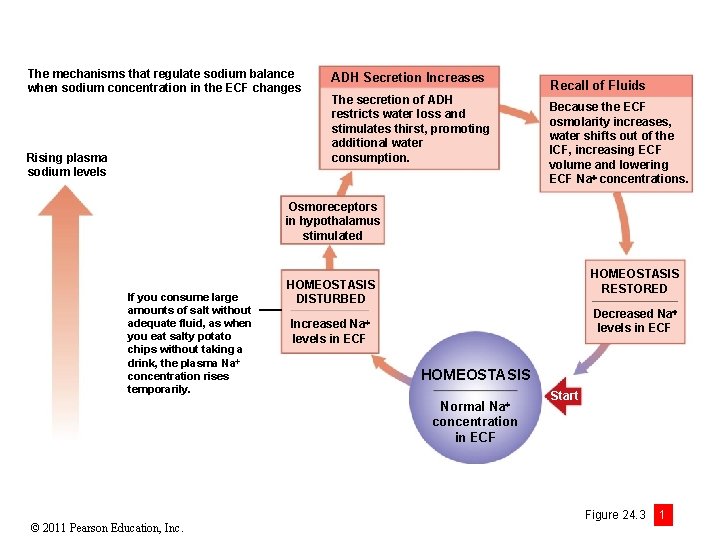
The mechanisms that regulate sodium balance when sodium concentration in the ECF changes Rising plasma sodium levels ADH Secretion Increases The secretion of ADH restricts water loss and stimulates thirst, promoting additional water consumption. Recall of Fluids Because the ECF osmolarity increases, water shifts out of the ICF, increasing ECF volume and lowering ECF Na concentrations. Osmoreceptors in hypothalamus stimulated If you consume large amounts of salt without adequate fluid, as when you eat salty potato chips without taking a drink, the plasma Na concentration rises temporarily. HOMEOSTASIS RESTORED HOMEOSTASIS DISTURBED Decreased Na levels in ECF Na Increased levels in ECF HOMEOSTASIS Normal Na concentration in ECF © 2011 Pearson Education, Inc. Start Figure 24. 3 1
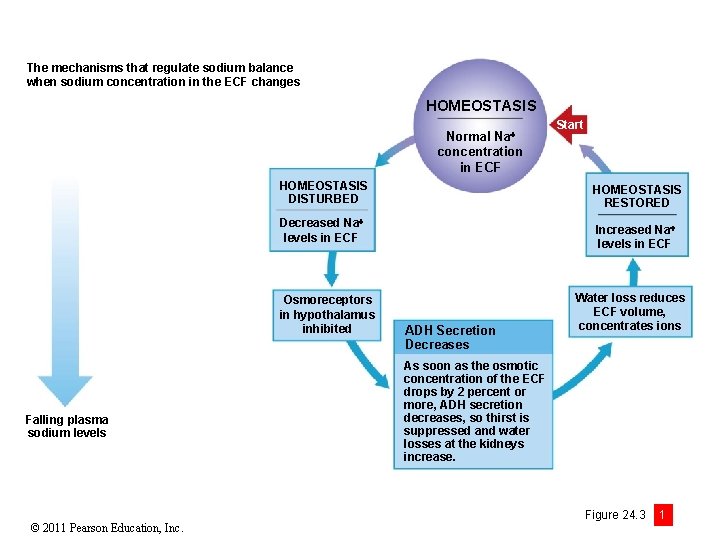
The mechanisms that regulate sodium balance when sodium concentration in the ECF changes HOMEOSTASIS Normal Na concentration in ECF HOMEOSTASIS DISTURBED HOMEOSTASIS RESTORED Decreased Na levels in ECF Osmoreceptors in hypothalamus inhibited Falling plasma sodium levels © 2011 Pearson Education, Inc. Start Increased Na levels in ECF ADH Secretion Decreases Water loss reduces ECF volume, concentrates ions As soon as the osmotic concentration of the ECF drops by 2 percent or more, ADH secretion decreases, so thirst is suppressed and water losses at the kidneys increase. Figure 24. 3 1

Module 24. 3: Water and sodium balance • Sodium balance (continued) • Exchanges in Na+ are accommodated by changes in blood pressure and volume • Hyponatremia (natrium, sodium) • Low ECF Na+ concentration (<136 m. Eq/L) • Can occur from overhydration or inadequate salt intake • Hypernatremia • High ECF Na+ concentration (>145 m. Eq/L) • Commonly from dehydration © 2011 Pearson Education, Inc.

Module 24. 3: Water and sodium balance • Sodium balance (continued) • Exchanges in Na+ are accommodated by changes in blood pressure and volume (continued) • Increased blood volume and pressure • Natriuretic peptides released • Increased Na+ and water loss in urine • Reduced thirst • Inhibition of ADH, aldosterone, epinephrine, and norepinephrine release • Decreased blood volume and pressure • Endocrine response • Increased ADH, aldosterone, RAAS mechanism • Opposite bodily responses to above © 2011 Pearson Education, Inc.
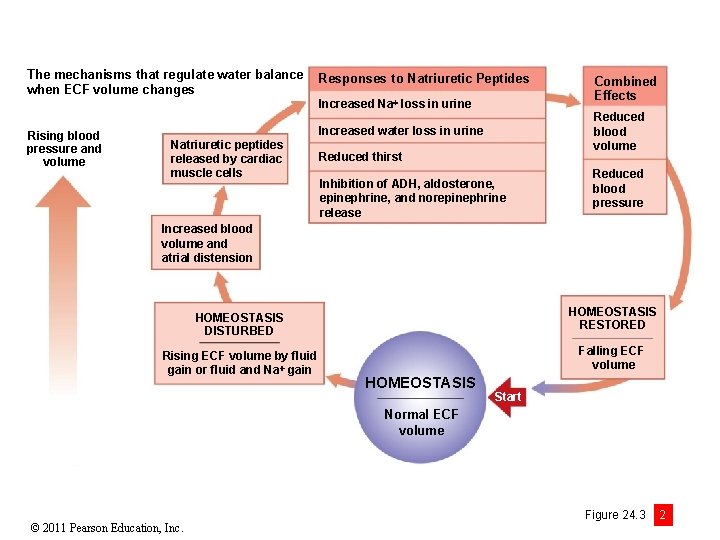
The mechanisms that regulate water balance when ECF volume changes Responses to Natriuretic Peptides Rising blood pressure and volume Increased water loss in urine Natriuretic peptides released by cardiac muscle cells Increased Na loss in urine Combined Effects Reduced blood volume Reduced thirst Inhibition of ADH, aldosterone, epinephrine, and norepinephrine release Reduced blood pressure Increased blood volume and atrial distension HOMEOSTASIS RESTORED HOMEOSTASIS DISTURBED Rising ECF volume by fluid gain or fluid and Na gain Falling ECF volume HOMEOSTASIS Start Normal ECF volume © 2011 Pearson Education, Inc. Figure 24. 3 2
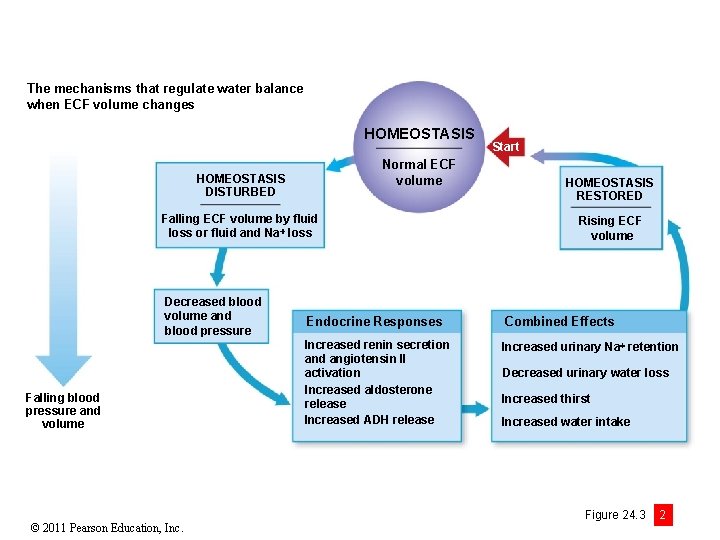
The mechanisms that regulate water balance when ECF volume changes HOMEOSTASIS Normal ECF volume HOMEOSTASIS DISTURBED Falling ECF volume by fluid loss or fluid and Na loss Decreased blood volume and blood pressure Falling blood pressure and volume © 2011 Pearson Education, Inc. Start HOMEOSTASIS RESTORED Rising ECF volume Endocrine Responses Combined Effects Increased renin secretion and angiotensin II activation Increased aldosterone release Increased ADH release Increased urinary Na retention Decreased urinary water loss Increased thirst Increased water intake Figure 24. 3 2

Module 24. 3 Review a. What effect does inhibition of osmoreceptors have on ADH secretion and thirst? b. What effect does aldosterone have on sodium ion concentration in the ECF? c. Briefly summarize the relationship between sodium ion concentration and the ECF. © 2011 Pearson Education, Inc.

CLINICAL MODULE 24. 4: Potassium imbalance • Potassium balance (K+ gain = loss) • Major gain is through digestive tract absorption • ~100 m. Eq (1. 9– 5. 8 g)/day • Major loss is excretion by kidneys • Primary ECF potassium regulation by kidneys since intake fairly constant • Controlled by aldosterone regulating Na+/K+ exchange pumps in DCT and collecting duct of nephron • Low ECF p. H can cause H+ to be substituted for K+ • Potassium is highest in ICF due to Na+/K+ exchange pump • ~135 m. Eq/L in ICF vs. ~5 m. Eq/L in ECF © 2011 Pearson Education, Inc.
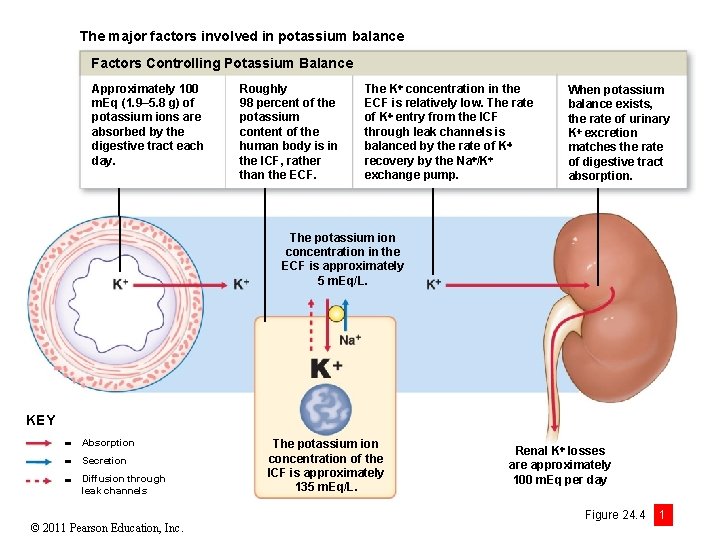
The major factors involved in potassium balance Factors Controlling Potassium Balance Approximately 100 m. Eq (1. 9– 5. 8 g) of potassium ions are absorbed by the digestive tract each day. Roughly 98 percent of the potassium content of the human body is in the ICF, rather than the ECF. The K concentration in the ECF is relatively low. The rate of K entry from the ICF through leak channels is balanced by the rate of K recovery by the Na /K exchange pump. When potassium balance exists, the rate of urinary K excretion matches the rate of digestive tract absorption. The potassium ion concentration in the ECF is approximately 5 m. Eq/L. KEY Absorption Secretion Diffusion through leak channels © 2011 Pearson Education, Inc. The potassium ion concentration of the ICF is approximately 135 m. Eq/L. Renal K losses are approximately 100 m. Eq per day Figure 24. 4 1
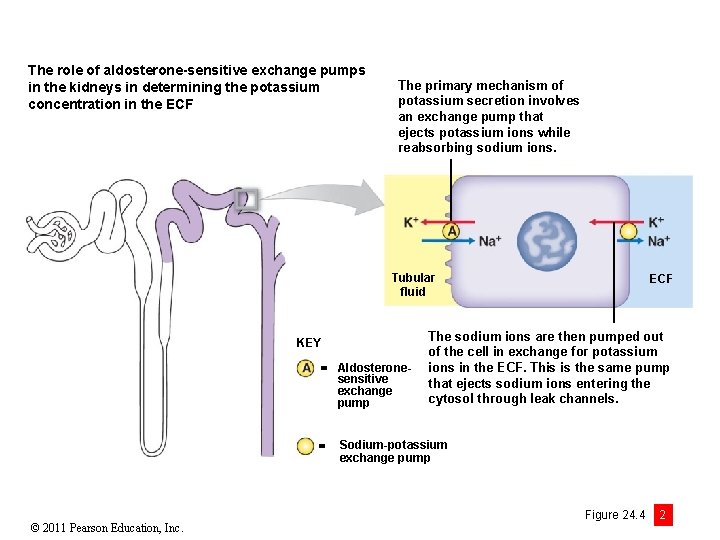
The role of aldosterone-sensitive exchange pumps in the kidneys in determining the potassium concentration in the ECF The primary mechanism of potassium secretion involves an exchange pump that ejects potassium ions while reabsorbing sodium ions. Tubular fluid KEY Aldosteronesensitive exchange pump © 2011 Pearson Education, Inc. ECF The sodium ions are then pumped out of the cell in exchange for potassium ions in the ECF. This is the same pump that ejects sodium ions entering the cytosol through leak channels. Sodium-potassium exchange pump Figure 24. 4 2
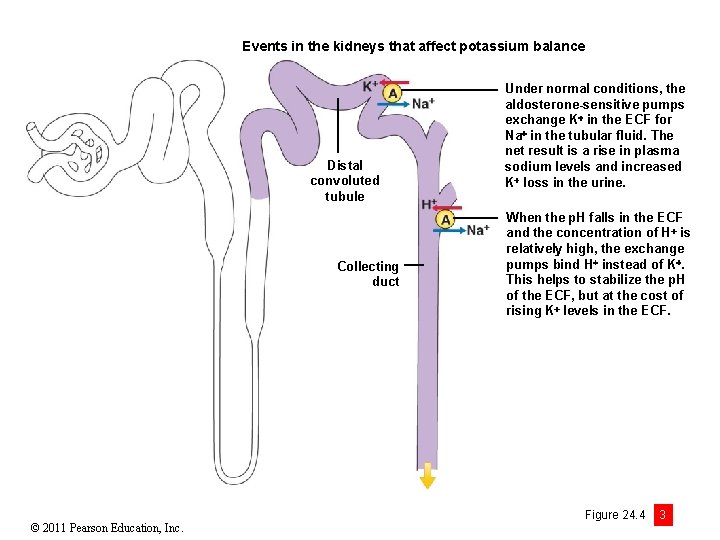
Events in the kidneys that affect potassium balance Distal convoluted tubule Collecting duct © 2011 Pearson Education, Inc. Under normal conditions, the aldosterone-sensitive pumps exchange K in the ECF for Na in the tubular fluid. The net result is a rise in plasma sodium levels and increased K loss in the urine. When the p. H falls in the ECF and the concentration of H is relatively high, the exchange pumps bind H instead of K . This helps to stabilize the p. H of the ECF, but at the cost of rising K levels in the ECF. Figure 24. 4 3

CLINICAL MODULE 24. 4: Potassium imbalance • Disturbances of potassium balance • Hypokalemia (kalium, potassium) • Below 2 m. Eq/L in plasma • Can be caused by: • Diuretics • Aldosteronism (excessive aldosterone secretion) • Symptoms • Muscular weakness, followed by paralysis • Potentially lethal when affecting heart © 2011 Pearson Education, Inc.

CLINICAL MODULE 24. 4: Potassium imbalance • Disturbances of potassium balance (continued) • Hyperkalemia • Above 8 m. Eq/L in plasma • Can be caused by: • Chronically low p. H • Kidney failure • Drugs promoting diuresis by blocking Na+/K+ pumps • Symptoms • Muscular spasm including heart arrhythmias © 2011 Pearson Education, Inc.

CLINICAL MODULE 24. 4 Review a. Define hypokalemia and hyperkalemia. b. What organs are primarily responsible for regulating the potassium ion concentration of the ECF? c. Identify factors that cause potassium excretion. © 2011 Pearson Education, Inc.

Section 2: Acid-Base Balance • Learning Outcomes • 24. 5 Explain the role of buffer systems in maintaining acid-base balance and p. H. • 24. 6 Explain the role of buffer systems in regulating the p. H of the intracellular fluid and the extracellular fluid. • 24. 7 Describe the compensatory mechanisms involved in the maintenance of acid-base balance. • 24. 8 CLINICAL MODULE Describe respiratory acidosis and respiratory alkalosis. © 2011 Pearson Education, Inc.

Section 2: Acid-Base Balance • Acid-base balance (H+ production = loss) • Normal plasma p. H: 7. 35– 7. 45 • H+ gains: many metabolic activities produce acids • CO 2 (to carbonic acid) from aerobic respiration • Lactic acid from glycolysis • H+ losses and storage • Respiratory system eliminates CO 2 • H+ excretion from kidneys • Buffers temporarily store H+ © 2011 Pearson Education, Inc.
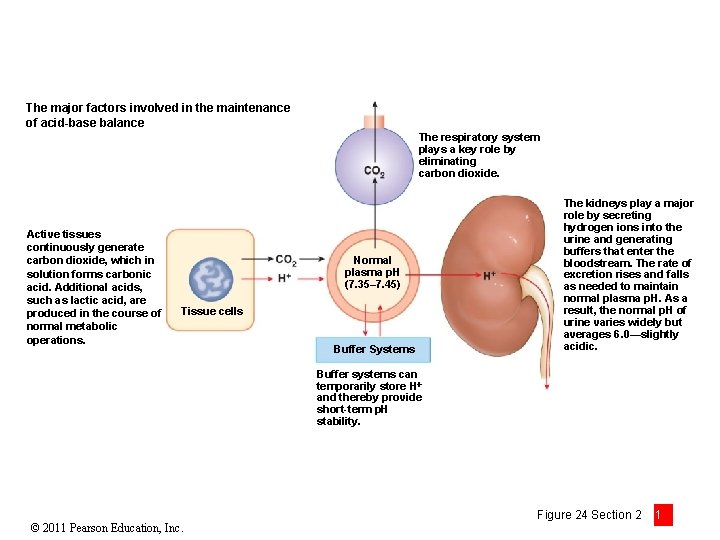
The major factors involved in the maintenance of acid-base balance The respiratory system plays a key role by eliminating carbon dioxide. Active tissues continuously generate carbon dioxide, which in solution forms carbonic acid. Additional acids, such as lactic acid, are produced in the course of normal metabolic operations. Normal plasma p. H (7. 35– 7. 45) Tissue cells Buffer Systems The kidneys play a major role by secreting hydrogen ions into the urine and generating buffers that enter the bloodstream. The rate of excretion rises and falls as needed to maintain normal plasma p. H. As a result, the normal p. H of urine varies widely but averages 6. 0—slightly acidic. Buffer systems can temporarily store H and thereby provide short-term p. H stability. © 2011 Pearson Education, Inc. Figure 24 Section 2 1

Section 2: Acid-Base Balance • Classes of acids • Fixed acids • Do not leave solution • • Examples: sulfuric and phosphoric acid • • Remain in body fluids until kidney excretion Generated during catabolism of amino acids, phospholipids, and nucleic acids Organic acids • Part of cellular metabolism • • Examples: lactic acid and ketones Most metabolized rapidly so no accumulation © 2011 Pearson Education, Inc.

Section 2: Acid-Base Balance • Classes of acids (continued) • Volatile acids • Can leave body by external respiration • Example: carbonic acid (H 2 CO 3) © 2011 Pearson Education, Inc.

Module 24. 5: Buffer systems • p. H imbalance • ECH p. H normally between 7. 35 and 7. 45 • Acidemia (plasma p. H <7. 35): acidosis (physiological state) • More common due to acid-producing metabolic activities • Effects • CNS function deteriorates, may cause coma • Cardiac contractions grow weak and irregular • Peripheral vasodilation causes BP drop • Alkalemia (plasma p. H >7. 45): alkalosis (physiological state) • Can be dangerous but relatively rare © 2011 Pearson Education, Inc.
© 2011 Pearson Education, Inc. Figure 24. 5 1
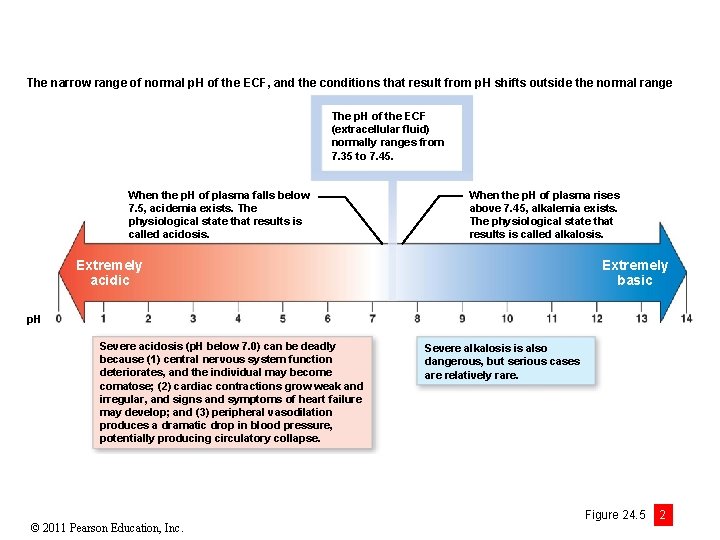
The narrow range of normal p. H of the ECF, and the conditions that result from p. H shifts outside the normal range The p. H of the ECF (extracellular fluid) normally ranges from 7. 35 to 7. 45. When the p. H of plasma falls below 7. 5, acidemia exists. The physiological state that results is called acidosis. When the p. H of plasma rises above 7. 45, alkalemia exists. The physiological state that results is called alkalosis. Extremely acidic Extremely basic p. H Severe acidosis (p. H below 7. 0) can be deadly because (1) central nervous system function deteriorates, and the individual may become comatose; (2) cardiac contractions grow weak and irregular, and signs and symptoms of heart failure may develop; and (3) peripheral vasodilation produces a dramatic drop in blood pressure, potentially producing circulatory collapse. © 2011 Pearson Education, Inc. Severe alkalosis is also dangerous, but serious cases are relatively rare. Figure 24. 5 2

Module 24. 5: Buffer systems • CO 2 partial pressure effects on p. H • Most important factor affecting body p. H • H 2 O + CO 2 H 2 CO 3 H+ + HCO 3– • Reversible reaction that can buffer body p. H • Adjustments in respiratory rate can affect body p. H © 2011 Pearson Education, Inc.
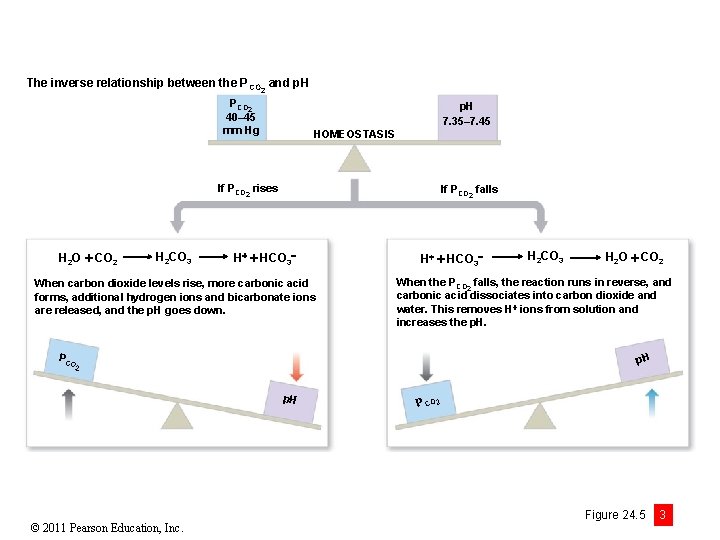
The inverse relationship between the P CO 2 and p. H PCO 2 40– 45 mm Hg p. H 7. 35– 7. 45 HOMEOSTASIS If PCO 2 rises H 2 O CO 2 H 2 CO 3 If PCO 2 falls H HCO 3 When carbon dioxide levels rise, more carbonic acid forms, additional hydrogen ions and bicarbonate ions are released, and the p. H goes down. H HCO 3 H 2 O CO 2 When the PCO 2 falls, the reaction runs in reverse, and carbonic acid dissociates into carbon dioxide and water. This removes H ions from solution and increases the p. H. PCO p. H 2 p. H © 2011 Pearson Education, Inc. H 2 CO 3 P CO 2 Figure 24. 5 3

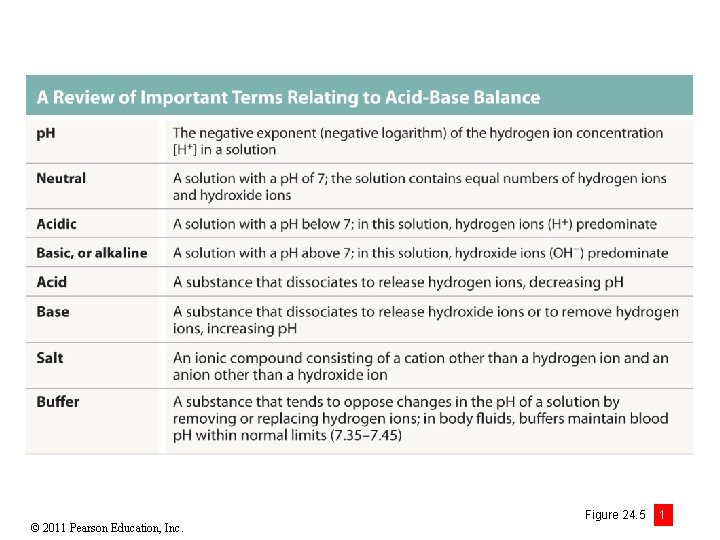
Module 24. 5: Buffer systems • Buffer • Substance that opposes changes to p. H by removing or adding H+ • Generally consists of: • Weak acid (HY) • Anion released by its dissociation (Y–) • HY H+ + Y– and H+ + Y– HY © 2011 Pearson Education, Inc.

The reactions that occur when p. H buffer systems function A buffer system in body fluids generally consists of a combination of a weak acid (HY) and the anion (Y ) released by its dissociation. The anion functions as a weak base. In solution, molecules of the weak acid exist in equilibrium with its dissociation products. HY H © 2011 Pearson Education, Inc. Y Adding H to the solution upsets the equilibrium and results in the formation of additional molecules of the weak acid. H Y H H HY Removing H from the solution also upsets the equilibrium and results in the dissociation of additional molecules of HY. This releases H. H HY H Figure 24. 5 4

Module 24. 5 Review a. Define acidemia and alkalemia. b. What is the most important factor affecting the p. H of the ECF? c. Summarize the relationship between CO 2 levels and p. H. © 2011 Pearson Education, Inc.

Module 24. 6: Major body buffer systems • Three major body buffer systems • All can only temporarily affect p. H (H+ not eliminated) 1. Phosphate buffer system • Buffers p. H of ICF and urine 2. Carbonic acid–bicarbonate buffer system • Most important in ECF • Fully reversible • Bicarbonate reserves (from Na. HCO 3 in ECF) contribute © 2011 Pearson Education, Inc.

Module 24. 6: Major body buffer systems • Three major body buffer systems (continued) 3. Protein buffer systems (in ICF and ECF) • Usually operate under acid conditions (bind H+) • • Binding to carboxyl group (COOH–) and amino group ( —NH 2) Examples: • Hemoglobin buffer system • CO 2 + H 2 O H 2 CO 3 HCO 3– + Hb-H+ • Only intracellular system with immediate effects • Amino acid buffers (all proteins) • Plasma proteins © 2011 Pearson Education, Inc.
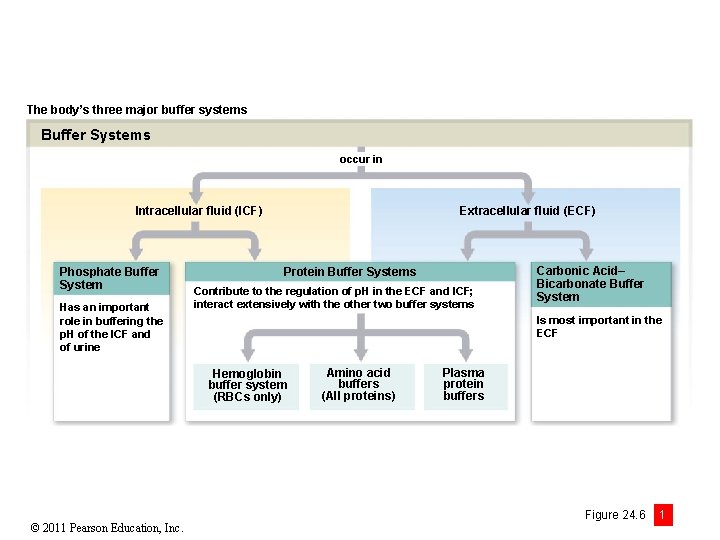
The body’s three major buffer systems Buffer Systems occur in Intracellular fluid (ICF) Phosphate Buffer System Has an important role in buffering the p. H of the ICF and of urine Extracellular fluid (ECF) Protein Buffer Systems Contribute to the regulation of p. H in the ECF and ICF; interact extensively with the other two buffer systems Is most important in the ECF Hemoglobin buffer system (RBCs only) © 2011 Pearson Education, Inc. Carbonic Acid– Bicarbonate Buffer System Amino acid buffers (All proteins) Plasma protein buffers Figure 24. 6 1
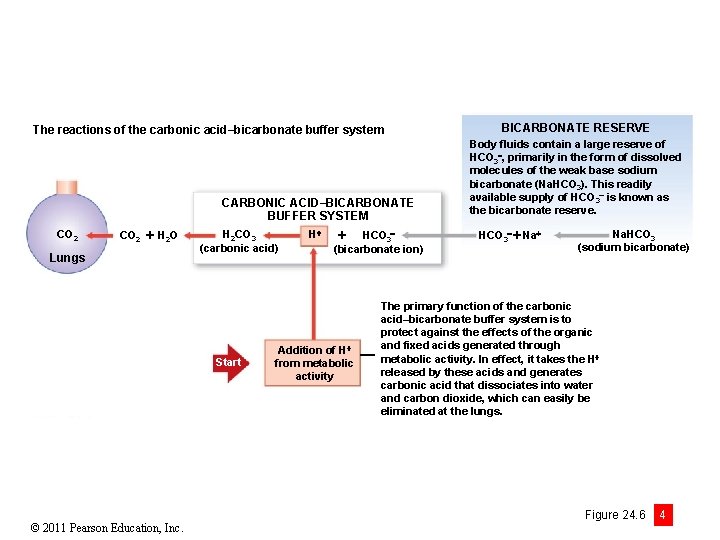
The reactions of the carbonic acid–bicarbonate buffer system CARBONIC ACID–BICARBONATE BUFFER SYSTEM CO 2 H 2 O Lungs H 2 CO 3 (carbonic acid) Start © 2011 Pearson Education, Inc. H HCO 3 (bicarbonate ion) Addition of H from metabolic activity BICARBONATE RESERVE Body fluids contain a large reserve of HCO 3 , primarily in the form of dissolved molecules of the weak base sodium bicarbonate (Na. HCO 3). This readily available supply of HCO 3 is known as the bicarbonate reserve. HCO 3 Na. HCO 3 (sodium bicarbonate) The primary function of the carbonic acid–bicarbonate buffer system is to protect against the effects of the organic and fixed acids generated through metabolic activity. In effect, it takes the H released by these acids and generates carbonic acid that dissociates into water and carbon dioxide, which can easily be eliminated at the lungs. Figure 24. 6 4
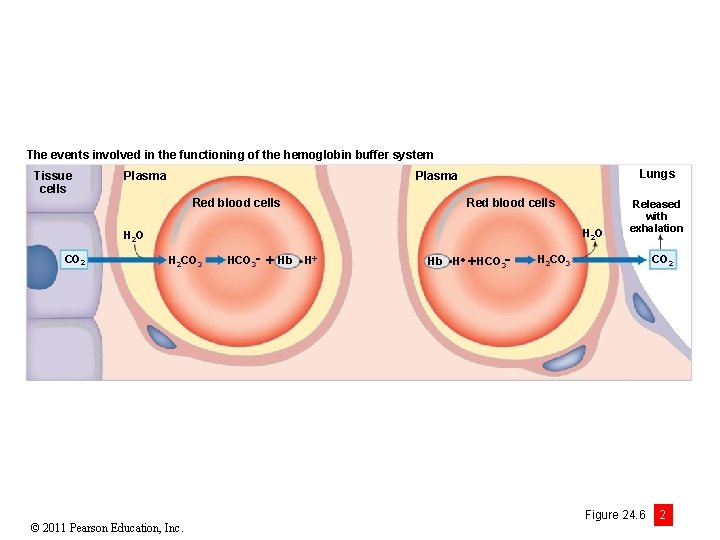
The events involved in the functioning of the hemoglobin buffer system Tissue cells Plasma Lungs Plasma Red blood cells H 2 O CO 2 H 2 CO 3 © 2011 Pearson Education, Inc. HCO 3 Hb H HCO 3 Released with exhalation H 2 CO 3 CO 2 Figure 24. 6 2
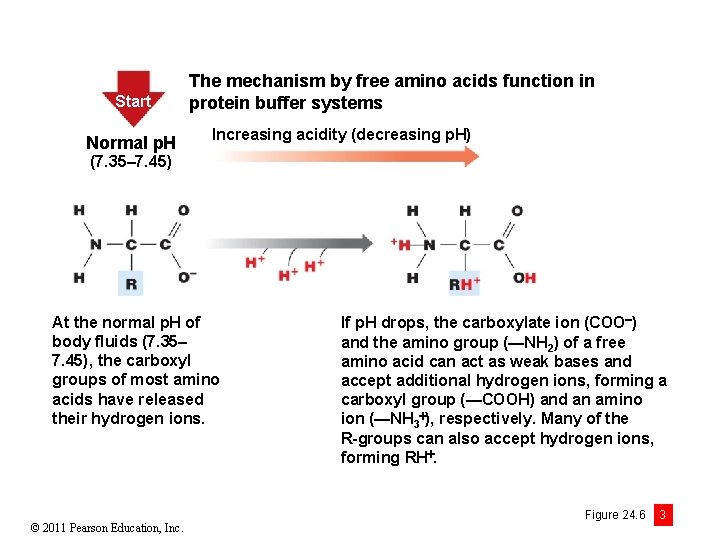
Start Normal p. H The mechanism by free amino acids function in protein buffer systems Increasing acidity (decreasing p. H) (7. 35– 7. 45) At the normal p. H of body fluids (7. 35– 7. 45), the carboxyl groups of most amino acids have released their hydrogen ions. © 2011 Pearson Education, Inc. If p. H drops, the carboxylate ion (COO ) and the amino group (—NH 2) of a free amino acid can act as weak bases and accept additional hydrogen ions, forming a carboxyl group (—COOH) and an amino ion (—NH 3 ), respectively. Many of the R-groups can also accept hydrogen ions, forming RH. Figure 24. 6 3

Module 24. 6: Major body buffer systems • Disorders • Metabolic acid-base disorders • Production or loss of excessive amounts of fixed or organic acids • Carbonic acid–bicarbonate system works to counter • Respiratory acid-base disorders • Imbalance of CO 2 generation and elimination • Must be corrected by depth and rate of respiration changes © 2011 Pearson Education, Inc.

Module 24. 6 Review a. Identify the body’s three major buffer systems. b. Describe the carbonic acid–bicarbonate buffer system. c. Describe the roles of the phosphate buffer system. © 2011 Pearson Education, Inc.

Module 24. 7: Metabolic acid-base disorders • Metabolic acidosis • • Develops when large numbers of H+ are released by organic or fixed acids Accommodated by respiratory and renal responses • • Respiratory response • Increased respiratory rate lowers PCO 2 • H+ + HCO 3– H 2 CO 3 H 2 O + CO 2 Renal response • Occurs in PCT, DCT, and collecting system • H 2 O + CO 2 H 2 CO 3 H+ + HCO 3– H+ secreted into urine HCO 3– reabsorbed into ECF © 2011 Pearson Education, Inc.
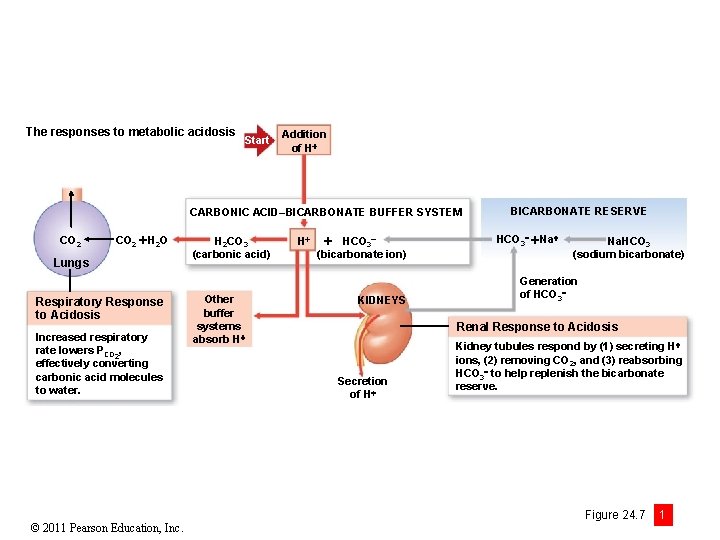
The responses to metabolic acidosis Start Addition of H CARBONIC ACID–BICARBONATE BUFFER SYSTEM CO 2 H 2 O Lungs Respiratory Response to Acidosis Increased respiratory rate lowers PCO 2, effectively converting carbonic acid molecules to water. © 2011 Pearson Education, Inc. H 2 CO 3 (carbonic acid) Other buffer systems absorb H H HCO 3 (bicarbonate ion) KIDNEYS BICARBONATE RESERVE HCO 3 Na. HCO 3 (sodium bicarbonate) Generation of HCO 3 Renal Response to Acidosis Secretion of H Kidney tubules respond by (1) secreting H ions, (2) removing CO 2, and (3) reabsorbing HCO 3 to help replenish the bicarbonate reserve. Figure 24. 7 1
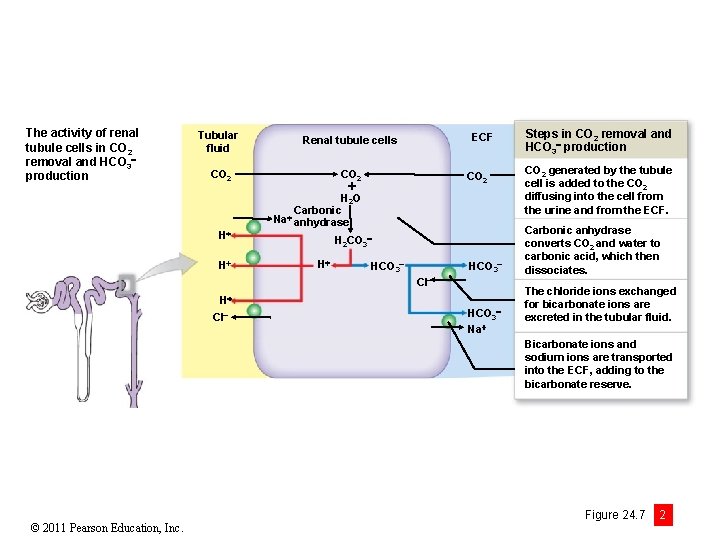
The activity of renal tubule cells in CO 2 removal and HCO 3 production Tubular fluid CO 2 H 2 O Carbonic Na anhydrase H H ECF Renal tubule cells CO 2 H 2 CO 3 H HCO 3 Cl HCO 3 Na Steps in CO 2 removal and HCO 3 production CO 2 generated by the tubule cell is added to the CO 2 diffusing into the cell from the urine and from the ECF. Carbonic anhydrase converts CO 2 and water to carbonic acid, which then dissociates. The chloride ions exchanged for bicarbonate ions are excreted in the tubular fluid. Bicarbonate ions and sodium ions are transported into the ECF, adding to the bicarbonate reserve. © 2011 Pearson Education, Inc. Figure 24. 7 2

Module 24. 7: Metabolic acid-base disorders • Metabolic alkalosis • Develops when large numbers of H+ are removed from body fluids • Rate of kidney H+ secretion declines • Tubular cells do not reclaim bicarbonate • Collecting system transports bicarbonate into urine and retains acid (HCl) in ECF © 2011 Pearson Education, Inc.

Module 24. 7: Metabolic acid-base disorders • Metabolic alkalosis (continued) • Accommodated by respiratory and renal responses • • Respiratory response • Decreased respiratory rate raises PCO 2 • H 2 O + CO 2 H 2 CO 3 H+ + HCO 3– Renal response • Occurs in PCT, DCT, and collecting system • H 2 O + CO 2 H 2 CO 3 H+ + HCO 3– • HCO 3– secreted into urine (in exchange for Cl–) • H+ actively reabsorbed into ECF © 2011 Pearson Education, Inc.
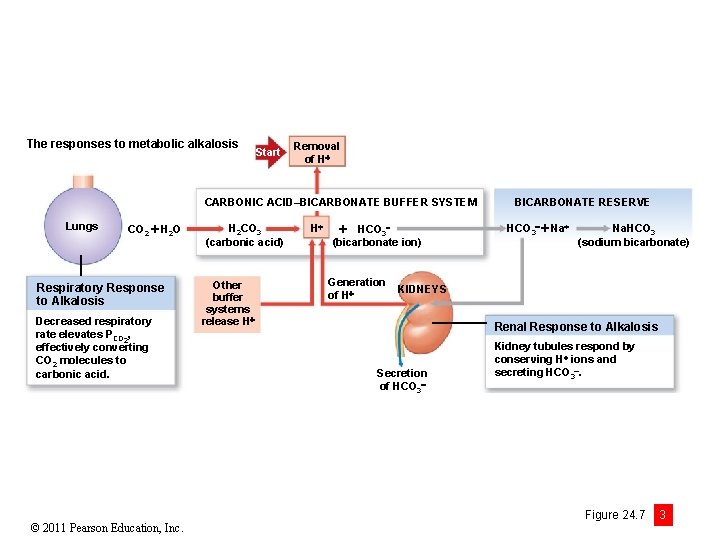
The responses to metabolic alkalosis Start Removal of H CARBONIC ACID–BICARBONATE BUFFER SYSTEM Lungs CO 2 H 2 O Respiratory Response to Alkalosis Decreased respiratory rate elevates PCO 2, effectively converting CO 2 molecules to carbonic acid. © 2011 Pearson Education, Inc. H 2 CO 3 (carbonic acid) Other buffer systems release H H HCO 3 (bicarbonate ion) Generation of H BICARBONATE RESERVE HCO 3 Na. HCO 3 (sodium bicarbonate) KIDNEYS Renal Response to Alkalosis Secretion of HCO 3 Kidney tubules respond by conserving H ions and secreting HCO 3. Figure 24. 7 3
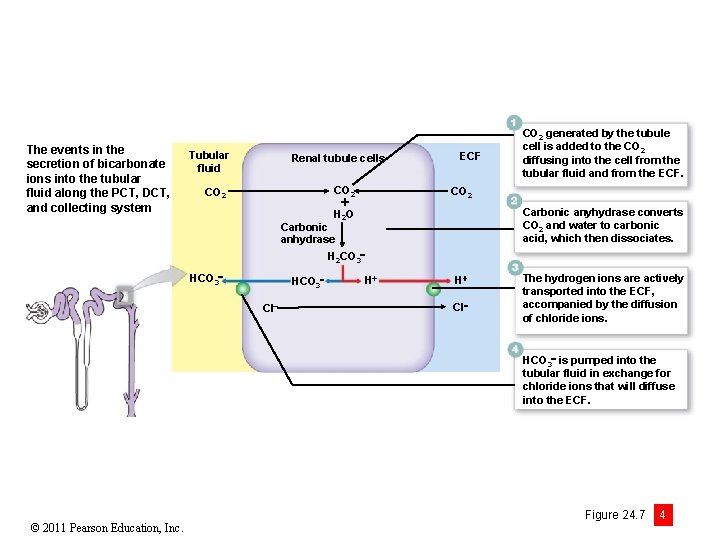
The events in the secretion of bicarbonate ions into the tubular fluid along the PCT, DCT, and collecting system Tubular fluid Renal tubule cells CO 2 H 2 O CO 2 ECF CO 2 generated by the tubule cell is added to the CO 2 diffusing into the cell from the tubular fluid and from the ECF. CO 2 Carbonic anyhydrase converts CO 2 and water to carbonic acid, which then dissociates. Carbonic anhydrase H 2 CO 3 HCO 3 Cl H H Cl The hydrogen ions are actively transported into the ECF, accompanied by the diffusion of chloride ions. HCO 3 is pumped into the tubular fluid in exchange for chloride ions that will diffuse into the ECF. © 2011 Pearson Education, Inc. Figure 24. 7 4

Module 24. 7 Review a. Describe metabolic acidosis. b. Describe metabolic alkalosis. c. lf the kidneys are conserving HCO 3– and eliminating H+ in acidic urine, which is occurring: metabolic alkalosis or metabolic acidosis? © 2011 Pearson Education, Inc.

CLINICAL MODULE 24. 8: Respiratory acid-base disorders • Respiratory acidosis • CO 2 generation outpaces rate of CO 2 elimination at lungs • Shifts bicarbonate buffer system toward generating more carbonic acid • H 2 O + CO 2 H 2 CO 3 H+ + HCO 3– • HCO 3– goes into bicarbonate reserve • H+ must be neutralized by any of the buffer systems • Respiratory (increased respiratory rate) • Renal (H+ secreted and HCO 3– reabsorbed) • Proteins (bind free H+) © 2011 Pearson Education, Inc.
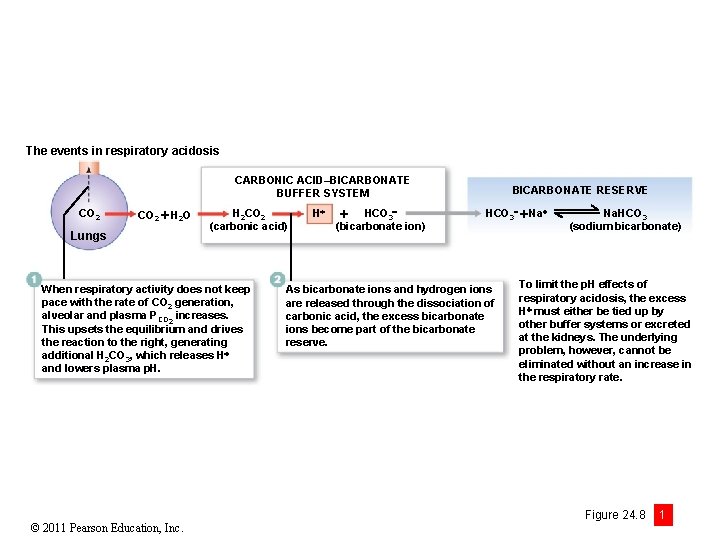
The events in respiratory acidosis CARBONIC ACID–BICARBONATE BUFFER SYSTEM CO 2 H 2 O Lungs H 2 CO 2 (carbonic acid) When respiratory activity does not keep pace with the rate of CO 2 generation, alveolar and plasma PCO 2 increases. This upsets the equilibrium and drives the reaction to the right, generating additional H 2 CO 3, which releases H and lowers plasma p. H. © 2011 Pearson Education, Inc. H HCO 3 (bicarbonate ion) BICARBONATE RESERVE HCO 3 Na As bicarbonate ions and hydrogen ions are released through the dissociation of carbonic acid, the excess bicarbonate ions become part of the bicarbonate reserve. Na. HCO 3 (sodium bicarbonate) To limit the p. H effects of respiratory acidosis, the excess H must either be tied up by other buffer systems or excreted at the kidneys. The underlying problem, however, cannot be eliminated without an increase in the respiratory rate. Figure 24. 8 1
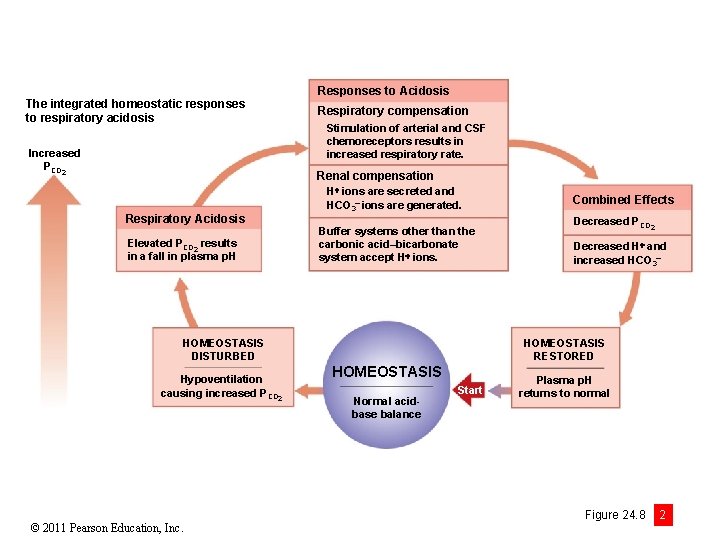
The integrated homeostatic responses to respiratory acidosis Increased PCO 2 Responses to Acidosis Respiratory compensation Stimulation of arterial and CSF chemoreceptors results in increased respiratory rate. Renal compensation Respiratory Acidosis Elevated PCO 2 results in a fall in plasma p. H H ions are secreted and HCO 3 ions are generated. Buffer systems other than the carbonic acid–bicarbonate system accept H ions. © 2011 Pearson Education, Inc. Decreased PCO 2 Decreased H and increased HCO 3 HOMEOSTASIS RESTORED HOMEOSTASIS DISTURBED Hypoventilation causing increased PCO 2 Combined Effects HOMEOSTASIS Normal acidbase balance Start Plasma p. H returns to normal Figure 24. 8 2

CLINICAL MODULE 24. 8: Respiratory acid -base disorders • Respiratory alkalosis • CO 2 elimination at lungs outpaces CO 2 generation rate • Shifts bicarbonate buffer system toward generating more carbonic acid • H+ + HCO 3– H 2 CO 3 H 2 O + CO 2 • • H+ removed as CO 2 exhaled and water formed Buffer system responses • Respiratory (decreased respiratory rate) • Renal (HCO 3– secreted and H+ reabsorbed) • Proteins (release free H+) © 2011 Pearson Education, Inc.
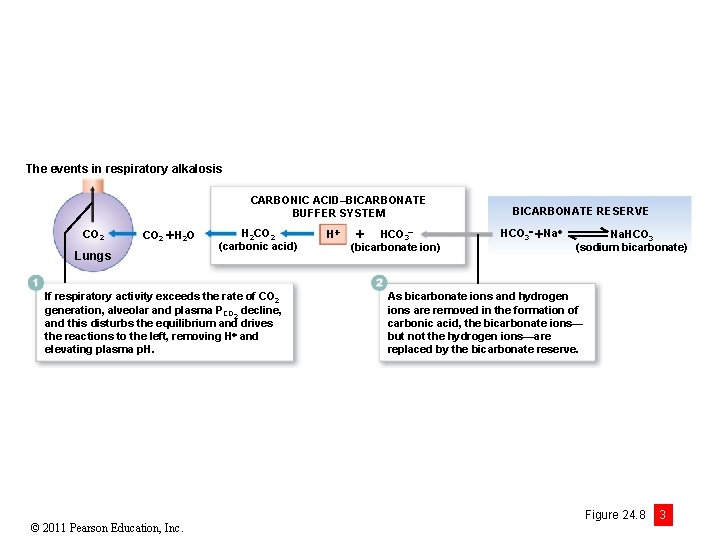
The events in respiratory alkalosis CARBONIC ACID–BICARBONATE BUFFER SYSTEM CO 2 H 2 O Lungs H 2 CO 2 (carbonic acid) If respiratory activity exceeds the rate of CO 2 generation, alveolar and plasma PCO 2 decline, and this disturbs the equilibrium and drives the reactions to the left, removing H and elevating plasma p. H. © 2011 Pearson Education, Inc. H HCO 3 (bicarbonate ion) BICARBONATE RESERVE HCO 3 Na. HCO 3 (sodium bicarbonate) As bicarbonate ions and hydrogen ions are removed in the formation of carbonic acid, the bicarbonate ions— but not the hydrogen ions—are replaced by the bicarbonate reserve. Figure 24. 8 3
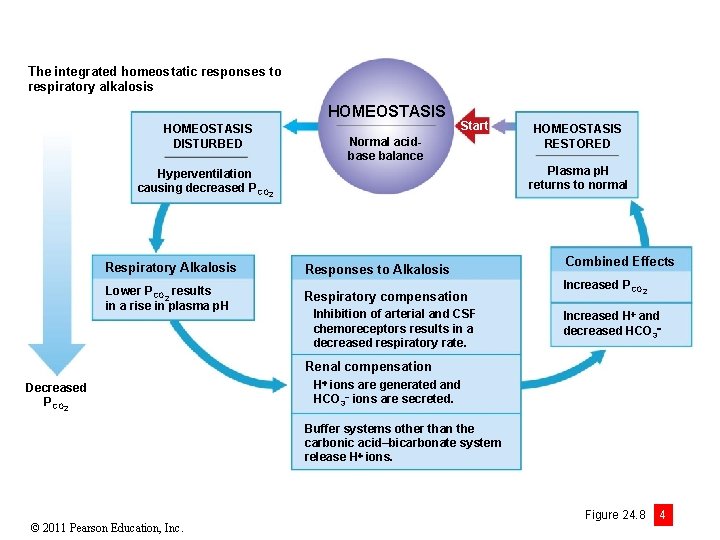
The integrated homeostatic responses to respiratory alkalosis HOMEOSTASIS DISTURBED Start Normal acidbase balance HOMEOSTASIS RESTORED Plasma p. H returns to normal Hyperventilation causing decreased PCO 2 Respiratory Alkalosis Responses to Alkalosis Lower PCO 2 results in a rise in plasma p. H Respiratory compensation Inhibition of arterial and CSF chemoreceptors results in a decreased respiratory rate. Combined Effects Increased PCO 2 Increased H and decreased HCO 3 Renal compensation Decreased PCO 2 H ions are generated and HCO 3 ions are secreted. Buffer systems other than the carbonic acid–bicarbonate system release H ions. © 2011 Pearson Education, Inc. Figure 24. 8 4

CLINICAL MODULE 24. 8 Review a. Define respiratory acidosis and respiratory alkalosis. b. What would happen to the plasma PCO 2 of a patient who has an airway obstruction? c. How would a decrease in the p. H of body fluids affect the respiratory rate? © 2011 Pearson Education, Inc.
- Slides: 69