Fundamentals of Fluid and Electrolyte Balance Parenteral Solutions

Fundamentals of Fluid and Electrolyte Balance Parenteral Solutions ADN 136 Fall 09

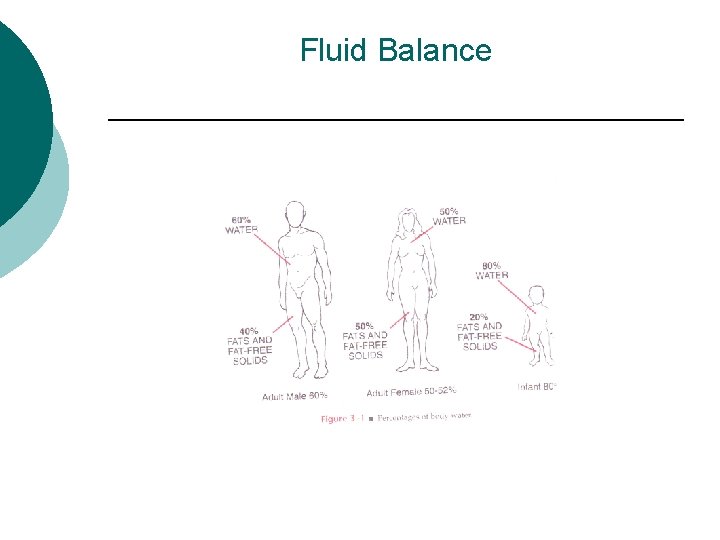
Fluid Balance Body fluid is body water in which electrolytes are dissolved ¡ Bodywater makes up ¡ l l l 60% of Total Body weight in young men 50 -55% in women 70 -80% in infants
Fluid Balance

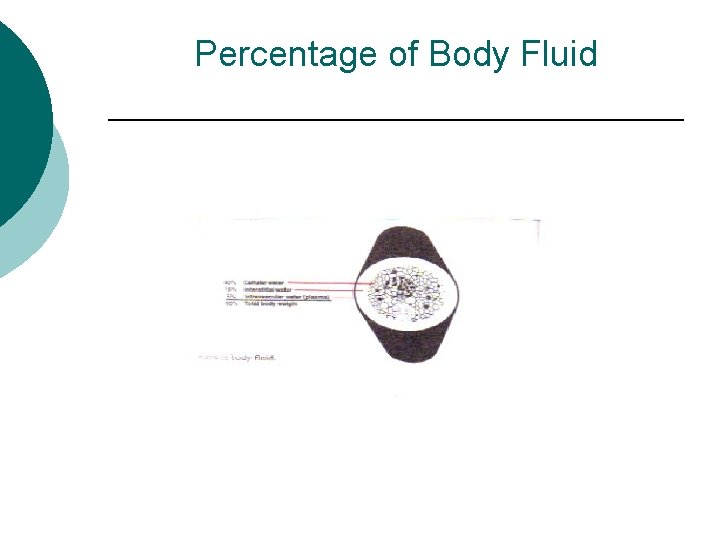
Fluid Balance (cont) ¡ Homeostasis- Dependent on fluid and electrolyte intake physiologic factors, disease state factors, external environmental factors and pharmacologic intervention. l l Intracellular fluid (ICF) water in the cells = 40% Extracellular fluid (ECF) fluid out side the cells = 20% ¡ 15% in tissue space (interstitial) outside the blood vessel, between cells ¡ 5% in plasma (intravascular space)
Percentage of Body Fluid

Fluid Balance Normal intake 1 -3 L/day ¡ 200 -300 ml produced by oxidation ¡ Normal intake and output will balance approximately every 72 hours ¡

Fluid Balance ¡ Elimination of fluids is considered l Sensible (measurable) loss Skin, Kidneys, Bowels, lungs lose fluid ¡ 300 -500 ml lost through lungs every 24 hrs. ¡ 500 ml lost with perspiration ¡ l Insensible (not measurable) loss ¡ Considered to be 500 -1000 ml/day l Lost through GI tract only 100 -200 ml/day

Fluid Balance (cont) Loss from Diarrhea or intestinal fistula ¡ Significant sweat loss when body temp >101 F-38. 3 C or room temp > 90 F ¡ When respirations > 20/min ¡ Formula to calculate insensible loss ¡ l 300 -400 ml/m 2/day

Fluid Function The fluid in the body has the following function: 1. Maintains blood volume 2. Regulates body temperature 3. Transports material to and from cells 4. Serves as an aqueous medium for cellular metabloism 5. Assists digestion of food through hydrolysis 6. Acts as a solvent in which solutes are available for cell function 7. Serves as a medium for the excretion of waste

Fluid Transport ¡ 4 transport mechanisms l Passive transport Passive diffusion¡ Osmosis ¡ Filtration ¡ l Active transport

Fluid Transport ¡ Passive transport- non carrier mediated transport- Fluid moves through membranes with out the expenditure of energy l l Passive diffusion - movement of water and other elements in all directions from high concentration to low concentration Osmosis – passage of water from low particle concentration toward one of higher particle concentration ¡ Normal osmolarity – 280 -295 m. Osm/L l Osmolarity of ICF and ECF is always equal

Fluid Transport (cont) ¡ Filtration – the transfer of water and a dissolved substance from a region of high pressure to a region of low pressure. Force behind it is hydrostatic pressure (the pressure of water at rest) l Pumping heat provides hydrostatic pressure in the movement of water and electrolytes from the arterial capillary bed to the interstitial fluid.

Fluid Transport (cont) Plasma protein creates and osmotic pressure at the capillary membrane, preventing fluid from plasma leaking into interstitial spaces ¡ Osmotic pressure (created within the plasma) keeps water in the vascular system ¡

Fluid Transport (cont) ¡ Starling’s law of capillaries l Under normal circumstances fluid filtered out of the arterial end of a capillary bed and reabsorbed at the venous end is exactly the same, creating a state of near equilibrium

Fluid Transport (cont) ¡ Active Transport – acts as a concentration gradient l l ATP – released from the cell to enable substances to acquire the energy needed to pass through the cell membrane Active Transport is vital for maintaining the unique composition of both the intracellular and intracellular compartments

Tonicity of Solution ¡ Isotonic -. 9% saline, 5% dextrose same as body fluids l Osmolarity of 250 -375 m. Osm/L l Remains within the ECF space l Used to expand ECF compartment l

Isotonic Solution

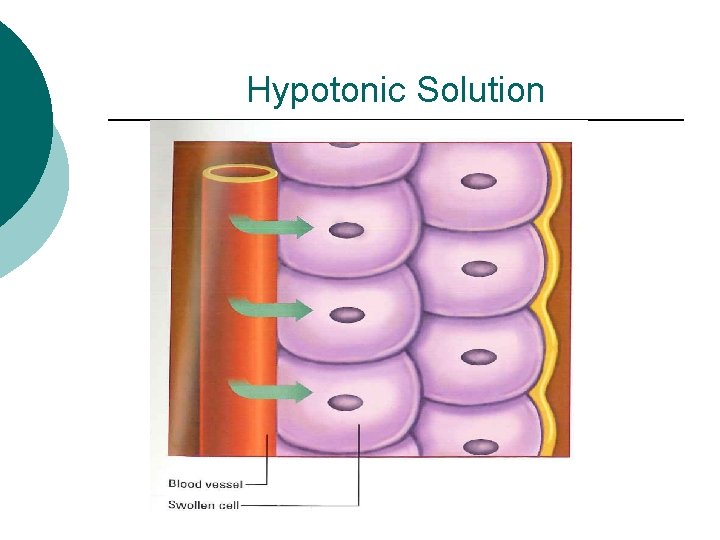
Tonicity of Solution ¡ Hypotonic – contains less salt than the intracellular space 2. 5% DW l Osmolarity below 250 m. Osm/L ¡ Hydrates cells ¡ Depletes the circulatory system
Hypotonic Solution

Tonicity of Solution ¡ Hypertonic – causes water from within a cell to move to the ECF compartment l l Osmolarity of 375 m. Osm/l or greater Used to replace electrolytes Used to shift EDF from interstitial tissue to plasma D 5 W, . 9 Normal Saline

Hypertonic Solution

Homeostatic Mechanism ¡ Regulation of body water is maintained l l Exogenous sources - Intake of food & Fluids (nurse’s responsibility) Endogenous sources – produce with in the body through chemical exidation process (various body systems responsible)

Homeostatic Mechanisms ¡ Renal System – Kidneys filter 170 L l of plasma/day and excrete 1. 5 L of urine l l Regulation of fluid volume and osmolarity by selective retention and secretion of body fluid Regulation of electrolyte levels by selective retention of needed substances and excretion of unneeded substances Regulation of p. H of ECF by excretion or retention of hydrogen ions Excretion of metabolic wastes (primarily acids) and toxic substances

Homeostatic Mechanism ¡ Cardiovascular System – l l l Pumping action of the heart provides circulation of blood through the kidneys under pressure Allow urine to form Renal perfusion makes renal function possible

Homeostatic Mechanism ¡ Lymphatic system – l l Serves as an adjunct to the cardio vascular system by removing excess interstitial fluid (lymph) and returning it to the circulatory system Prevents fluid overload

Homeostatic Mechanism ¡ Respiratory System l l Lungs are vital for maintaining homeostasis and constitute one of the main regulatory orgnas of fluid and acid base balance Functions of the lungs Regulation of metabolic alkalosis by compensatory hypoventilation ¡ Regulation of metabolic acidosis by causing compensatory hyperventilation ¡ Removal of 300 -500 ml of water daily through exhalation ¡

Homeostatic Mechanism ¡ Nervous system l ¡ Master controller in fluid and electrolyte balance through the regulation of sodium and water Endocrine system l Responsible for aiding homeostasis through production of various hormones Antidiuretic hormones (ADH) ¡ Parathyroid Hormones ¡ Aldosterone ¡ Epinephrine ¡

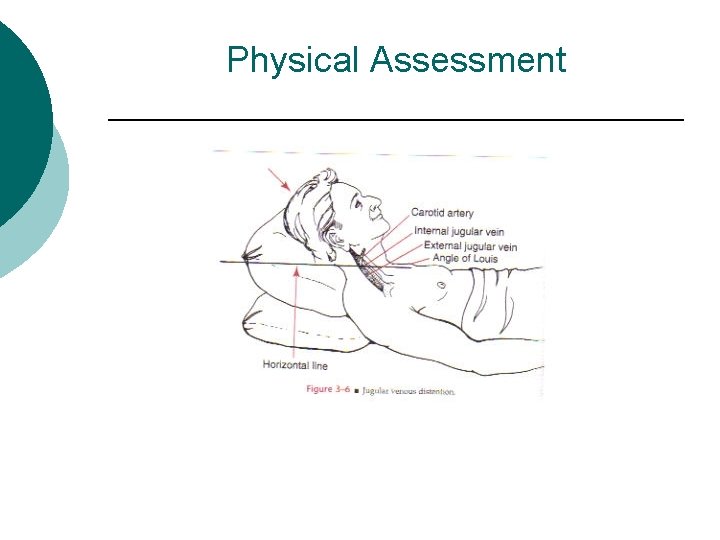
Physical Assessment Vital signs, infusion rate of IV fluids, intake and output. ¡ Neurological - Changes in orientation, irritability, lethargy, confusion, seizures or coma ¡ Cardiovascular – ¡ l l Quality and rate of pulse Peripheral vein filling Orthostatic hypertension Distended or Flat neck veins
Physical Assessment

Physical Assessment ¡ Respiratory l Changes in respiratory rate Tachypnea > 20/min or dysphnea indicate excess Fluid Volume (FVE) ¡ Moist crackles (FVE) ¡ Shallow Slow breathing- Metabolic Acidosis ¡ Deep rapid Breathing – Metabolic Alkalosis ¡
Physical Assessment ¡ Skin Appearance & Temperature l l Access skin turgor Appearance of the tongue

Physical Assessment ¡ Body Weight l l l Weigh Daily – better indicator than I&O records Loss or gain of 1 kg indicates a loss or gain of 1 L of body fluid 15% flucation is considered sever

Fluid Volume Imbalance ¡ ¡ Fluid Volume Deficit Common Causes of Isotonic Dehydration l l l Hemorrhage resulting in loss of fluid, electrolytes, proteins and blood cells resulting in inadequate vascular volume Gastrointestinal losses Fever, environmental heat, profuse sweating Burns Diuretics Third spaced fluids

Fluid Volume Imbalance ¡ Causes of Hypertonic Fluid Dehydration l l Inadequate fluid intake Decreased water intake results in ECF solute concentration and leads to cellular dehydration

Fluid Volume Imbalance ¡ Fluid Volume Excess l ¡ Causes of isotonic over hydration l l ¡ Primary cause – Cardiovascular dysfunction secondary to an increase in total body sodium content Renal failure leading to decrease excretion of water and sodium Heart failure leading to stasis of blood Excess fluid intake of isotonic IV solution High corticosteroid levels High Aldosterone levels

Fluid Volume Imbalance ¡ Common causes of Hypotonic Over hydration (Water intoxication) l l l ¡ More fluid is gained than solute Serum osmolality falls causing cells to swell Repeated water enemas Overuse of hypotonic IV fluids Ingestion of inappropriately prepared formula SIADH causes kidneys to retain large amounts of water without sodium Treatment- sodium and fluid restriction, diuretics, treat underlying cause.

Electrolyte Balance ¡ ¡ ¡ Major electrolytes in body fluid are sodium, potassium, calcium, magnesium, chloride, phosphorus and bicarb Expressed in meq/liter. Measures chemical activity or combining power rather than weight Each water compartment of the body contains electrolytes l Concentration and composition vary from compartment to compartment

Electrolyte Balance (cont) ¡ Physiological role of electrolytes l l l Maintaining electroneutrality in fluid compartments Mediating enzyme reactions Altering cell membrane permeability Regulating muscle contraction and relaxation Regulating nerve impulse transmission Influencing blood clotting time

Electrolyte Balance (cont) Sodium- 135 -145 m. Eq/L ¡ Physiologic role of sodium: ¡ l l Regulation of fluid distribution in body: water follows sodium Maintenance of body fluid osmolarity Promotion of neuromuscular response: Transmission of nerve and muscle impulses depends on sodium, gradient between ECF and ICF Regulation of acid-base balance: Sodium combines with chloride and bicarbonate to alter p. H

Electrolyte Balance (cont) ¡ ¡ ¡ Sodium represents 90% of the extracellular cations Serum plasma levels of electrolytes are important in the assessment and management of patients with electrolyte imbalances Normal daily requirement 100 m. Eq Hyponatermia is a common complication of adrenal insufficiency Hypernatermia – Serum Sodium excess great that 145 m. Eq/L can occur with deprivation of water

Electrolyte Balance (cont) ¡ Signs and Symptoms - Marked thirst, elevated body temperature, swollen tongue. l ¡ Chronic Hyponatremia: impaired sensation of taste, anorexia, muscle cramps, feeling of exhaustion, apprehension, feeling of impending doom and focal weakness. Treatment: Gradually lower seum sodium level by infusion of hypotonic electrolyte solution. 45 Normal Saline or D 5 W. Level lowered no more than 15 m. Eq/L in 8 hr.

Electrolyte Balance (cont) ¡ Potassium: Physiological role l l l Regulation of fluid volume within the cell Promotion of nerve impulse transmission Contraction of skeletal smooth and cardiac muscle Control of hydrogen ion concentration, acidbase balance Role of enzyme action for cellular energy production.

Electrolyte Balance (cont) Potassium is an intracellular electrolyte with 98% in ICF and 2% in the ECF ¡ Acquired thru diet and must be ingested daily ¡ Daily requirement is 40 m. Eq ¡ Involved in muscle activity and transmission of nerve impulses. ¡

Electrolyte Balance (cont) ¡ Hypokalemia (cont) – Can cause alkalosis l l S&S fatigue, muscle weakness, anorexia, nausea and vomiting, irregularity Treatment – at level less than 3. 5 m. Eq/L replacement must be slow to prevent hyperkalemia

Electrolyte Balance (cont) ¡ Hyperkalemia- Serum plasma level greater than 5. 5 m. Eq/L l l l ¡ Increased intake of potassium Decreased urinary excretion Movement out of cells into extra cellular space. Signs & Symptoms l l l Changes shown on ECG Vague muscle weakness Flaccid paralysis Anxiety Nausea and vomiting Cramping and diaherrea

Electrolyte Balance (cont) ¡ Calcium: Physiological role l l ¡ Maintaining skeletal elements; calcium is needed for strong bones and teeth Regulating neuromuscular activity Influencing enzyme activity Converting prothrombin to thrombin, a necessary part of clotting. 99% resides in bones and teeth

Electrolyte Balance (cont) ¡ Hypocalcemia: reduction in total body calcium levels l ¡ S & S: l ¡ Because of increase calcium loss, reduced intake secondary to altered intestinal absorption, altered regulation hypoparathyroidism Numbness of fingers, muscle cramps, hyperactive deep tendon reflexes, positive Trousseaus’s sign and Chevostek’s sign Treatment with Calcium Gluconate oral or IV

Electrolyte Balance (cont) Hyperclacemia: Excessive release of calcium from bone ¡ S & S Neuromuscular symptoms, lethargy, bone pain, flank pain, pathological fractures, constipation, anorexia, N & V, Stone formation. ¡

Electrolyte Balance (cont) ¡ Magnesium: Physiological role l l l ¡ Enzyme action Regulation of neuromuscular activity Regulation of electrolyte balance, including facilitating transport of sodium and potassium across cell membranes, influencing the utilization of calcium, potassium, and protein. A major intracellular electrolyte

Electrolyte Balance (cont) ¡ ¡ Hypomagnesemia: often overlooked in critically ill patients Results from: l l l l Chronic alcoholism Malabsorption syndrome Prolonged malnutrition or starvation Prolonged diarrhea Acute pancreatitis Administration of magnesium-free solutions for more than one week Prolonged NG tube suctioning

Electrolyte Balance (cont) ¡ S&S l Neuromuscular symptoms Hyperactive reflexes, ¡ Coarse tremors ¡ Muscle cramps ¡ Positive Chvostek’s and Trousseau’s signs ¡ Seizures ¡ Paresthesia of the feet and legs ¡ Painfully cold hands and feet ¡ Disorientation dysrhythmias tachycardia and indreased potential for digitalis toxicity ¡

Electrolyte Balance (cont) ¡ ¡ Hypermagnesemia: renal failure, addison’s disease, and inadequate excretion of magnesium by kidneys S & S: l l l Neuromuscular symptoms ¡ Flushing and sense of skin warmth Lethargy Sedation Hypoactive deep tendon reflexes, Depressed respiration Weak or absent new born cry

Electrolyte Balance (cont) ¡ Phosphorus: physiologic role: l l l Essential to all cells Role in metabolism of proteins, carbohydrates and fats Essential to energy, necessary in the formation of high energy compounds adenosine triphosphate (ATP) and adenosine diphosphate (ADP) As a cellular building block, it is the backbone of nucleic acids and is essential to cell membrane formation’ Delivery of oxygen; functions information of red blood cell enzyme.

Electrolyte Balance (cont) ¡ ¡ Approximately 80% is contained in the bones and teeth 20% is abundant in the ICF Plays and important role in delivery of oxygen to tissues by regulating the level of 2, 3 -DPG Hyphphosphatemia: results from l l l Overzealous refeeding, TPN administered without adequate phosphorus Malabsorption Alcohol withdrawal Vomiting, chronic diarrhea, and malabsorption syndromes

Electrolyte Balance (cont) ¡ Other Electrolyte imbalance: l l l Hyperphosphatemia Hypochloremia Hyperchloremia
- Slides: 55