FLUID ELECTROLYTE AND ACIDBASE BALANCE FLUID ELECTROLYTE AND

FLUID, ELECTROLYTE, AND ACID-BASE BALANCE

FLUID, ELECTROLYTE, AND ACID-BASE BALANCE • Cell function depends not only on a continuous supply of nutrients and removal of metabolic wastes, but also on the physical and chemical homeostasis of the surrounding fluids

BODY FLUIDS Body Water Content • Total body water is a function of age, body mass, and body fat • Due to their low body fat and bone mass, infants are about 73% water • The body water content of men is about 60%, but since women have relatively more body fat and less skeletal muscle than men, theirs is about 50% • Of all body tissues, adipose tissue is least hydrated (up to about 20% water) • Skeletal muscle is about 65% water • Body water declines throughout life, ultimately comprising about 45% of total body mass in old age

BODY FLUIDS Fluid Compartments • There are two main fluid compartments of the body: – Intracellular fluid compartment: ICF • • Contains slightly less than two-thirds by volume Cells – Extracellular fluid compartment: ECF • • The remaining third Outside cells Constitutes the body’s internal environment but external environment of the cell Two Sub-compartments: – – • Blood plasma: fluid portion of blood Interstitial fluid (IF): fluid in the microscopic spaces between tissue cells Numerous other examples that are distinct from both plasma and interstitial fluid—lymph, cerebrospinal fluid, humors of the eye, synovial fluid, serous fluid, secretions of the gastrointestinal tract—but most of these are similar to interstitial fluid (IF) and are usually considered part of it
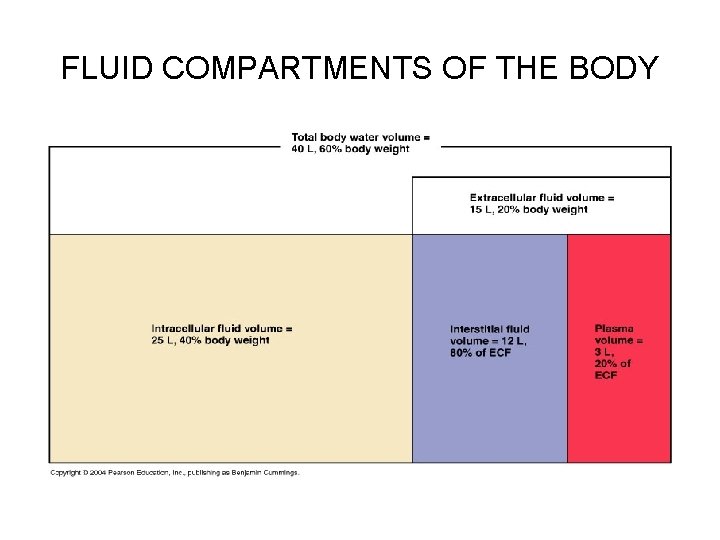
FLUID COMPARTMENTS OF THE BODY

Composition of Body Fluids Electrolytes and Nonelectrolytes • Water serves as the universal solvent in which a variety of solutes are dissolved: – Solutes may be classified broadly as: • Electrolytes: dissociate in water to ions – Include inorganic salts, both inorganic and organic acids and bases, and some proteins – Conduct electric current • Nonelectrolytes: do not dissociate in water – Have bonds (usually covalent) that prevent them from dissociating in solution – Include most organic molecules – Carry no net electrical charge when dissolved in water • Although all dissolve solutes contribute to the osmotic activity of a fluid: – Electrolytes have greater osmotic power because they dissociate in water and contribute at least two particles to solution • Examples: – Na. Cl → Na+ + Cl- (electrolyte: two particles) – Mg. Cl 2 → Mg 2+ + 2 Cl- (electrolyte: three particles) – Glucose → Glucose (nonelectrolyte: one particle)

Composition of Body Fluids Electrolytes and Nonelectrolytes • Regardless of the type of solute particle, water moves according to osmotic gradients—from an area of lesser osmolality (less dissolved substances per unit of solvent) to an area of greater osmolality (more dissolved substances per unit of solvent) – Osmolality: the number of solute particles dissolved in one liter (1000 g) of water • Reflects the solution’s ability to cause osmosis • Osmolarity: total concentration of all solute particles in a solution – Thus, electrolytes have the greatest ability to cause fluid shifts • Electrolyte concentrations of body fluids are usually expressed in milliequivalents per liter (m. Eq/L) – A measure of the number of electrical charges in 1 liter of solution
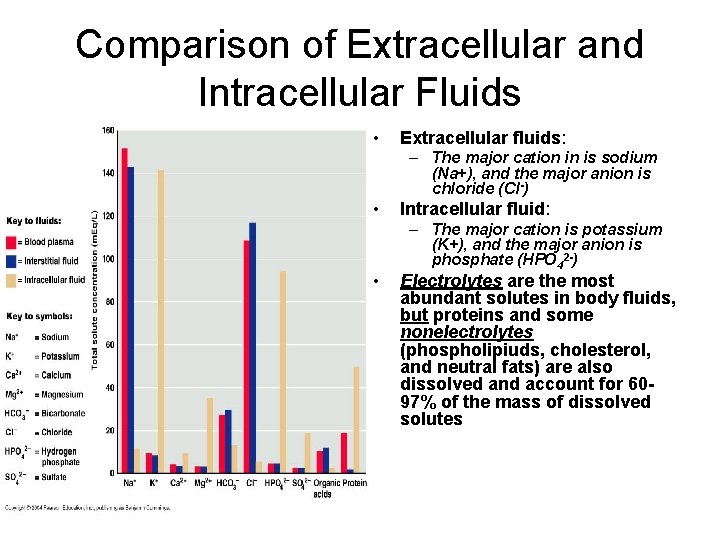
Comparison of Extracellular and Intracellular Fluids • Extracellular fluids: – The major cation in is sodium (Na+), and the major anion is chloride (Cl-) • • Intracellular fluid: – The major cation is potassium (K+), and the major anion is phosphate (HPO 42 -) Electrolytes are the most abundant solutes in body fluids, but proteins and some nonelectrolytes (phospholipiuds, cholesterol, and neutral fats) are also dissolved and account for 6097% of the mass of dissolved solutes
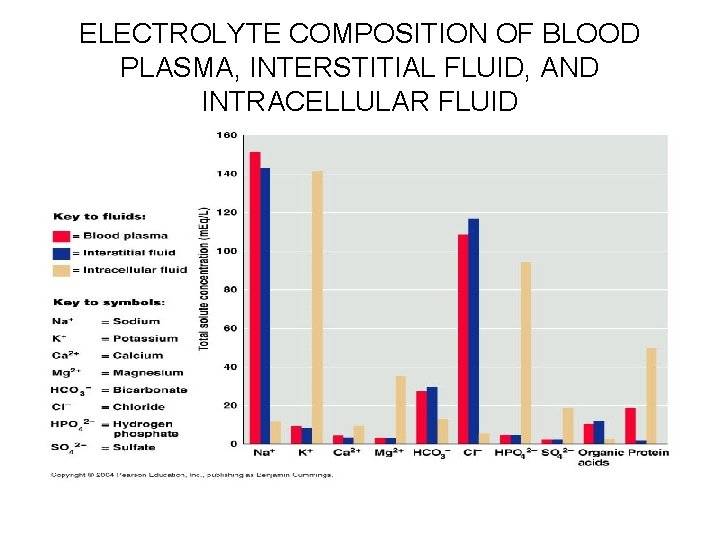
ELECTROLYTE COMPOSITION OF BLOOD PLASMA, INTERSTITIAL FLUID, AND INTRACELLULAR FLUID

Fluid Movement Among Compartments • The continuous exchange and mixing of body fluids are regulated by osmotic pressure (pressure that develops when two solutions of different concentrations are separated by a semipermeable membrane) and hydrostatic pressures (pressures exerted on liquids) • Although water moves freely between the compartments along osmotic gradients, solutes are unequally distributed because of their size, electrical charge, or dependence on active transport – Anything that changes solute concentration in any compartment leads to net water flows

Fluid Movement Among Compartments • Exchanges between plasma and Interstitial Fluid (IF) occur across capillary membranes – Nearly protein-free plasma is forced out of the blood by hydrostatic pressure, and almost completely reabsorbed due to colloid osmotic (oncotic) pressure of plasma proteins (review blood capillaries Chapter) – Movement of water between the interstitial fluid and intracellular fluid is more complex because of the selective permeability of cell membranes • As a rule: – Involves substantial two-way osmotic (water) flow that is equal in both directions – Ion fluxes are restricted and , in most cases, ions move selectively by active transport – Movement of nutrients, respiratory gases, and wastes are typically unidirectional
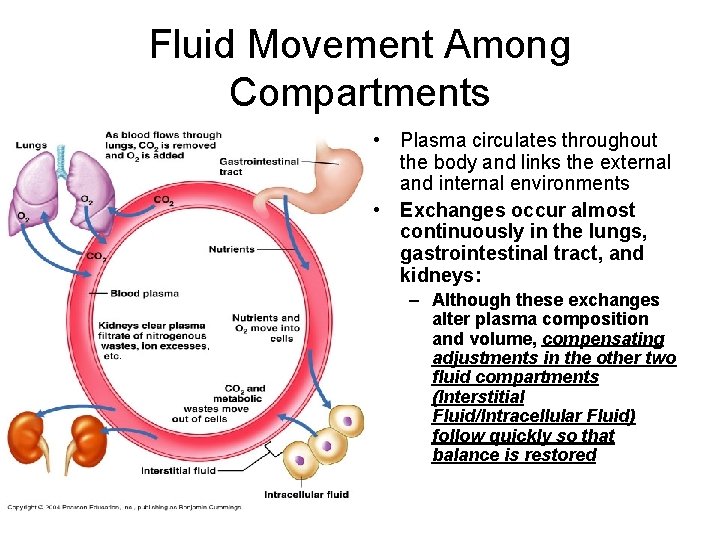
Fluid Movement Among Compartments • Plasma circulates throughout the body and links the external and internal environments • Exchanges occur almost continuously in the lungs, gastrointestinal tract, and kidneys: – Although these exchanges alter plasma composition and volume, compensating adjustments in the other two fluid compartments (Interstitial Fluid/Intracellular Fluid) follow quickly so that balance is restored
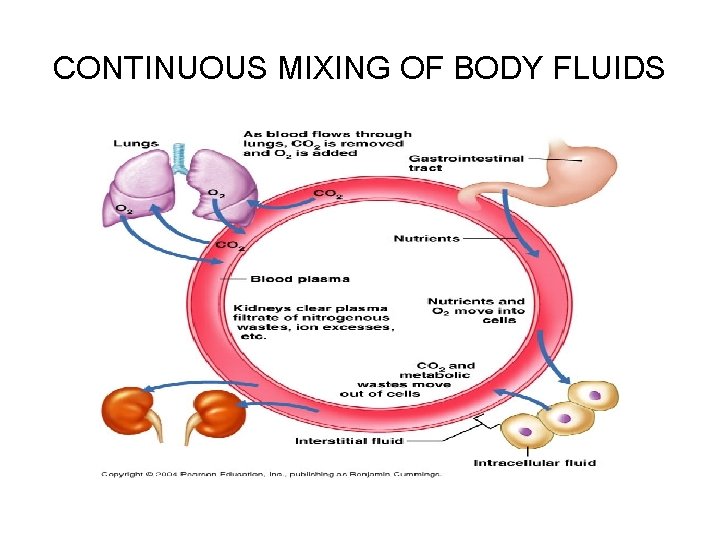
CONTINUOUS MIXING OF BODY FLUIDS
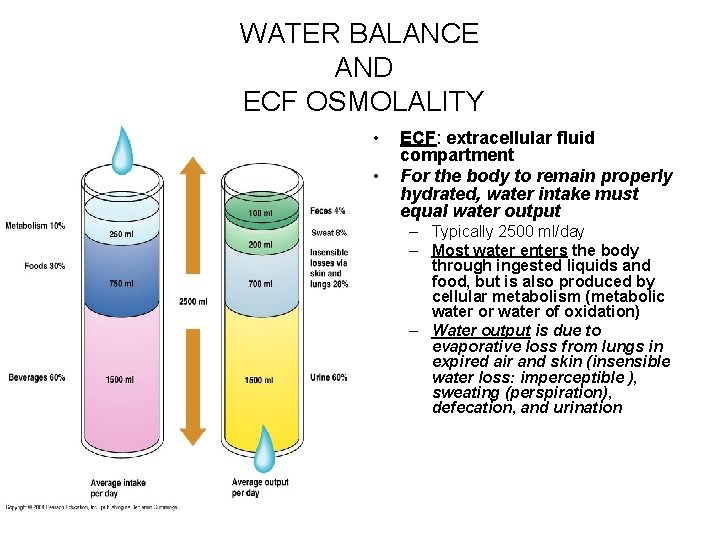
WATER BALANCE AND ECF OSMOLALITY • • ECF: extracellular fluid compartment For the body to remain properly hydrated, water intake must equal water output – Typically 2500 ml/day – Most water enters the body through ingested liquids and food, but is also produced by cellular metabolism (metabolic water or water of oxidation) – Water output is due to evaporative loss from lungs in expired air and skin (insensible water loss: imperceptible ), sweating (perspiration), defecation, and urination
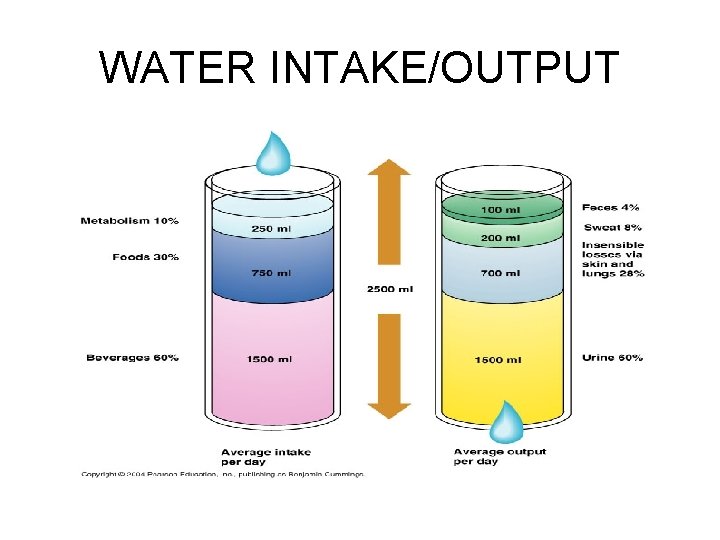
WATER INTAKE/OUTPUT

Regulation of Water Intake • The thirst mechanism is triggered by a increase in plasma osmolarity (2 -3%), which results in a dry mouth (less fluid leaves the bloodstream) and excites the hypothalamic thirst center – Less saliva is produced – Hypothalamic thirst center is stimulated when its osmoreceptors lose water by osmosis to the hypertonic ECF, or are excited by baroreceptor (pressure receptors in blood vessels) inputs, angiotensin (vasopressor-antidiuretic produced when renin is released by the kidneys), or other stimuli • Cause a subjective sensation of thrist • Free salty snacks in bar
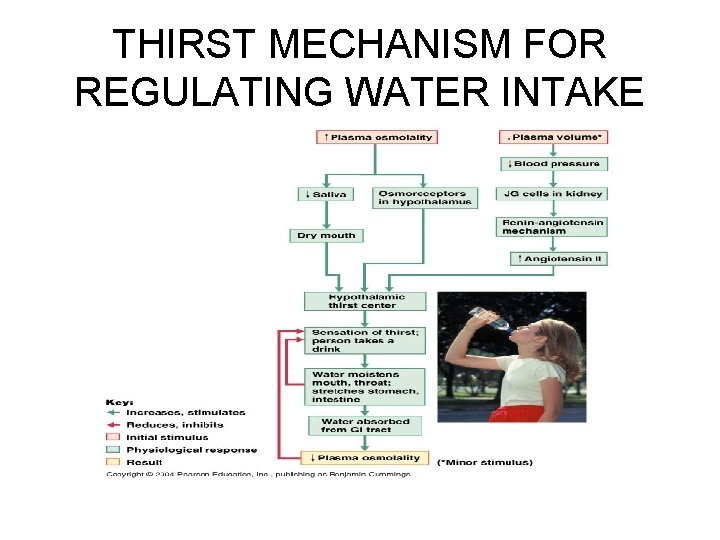
THIRST MECHANISM FOR REGULATING WATER INTAKE

Regulation of Water Intake • Thirst is quenched as the mucosa of the mouth is moistened, and continues with distention of the stomach and intestine, resulting in inhibition of the hypothalamus thirst center
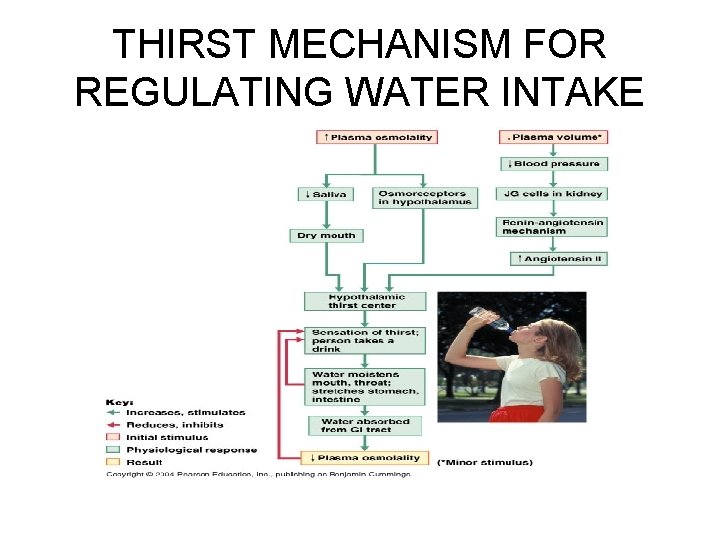
THIRST MECHANISM FOR REGULATING WATER INTAKE

Regulation of Water Output • Drinking is necessary since there is obligatory water loss due to the insensible (imperceptible/gradual) water losses – Even the most heroic conservation efforts by the kidneys cannot compensate for zero water intake • Beyond obligatory water losses, solute concentration and volume of urine depend on fluid intake
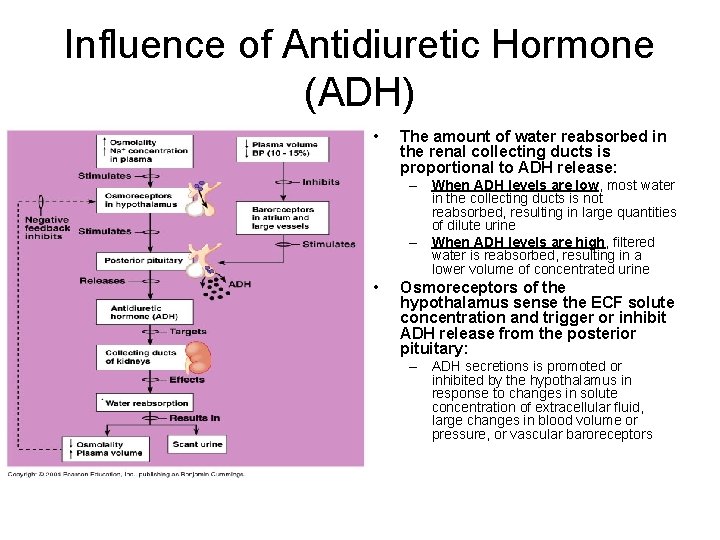
Influence of Antidiuretic Hormone (ADH) • The amount of water reabsorbed in the renal collecting ducts is proportional to ADH release: – When ADH levels are low, most water in the collecting ducts is not reabsorbed, resulting in large quantities of dilute urine – When ADH levels are high, filtered water is reabsorbed, resulting in a lower volume of concentrated urine • Osmoreceptors of the hypothalamus sense the ECF solute concentration and trigger or inhibit ADH release from the posterior pituitary: – ADH secretions is promoted or inhibited by the hypothalamus in response to changes in solute concentration of extracellular fluid, large changes in blood volume or pressure, or vascular baroreceptors

MECHANISMS AND CONSEQUENCES OF ADH RELEASE
MECHANISMS REGULATING SODIUM AND WATER BALANCE HELP MAINTAIN BLOOD PRESSURE HOMEOSTASIS
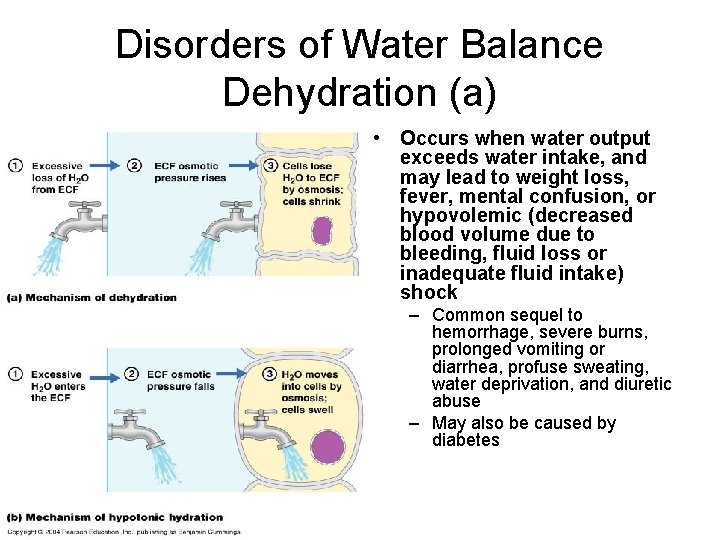
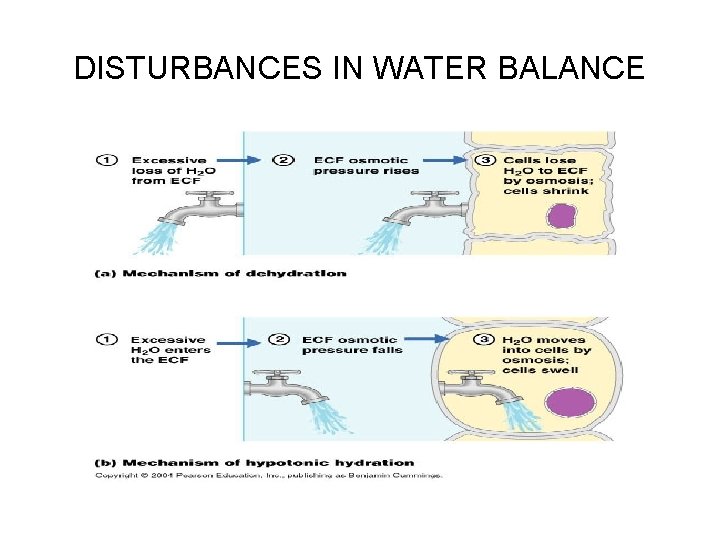
Disorders of Water Balance Dehydration (a) • Occurs when water output exceeds water intake, and may lead to weight loss, fever, mental confusion, or hypovolemic (decreased blood volume due to bleeding, fluid loss or inadequate fluid intake) shock – Common sequel to hemorrhage, severe burns, prolonged vomiting or diarrhea, profuse sweating, water deprivation, and diuretic abuse – May also be caused by diabetes
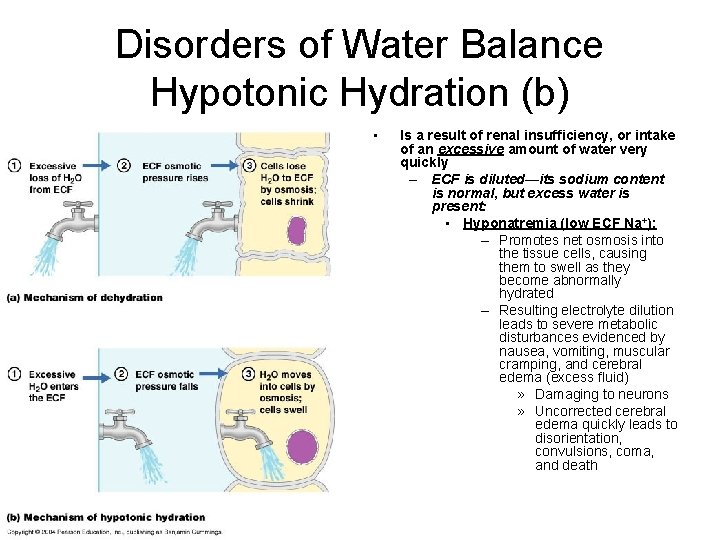
Disorders of Water Balance Hypotonic Hydration (b) • Is a result of renal insufficiency, or intake of an excessive amount of water very quickly – ECF is diluted—its sodium content is normal, but excess water is present: • Hyponatremia (low ECF Na+): – Promotes net osmosis into the tissue cells, causing them to swell as they become abnormally hydrated – Resulting electrolyte dilution leads to severe metabolic disturbances evidenced by nausea, vomiting, muscular cramping, and cerebral edema (excess fluid) » Damaging to neurons » Uncorrected cerebral edema quickly leads to disorientation, convulsions, coma, and death
DISTURBANCES IN WATER BALANCE
Disorders of Water Balance Edema • Is the accumulation of fluid in the interstitial space, which may impair tissue function: – Caused by any event that: • Steps up the flow of fluid out of the blood: – Blood pressure – Increased capillary permeability – Inflammation) • Or hinders its return: – Imbalance on the two sides of the capillary membranes – Hypoproteinemia: Low levels of plasma proteins » Fluids are forced out of the capillary beds at the arterial ends by blood pressure as usual, but fail to return to the blood at the venous end » Interstitial spaces become congested with fluid » Result of poor nutrition, liver disease, or renal failure

ELECTROLYTE BALANCE • Sodium is the most important cation to regulation of fluid and electrolyte balance in the body due to its abundance and osmotic pressure: – Since all body fluids are in chemical equilibrium, any change in sodium levels causes a compensatory shift in water, affecting plasma volume, blood pressure, and intracellular and interstitial fluid volumes

HOMEOSTATIC IMBALANCE • Severe electrolyte deficiencies prompt a craving for salty foods and often exotic foods, such as smoked meats or pickled eggs: – Common in Addison’s disease • Disorder entailing deficient mineralocorticoid hormone (aldosterone: regulates the retention and excretion of fluids and electrolytes) production by the adrenal cortex • Pica: appetite for abnormal substances – When electrolytes other than Na. Cl are deficient, a person may even eat substances not usually considered foods, like chalk, clay, starch, and burnt match tips

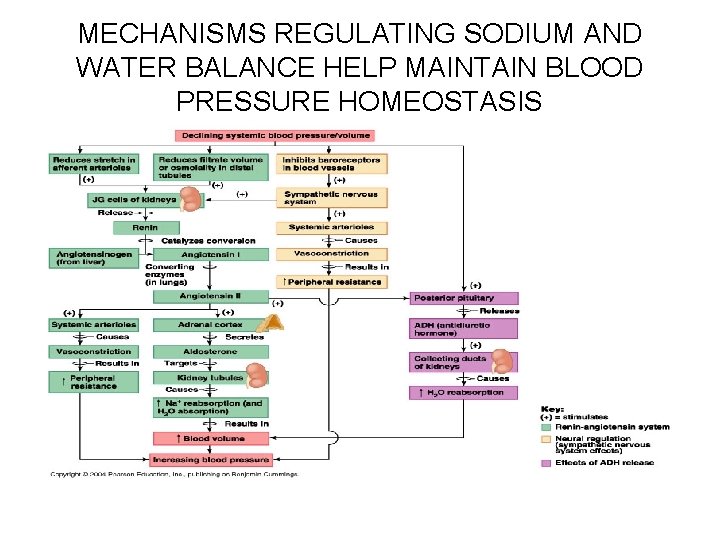
The Central Role of Sodium in Fluid and Electrolyte Balance • Sodium holds a central position in fluid and electrolyte balance and overall body homeostasis – Regulating the balance between sodium input and output is one of the most important renal functions • The salts Na. HCO 3 and Na. Cl account for 90 -95% of all solutes in ECF (extracellular fluid): – Na being the single most abundant – Cellular plasma membranes are relatively impermeable to Na+, but some does manage to diffuse in and must be pumped out against its electrochemical gradient • These two qualities give sodium the primary role in controlling ECF volume and water distribution in the body • It is important to understand that while the sodium content of the body may change, its ECF concentration normally remains stable because of immediate adjustments in water volume – Water follows salt
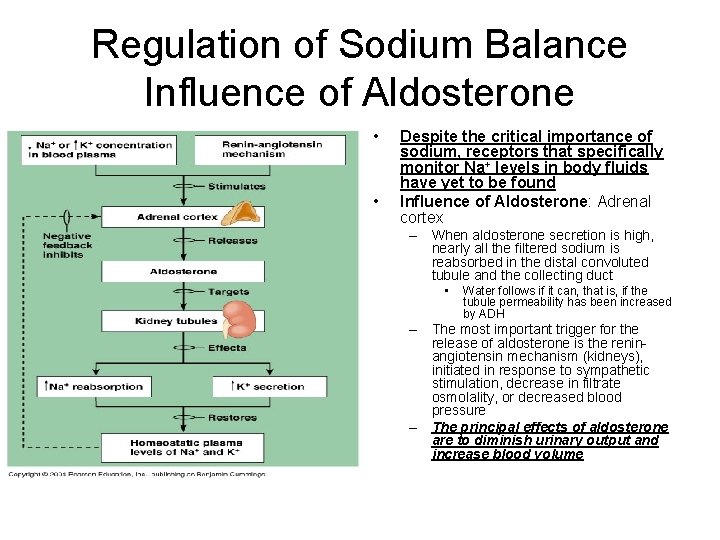
Regulation of Sodium Balance Influence of Aldosterone • • Despite the critical importance of sodium, receptors that specifically monitor Na+ levels in body fluids have yet to be found Influence of Aldosterone: Adrenal cortex – When aldosterone secretion is high, nearly all the filtered sodium is reabsorbed in the distal convoluted tubule and the collecting duct • Water follows if it can, that is, if the tubule permeability has been increased by ADH – The most important trigger for the release of aldosterone is the reninangiotensin mechanism (kidneys), initiated in response to sympathetic stimulation, decrease in filtrate osmolality, or decreased blood pressure – The principal effects of aldosterone are to diminish urinary output and increase blood volume
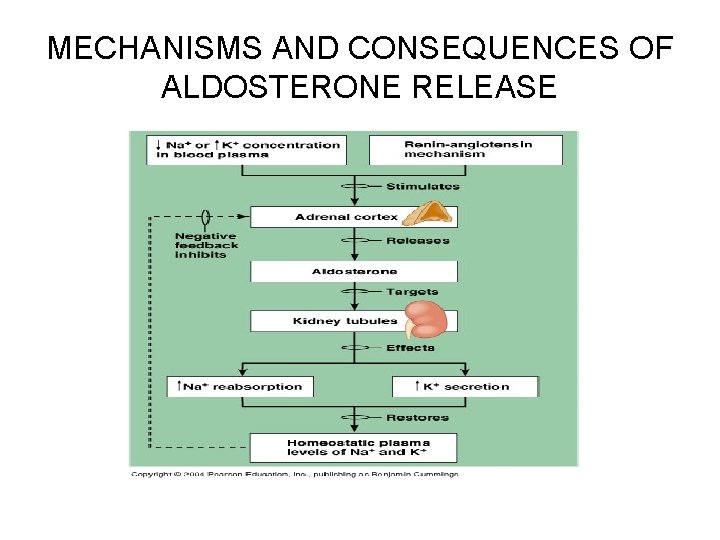
MECHANISMS AND CONSEQUENCES OF ALDOSTERONE RELEASE
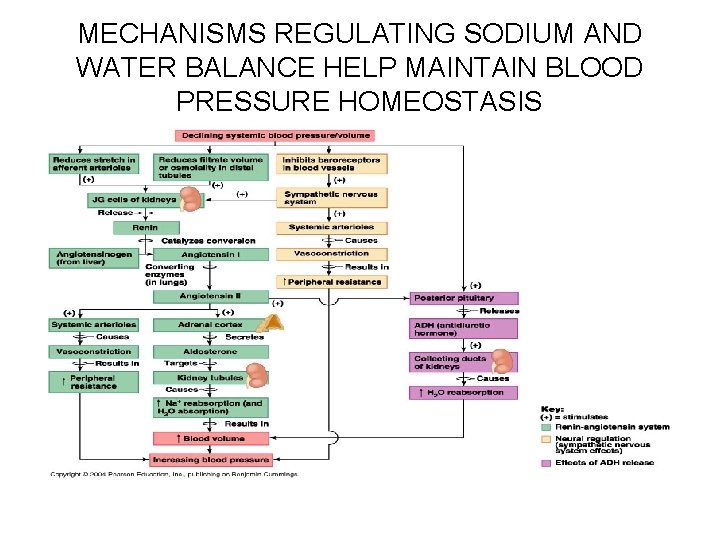
MECHANISMS REGULATING SODIUM AND WATER BALANCE HELP MAINTAIN BLOOD PRESSURE HOMEOSTASIS

HOMEOSTATIC IMBALANCE • People with Addison’s disease (hypoaldosteronism) lose tremendous amounts of Na. Cl and water to urine – As long as they ingest adequate amounts of salt and fluids, people with this condition can avoid problems, but they are perpetually teetering on the brink of hypovolemia (decreased blood volume) and dehydration

Regulation of Sodium Balance Cardiovascular Baroreceptors • Cardiovascular baroreceptors monitor blood volume so that blood pressure remains stable: – Because Na+ concentration determines fluid volume, the baroreceptors might be regarded as “sodium receptors”
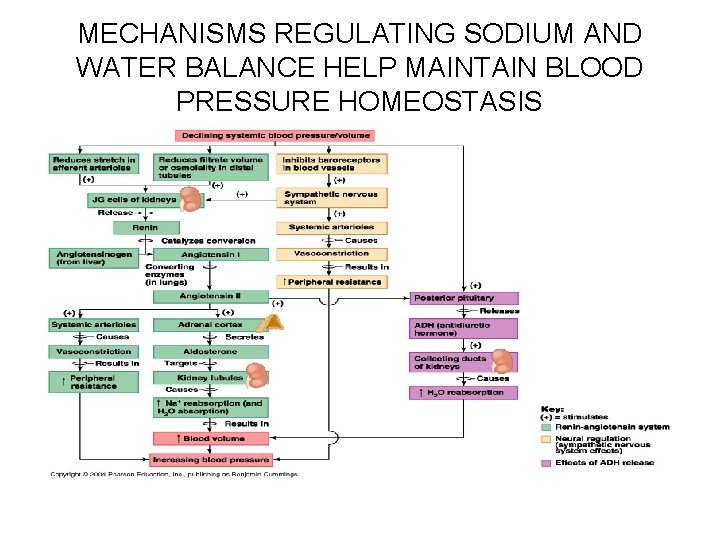
MECHANISMS REGULATING SODIUM AND WATER BALANCE HELP MAINTAIN BLOOD PRESSURE HOMEOSTASIS
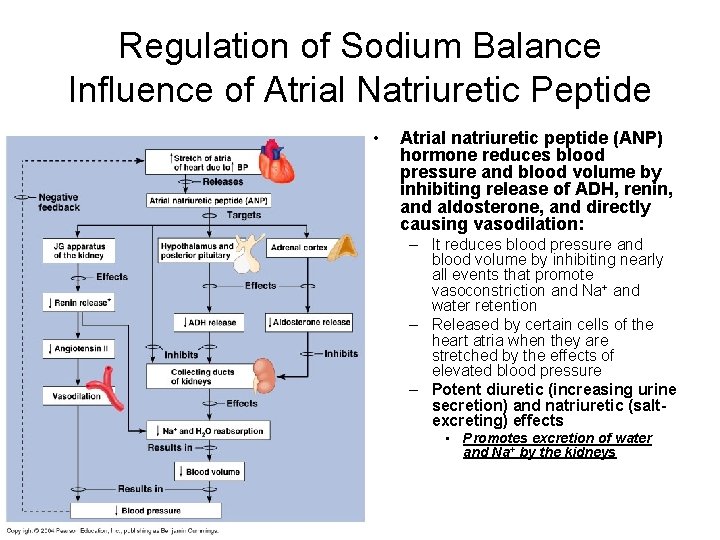
Regulation of Sodium Balance Influence of Atrial Natriuretic Peptide • Atrial natriuretic peptide (ANP) hormone reduces blood pressure and blood volume by inhibiting release of ADH, renin, and aldosterone, and directly causing vasodilation: – It reduces blood pressure and blood volume by inhibiting nearly all events that promote vasoconstriction and Na+ and water retention – Released by certain cells of the heart atria when they are stretched by the effects of elevated blood pressure – Potent diuretic (increasing urine secretion) and natriuretic (saltexcreting) effects • Promotes excretion of water and Na+ by the kidneys
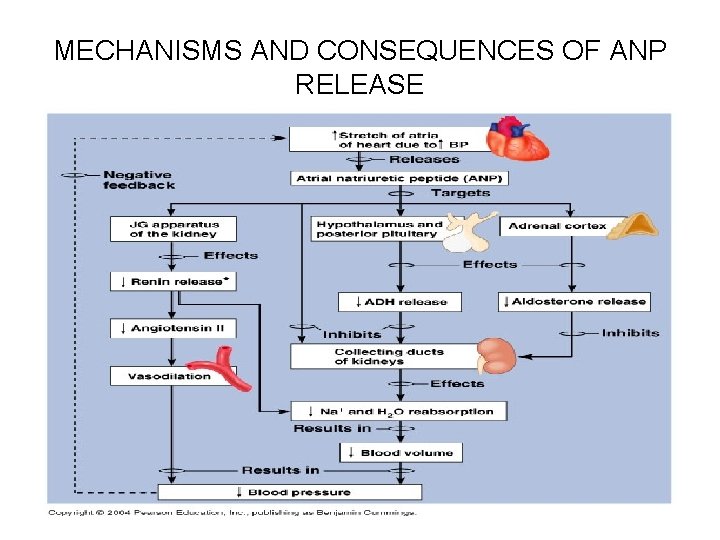
MECHANISMS AND CONSEQUENCES OF ANP RELEASE

Regulation of Sodium Balance Influence of Other Hormones • Estrogens are chemically similar to aldosterone, and enhance reabsorption of salt by the renal tubules: – Because water follows, many women retain fluid as their estrogen levels rise during the menstrual cycle – The edema experienced by many pregnant women is also largely due to the effect of estrogen • Progesterone appears to decrease Na+ reabsorption by blocking the effect aldosterone has on the renal tubules: – Thus, progesterone has a diuretic-like effect and promotes Na+ and water loss • Glucocorticoids (cortisol and hydrocortisol) enhance tubular reabsorption of sodium, but increase glomerular filtration

Regulation of Potassium Balance • Potassium, the chief intracellular cation, is required for normal neuromuscular functioning as well as for several essential activities – Potassium is critical to the maintenance of the membrane potential of neurons and muscle cells, and is a buffer that compensates for shifts of hydrogen ions in or out of the cell – The heart is particularly sensitive to K+ levels • Both too much (hyperkalemia) and too little (hypokalemia) can disrupt electrical conduction in the heart, leading to sudden death

Regulation of Potassium Balance Regulatory Site: The Cortical Collecting Duct • Like Na+ balance, K+ balance is maintained chiefly by renal mechanisms • Potassium balance is chiefly regulated by renal mechanisms, which control the amount of potassium secreted into the filtrate • The main thrust of renal regulation of K+ is to excrete it: – Because the kidneys have a limited ability to retain K+ it may be lost in urine even in the face of a deficiency • Consequently, failure to ingest potassium-rich substances eventually results in a severe deficiency

Regulation of Potassium Balance Influence of Plasma Potassium Concentration • The single most important factor influencing K+ secretion is the K+ concentration in blood plasma: – A high-potassium diet increases the K+ content of the ECF – This favors entry of K+ into the principal cells of the cortical collecting duct and prompts them to secrete K+ into the filtrate so that more of it is excreted • Conversely, a low-potassium diet or accelerated K+ loss depresses its secretion by the collecting ducts
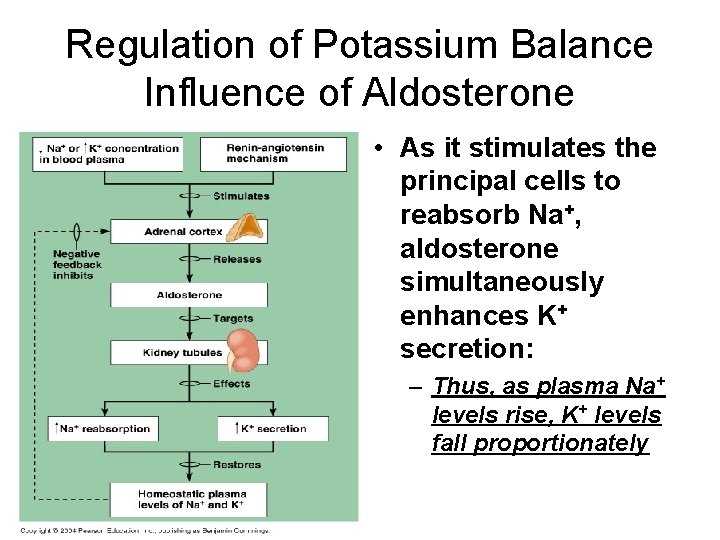
Regulation of Potassium Balance Influence of Aldosterone • As it stimulates the principal cells to reabsorb Na+, aldosterone simultaneously enhances K+ secretion: – Thus, as plasma Na+ levels rise, K+ levels fall proportionately
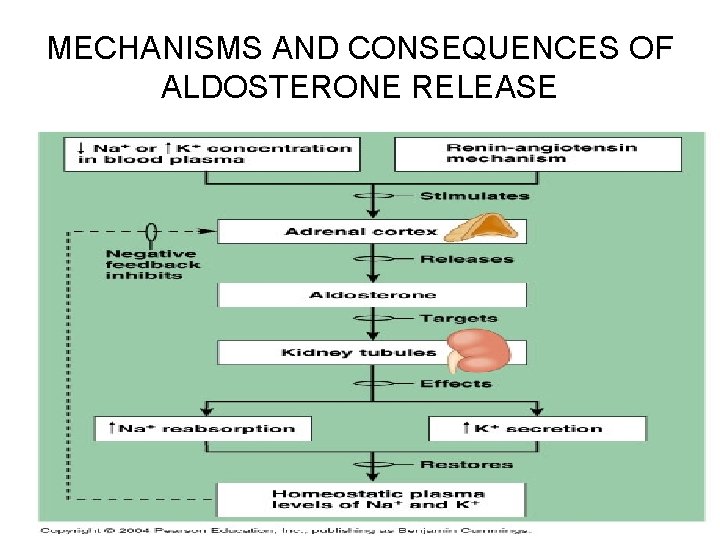
MECHANISMS AND CONSEQUENCES OF ALDOSTERONE RELEASE

HOMEOSTATIC IMBALANCE • In an attempt to reduce Na. Cl intake, many people have turned to salt substitutes, which are high in potassium: – Heavy consumption of these substitutes is safe only when aldosterone release in the body is normal – In the absence of aldosterone, hyperkalemia is swift and lethal regardless of K+ intake – High levels of aldosterone cause potassium levels to fall so low that neurons all over the body hyperpolarize and paralysis occurs

Regulation of Calcium and Phosphate Balance • About 99% of the body’s calcium is found in bones in the form of calcium phosphate salts, which provide strength and rigidity to the skeleton • Ionic calcium in the ECF is important for: – – Blood clotting Cell membrane permeability Secretory behavior Neuromuscular excitability

Regulation of Calcium and Phosphate Balance • Calcium ion levels are closely regulated by parathyroid hormone and calcitonin; about 98% is reabsorbed – Parathyroid hormone (PTH: parathormone) is released when blood calcium levels decline, and targets the bones, small intestine, and kidneys: • Promotes an increase in calcium levels • Targets: – Bone: breaking down bone matrix and liberating Ca 2+ – Small intestine: enhances intestinal absorption of Ca 2+ indirectly by stimulating the kidneys to transform vitamin D to its active form, which is a necessary cofactor for Ca 2+ absorption by the small intestine – Kidneys: increases Ca 2+ reabsorption while decreasing phosphate ion reabsorption » Thus, calcium conservation and phosphate excretion go hand in hand » Hence, the product of Ca 2+ and HPO 42 - remains constant, preventing calcium-salt deposit in bones or soft tissues

Regulation of Calcium and Phosphate Balance • Thyroid hormone (calcitonin) is an antagonist to parathyroid hormone, and is released when blood calcium rises, targeting bone: – Targets bone, where it encourages deposit of calcium salts and inhibits bone reabsorption (process of absorbing calcium again from the bone and transported to the interstitial fluid or blood)

Regulation of Anions • Chloride is the major anion reabsorbed with sodium, and helps maintain the osmotic pressure of the blood

ACID-BASE BALANCE • Because of the abundance of hydrogen bonds in the body’s functional proteins (enzymes, hemoglobin, cytochromes, and others) they are strongly influenced by hydrogen ion concentration: – It follows then that nearly all biochemical reactions are influenced by the p. H of their fluid environment, and the acid -base balance of body fluids is closely regulated – Optimal p. H varies from one body fluid to another: • When arterial blood p. H rises above 7. 45, the body is in alkalosis (alkalemia); when arterial p. H falls below 7. 35, the body is in acidosis (acidemia) – Between 7. 0 and 7. 35 is called physiological acidosis even though the value is slightly basic – Most hydrogen ions originate as metabolic by products, although they can also enter the body via ingested foods

ACID-BASE BALANCE • Dissociation of strong and weak acids: • (a): when added to water, the strong acid HCl dissociates completely into its ions (H+ and Cl-) • (b): dissociation of H 2 CO 3, a weak acid, is very incomplete, and some molecules of H 2 CO 3 remain undissociated in solution
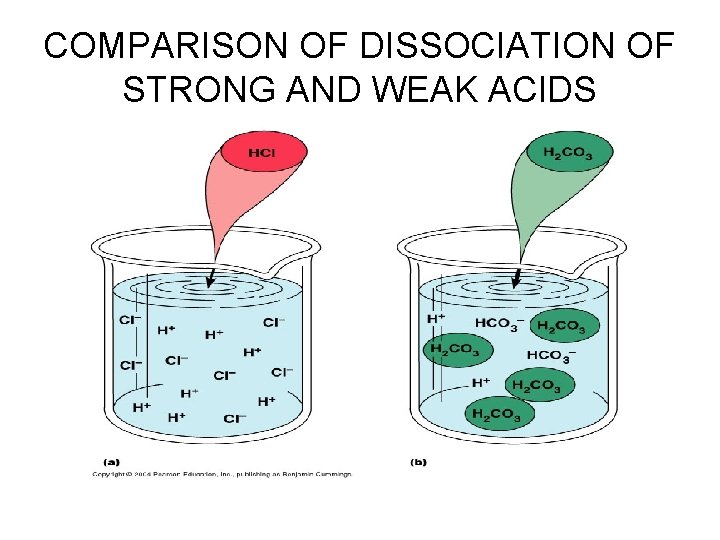
COMPARISON OF DISSOCIATION OF STRONG AND WEAK ACIDS

Chemical Buffer System Bicarbonate Buffer Systems • • A chemical buffer is a system of one or two molecules that acts to resist changes in p. H by binding H+ when the p. H drops, or releasing H+ when the p. H rises The bicarbonate buffer system is the main buffer of the extracellular fluid, and consists of carbonic acid and its salt, sodium bicarbonate: – When a strong acid is added to the solution, carbonic acid is mostly unchanged, but bicarbonate ions of the salt bind excess H +, forming more carbonic acid: • HCl + Na. HCO 3 → H 2 CO 3 + Na. Cl • Strong acid + weak base → weak acid + salt • p. H lowered slightly – When a strong base is added to solution, the sodium bicarbonate remains relatively unaffected, but carbonic acid dissociates further, donating more H+ to bind the excess hydroxide • Na. OH + H 2 CO 3 → Na. HCO 3 + H 2 O • Strong base + weak acid → weak base + water • p. H rises very little – Bicarbonates buffer system: sodium, potassium, and magnesium
Chemical Buffer System Phosphate Buffer System • The phosphate buffer system operates in the urine and intracellular fluid similar to the bicarbonate buffer system • The components of the phosphate system are the: – Sodium salts of dihydrogen phosphate (H 2 PO 4 -) – Sodium salts of monohydrogen phosphate (HPO 42 -) – Na. H 2 PO 4 acts as a weak acid – HCl + Na 2 HPO 4 → Na. H 2 PO 4 + Na. Cl • Strond acid + weak base → weak acid + salt – H+ released by strong acids is tied up in weak acids • Na. OH + Na. H 2 PO 4 • Strong base weak acid → Na 2 HPO 4 + H 2 O → weak base + water – Strong bases are converted to weak bases

Chemical Buffer System The Protein Buffer System • Proteins in plasma and in cells are the body’s most plentiful and powerful source of buffers, and constitute the protein buffer system: – At least ¾ of all the buffering power of body fluids resides in cells, and most of this reflects the buffering activity of intracellular proteins • Proteins are polymers of amino acids: – Consists of organic acids containing carboxyl groups that dissociate to: • Release H+ when the p. H begins to rise – R—COOH → R—COO- + H+ • Bind excess H+ when the p. H declines – R—COO- + H+ → R—COOH – Consists of an amide group that can act as a base and accept H+: • The exposed –NH 2 group can bind with hydrogen ions, becoming –NH 3 - – R—NH 2 + H+ → R—NH 3+ » Because this removes free hydrogen ions from the solution, it prevents the solution from becoming too acidic » Consequently, a single protein molecule can function reversibly as either an acid or a base depending on the p. H of its environment » Molecules with this ability are called amphoteric molecules » Example: hemoglobin

Respiratory Regulation of H+ • Carbon dioxide from cellular metabolism enters erythrocytes and is converted to bicarbonate ions for transport in the plasma: – carbonic – anhydrase – CO 2 + H 2 O ↔ H 2 CO 3 ↔ H+ + HCO 3– carbonic acid bicarbonate ion – – When hypercapnia (increased amount of carbon dioxide in the blood) occurs, blood p. H drops, activating medullary respiratory centers, resulting in increased rate and depth of breathing and increased unloading of CO 2 in the lungs • The reaction is pushed to the right – A rising plasma H+ concentration resulting from any metabolic process excites the respiratory center indirectly (peripheral chemoreceptors) to stimulate deeper, more rapid respiration • As ventilation increases, more CO 2 is removed from the blood, pushing the reaction to the left and reducing the H+ concentration

Renal Mechanisms of Acid-Base Balance • Chemical buffers can tie up excess acids or bases temporarily, but they cannot eliminate them from the body: – The lungs can dispose of the volatile acid carbonic acid by eliminating CO 2 – Only the kidneys can rid the body of acids (phosphoric, uric, and lactic acids, and ketone bodies) generated by cellular metabolism, while also regulating blood levels of alkaline substances and renewing chemical buffer components – Thus, the ultimate acid-base regulatory organs are the kidneys, which act slowly but surely to compensate for acidbase imbalances resulting from variations in diet or metabolism or from disease: • The most important renal mechanisms for regulating acid-base balance of the blood involve: – 1. Conserving (reabsorbing) or generating new HCO 3– 2. Excreting HCO 3 -

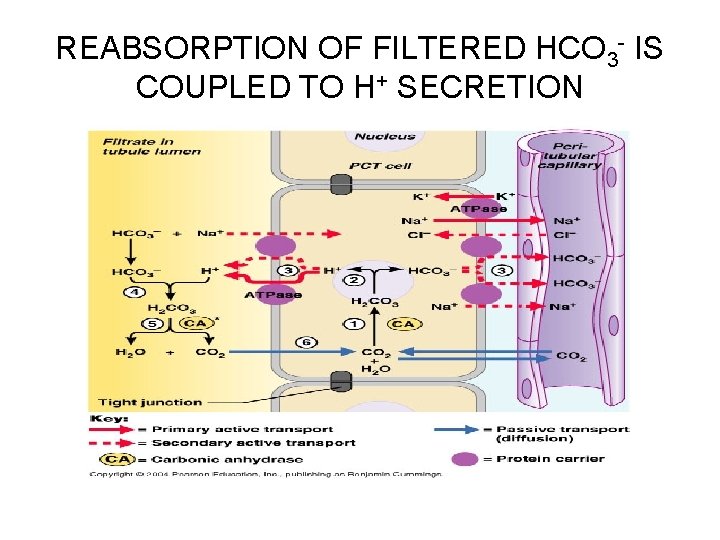
REABSORPTION OF FILTERED HCO 3 - IS COUPLED TO H+ SECRETION • 1. CO 2 combines with water within the tubule cell, forming H 2 CO 3 • 2. H 2 CO 3 is quickly split, forming H+ + HCO 3 • 3. For each H+ secreted into the filtrate, a bicarbonate ion (HCO 3 -) enters the peritubular capillary blood by cotransport either with Na+ or Cl-
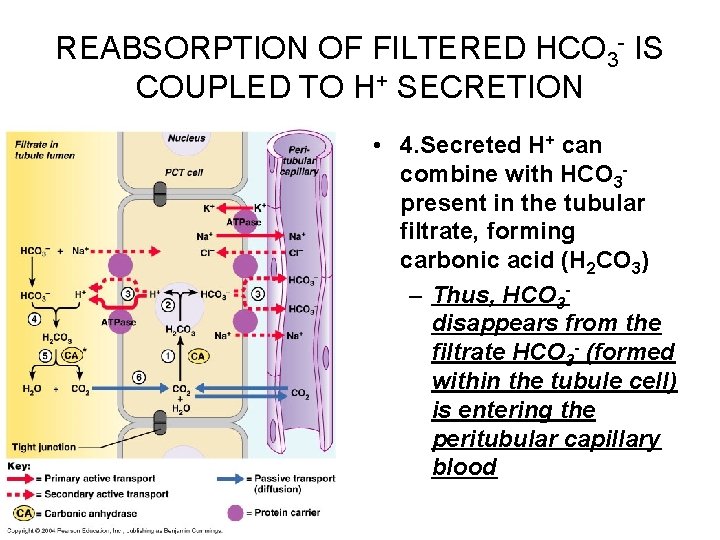
REABSORPTION OF FILTERED HCO 3 - IS COUPLED TO H+ SECRETION • 4. Secreted H+ can combine with HCO 3 present in the tubular filtrate, forming carbonic acid (H 2 CO 3) – Thus, HCO 3 disappears from the filtrate HCO 3 - (formed within the tubule cell) is entering the peritubular capillary blood

REABSORPTION OF FILTERED HCO 3 - IS COUPLED TO H+ SECRETION • 5. The H 2 CO 3 formed in the filtrate dissociates to release carbon dioxide and water • 6. Carbon dioxide then diffuses into the tubule cell, where it acts to trigger further H+ secretion
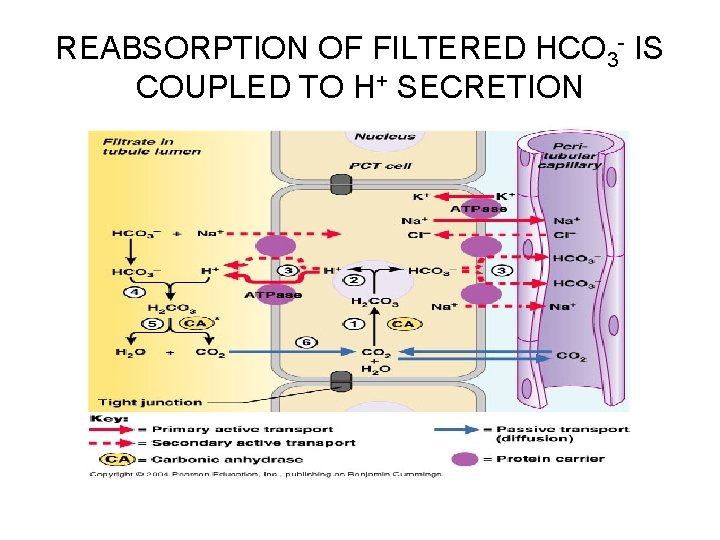
REABSORPTION OF FILTERED HCO 3 - IS COUPLED TO H+ SECRETION

Conserving Filtered Bicarbonate Ions: Bicarbonate Reabsorption • Bicarbonate ions (important part of buffer system) can be conserved from filtrate when depleted, and their reabsorption is dependent on H+ secretion

Generating New Bicarbonate Ions • Two renal mechanisms commonly carried out by the type A intercalated cells of the collecting ducts generate new HCO 3 - that can be added to plasma • Both mechanisms involve renal excretion of acid, via secretion and excretion of either H+ or ammonium ions in urine
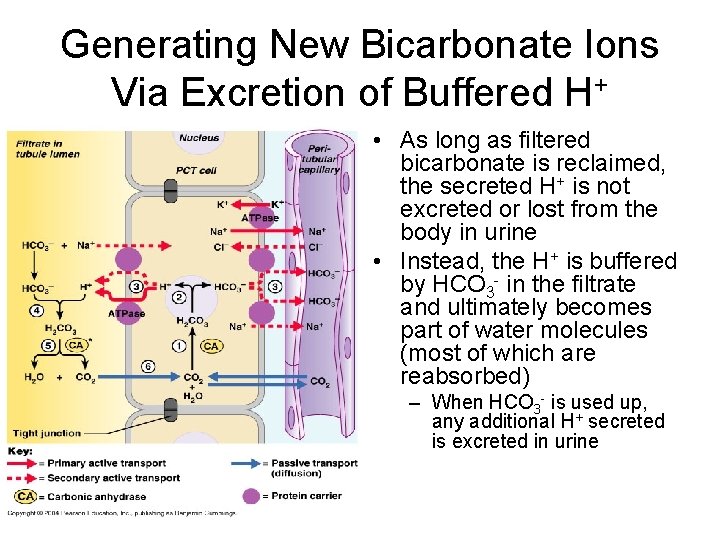
Generating New Bicarbonate Ions Via Excretion of Buffered H+ • As long as filtered bicarbonate is reclaimed, the secreted H+ is not excreted or lost from the body in urine • Instead, the H+ is buffered by HCO 3 - in the filtrate and ultimately becomes part of water molecules (most of which are reabsorbed) – When HCO 3 - is used up, any additional H+ secreted is excreted in urine
REABSORPTION OF FILTERED HCO 3 - IS COUPLED TO H+ SECRETION
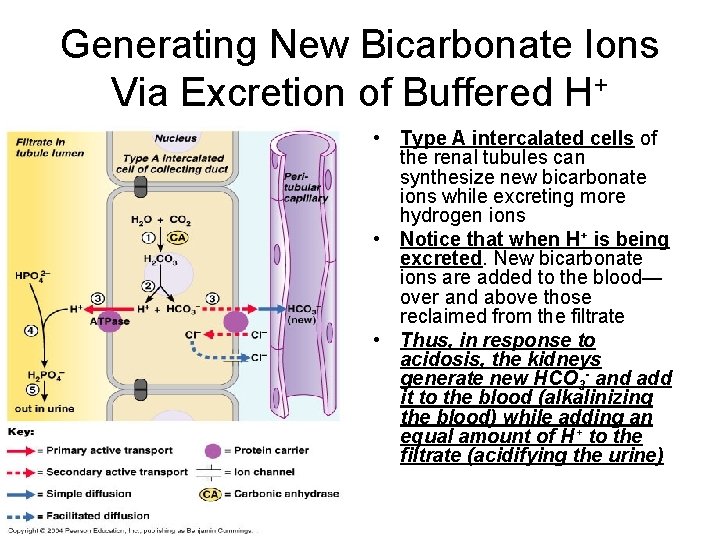
Generating New Bicarbonate Ions Via Excretion of Buffered H+ • Type A intercalated cells of the renal tubules can synthesize new bicarbonate ions while excreting more hydrogen ions • Notice that when H+ is being excreted. New bicarbonate ions are added to the blood— over and above those reclaimed from the filtrate • Thus, in response to acidosis, the kidneys generate new HCO 3 - and add it to the blood (alkalinizing the blood) while adding an equal amount of H+ to the filtrate (acidifying the urine)

Generating New Bicarbonate Ions Via Excretion of Buffered H+ • H+ from the dissociation of H 2 CO 3 is actively secreted by a H+-ATPase pump and combines with HPO 42 - in the lumen • HCO 3 - generated at the same time leaves the basolateral membrane via an antiport carrier in a HCO 3—Clexchange process which maintains electroneutrality in the duct cells
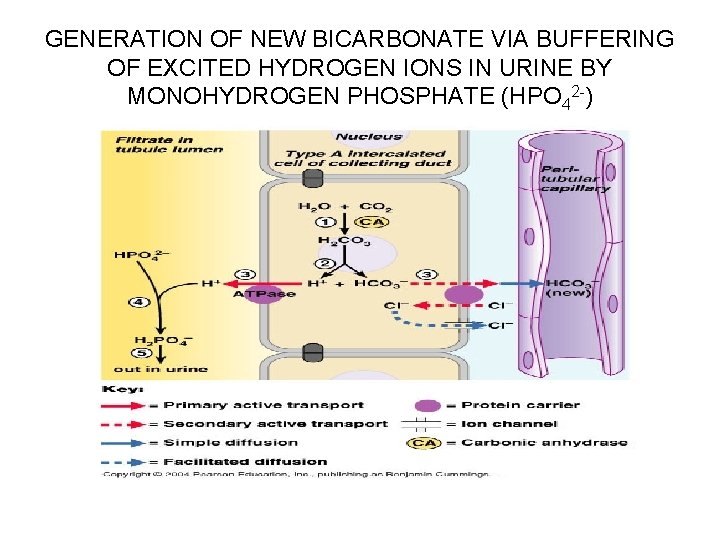
GENERATION OF NEW BICARBONATE VIA BUFFERING OF EXCITED HYDROGEN IONS IN URINE BY MONOHYDROGEN PHOSPHATE (HPO 42 -)
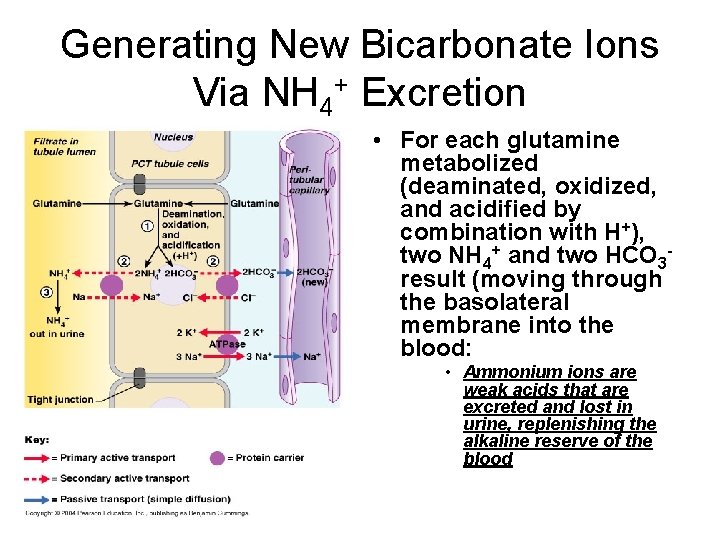
Generating New Bicarbonate Ions Via NH 4+ Excretion • For each glutamine metabolized (deaminated, oxidized, and acidified by combination with H+), two NH 4+ and two HCO 3 result (moving through the basolateral membrane into the blood: • Ammonium ions are weak acids that are excreted and lost in urine, replenishing the alkaline reserve of the blood
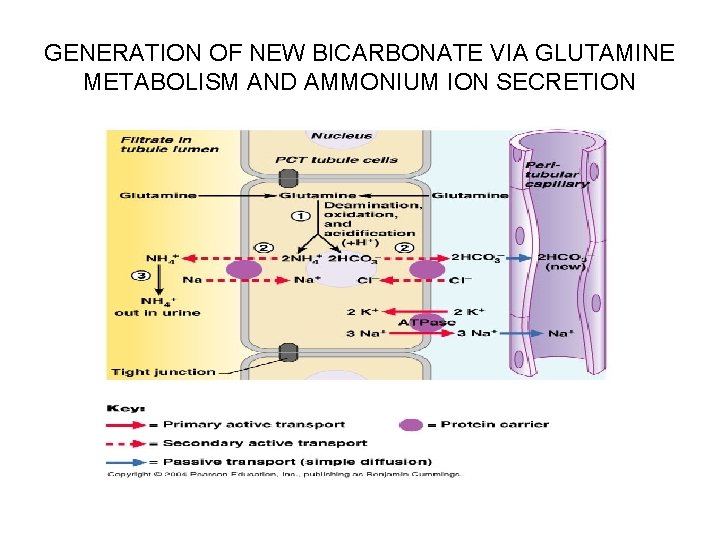
GENERATION OF NEW BICARBONATE VIA GLUTAMINE METABOLISM AND AMMONIUM ION SECRETION

Bicarbonate Ion Secretion • When the body is in alkalosis (increase in blood alkalinity), type B intercalated cells excrete bicarbonate, and reclaim hydrogen ions to acidify the blood

Abnormalities of Acid-Base Balance • Respiratory acidosis is characterized by falling blood p. H and rising PCO 2, which can result from: – Shallow breathing – Some respiratory diseases (pneumonia, cystic fibrosis, or emphysema) • • Respiratory alkalosis results when carbon dioxide is eliminated from the body faster than it is produced, such as during hyperventilation Metabolic acidosis is characterized by low blood p. H and bicarbonate levels, and is due to excessive loss of: – Bicarbonate ions (as might result from persistent diarrhea) – Ingestion of too much alcohol (which is metabolized to acetic acid) • Metabolic alkalosis is indicated by rising blood p. H and bicarbonate levels, and is the result of: – Vomiting (loss of acid contents of the blood) – Excessive base intake (antacids) – Constipation: more than the usual amount of HCO 3 - is reabsorbed by the colon
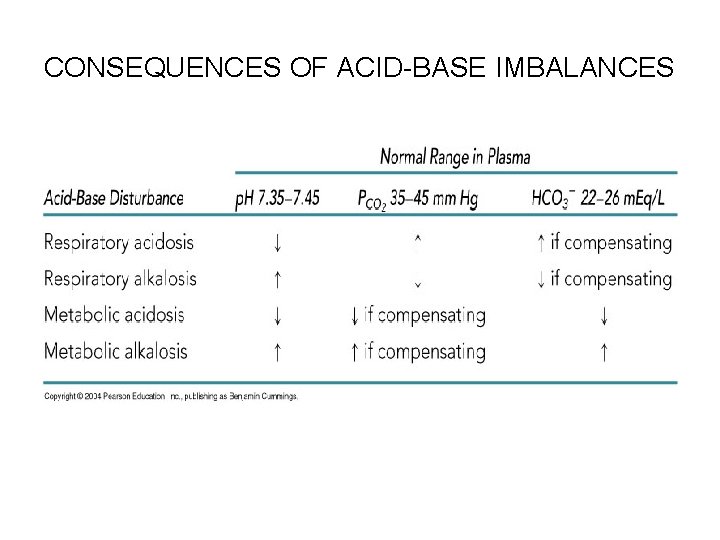
CONSEQUENCES OF ACID-BASE IMBALANCES

Effects of Acidosis and Alkalosis • Respiratory rate and depth: – Increases during metabolic acidosis: • When blood p. H falls below 7. 0, the central nervous system is so depressed that the person goes into coma and death soon follows – Decreases during metabolic alkalosis: • When blood p. H rises above 7. 8, the nervous system is overexcited, leading to such characteristic signs as muscle tetany, extreme nervousness, and convulsions • Death often results from respiratory arrest
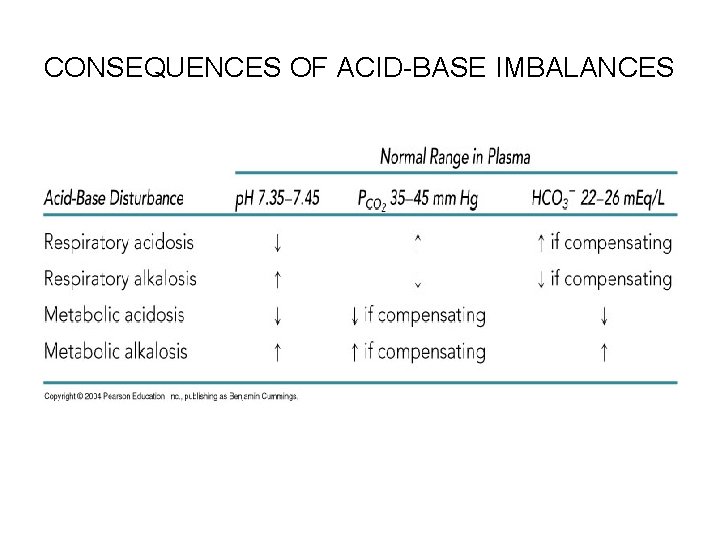
CONSEQUENCES OF ACID-BASE IMBALANCES

Respiratory and Renal Compensations • When an acid-base imbalance is due to inadequate functioning of one of the physiological buffer systems (lungs or kidneys), the other system tries to compensate: – The respiratory system attempts to compensate for metabolic acid-base imbalances, and the kidneys (although much slower) work to correct imbalances caused by respiratory disease

Respiratory Compensations • Compensates for metabolic acid-base imbalances • Metabolic acidosis, the respiratory rate and depth are usually elevated—an indication that the respiratory centers are stimulated by the high H+ levels – The blood p. H is low (below 7. 35) and the HCO 3 - level is below 22 m. Eq/L – As respiratory system exhales CO 2 to rid the blood of excess acid, the PCO 2 falls below 35 mm • Metabolic alkalosis involves slow, shallow breathing, which allows CO 2 to accumulate in the blood – A metabolic alkalosis being compensated by respiratory mechanisms is revealed by a p. H over 7. 45 at least initially, elevated bicarbonate levels (over 26 m. Eq/L), and a PCO 2 above 45 mm Hg

Renal Compensations • When an acid-base imbalance is of respiratory origin, renal mechanisms are stepped up to correct the imbalance • Hypoventilating individual will exhibit acidosis • Acidosis: when renal compensation is occurring, both the PCO 2 and the HCO 3 - levels are high – The high PCO 2 is the cause of the acidosis, and the rising HCO 3 - level indicates that the kidneys are retaining bicarbonate to offset the acidosis • Alkalosis: a person with renal-compensated respiratory alkalosis will have a high blood p. H and a low PCO 2 – Bicarbonate ion levels begin to fall as the kidneys eliminate more HCO 3 from the body by failing to reclaim it or by actively secreting it • Note that the kidneys cannot compensate for alkalosis or acidosis if that condition reflects a renal problem
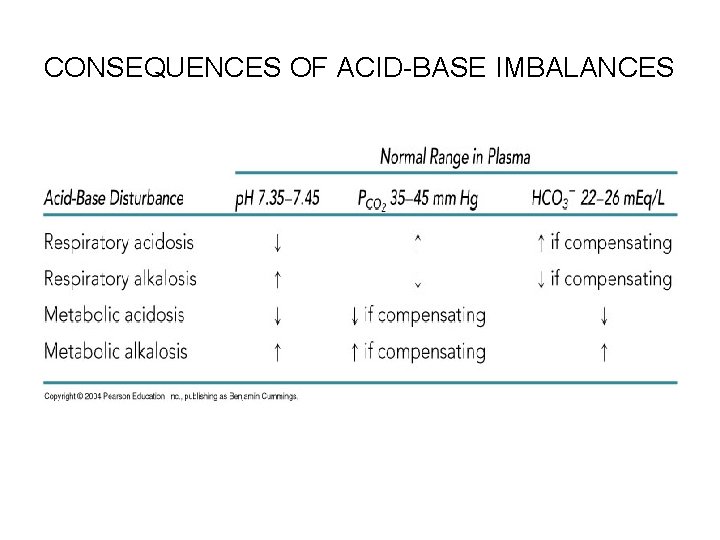
CONSEQUENCES OF ACID-BASE IMBALANCES

DEVELOPMENTAL ASPECTS OF FLUID, ELECTROLYTE, AND ACID-BASE BALANCE • An embryo and young fetus are more than 90% water, but as solids accumulate, the percentage declines to about 70 -80% at birth • Distribution of body water begins to change at 2 months of age, and takes on adult distribution by the time a child is 2 years of age • At puberty, sex differences in body water content appear as males develop more skeletal muscle • During infancy, problems with fluid, electrolyte, and acid-base balance are common, due to large-scale changes in. PCO 2 • In old age, body water loss is primarily from the intracellular compartment, due to decline in muscle mass, and increase in adipose tissue • Increased insensitivity to thirst cues makes the elderly vulnerable to dehydration, and electrolyte or acid-base imbalances
- Slides: 80