Unit 11 Solubility Water Solutions I Water A

Unit 11 - Solubility Water & Solutions
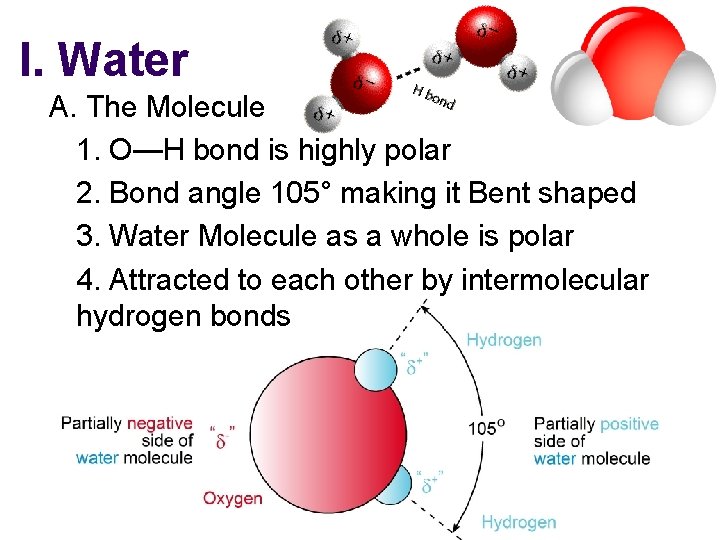
I. Water A. The Molecule 1. O—H bond is highly polar 2. Bond angle 105° making it Bent shaped 3. Water Molecule as a whole is polar 4. Attracted to each other by intermolecular hydrogen bonds

I. Water (cont. ) B. Important Properties 1. High surface tension 2. low vapor pressure l hydrogen bonds hold molecules to one another, tendency to escape surface is low 3. high specific heat capacity l 4. 18 J/g×°C 4. high melting and boiling points l 0°C and 100°C
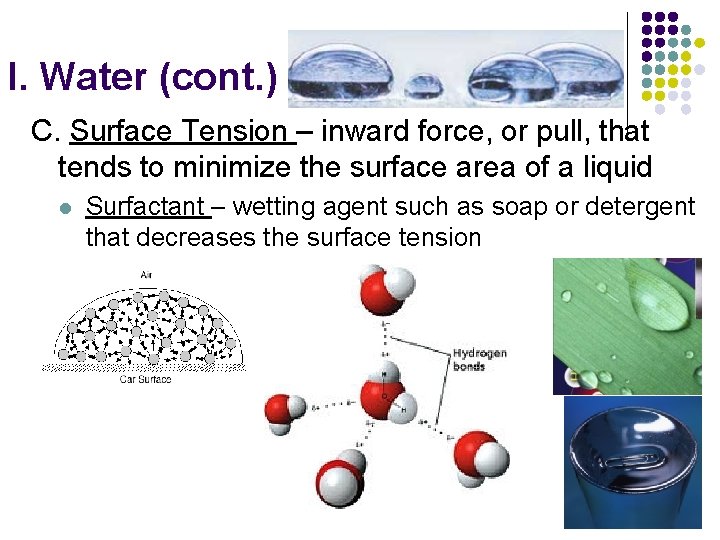
I. Water (cont. ) C. Surface Tension – inward force, or pull, that tends to minimize the surface area of a liquid l Surfactant – wetting agent such as soap or detergent that decreases the surface tension

I. Water (cont. ) D. Atypical Ice 1. As a typical liquid cools, density increases b/c Volume decreases as the mass stays constant 2. As water cools it first behaves like a typical liquid until it reaches 4°C 3. Below 4°C the density of water starts to decrease **Ice is one of only a few solids that float in their own liquid.
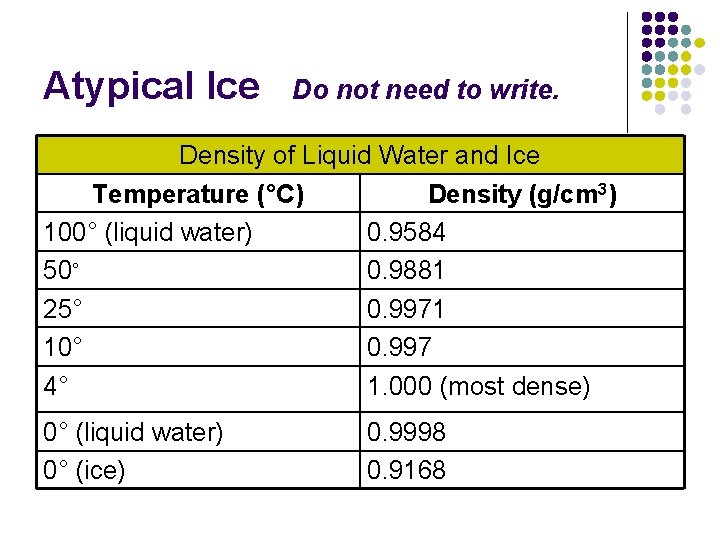
Atypical Ice Do not need to write. Density of Liquid Water and Ice Temperature (°C) Density (g/cm 3) 100° (liquid water) 0. 9584 50° 0. 9881 25° 0. 9971 10° 0. 997 4° 1. 000 (most dense) 0° (liquid water) 0° (ice) 0. 9998 0. 9168
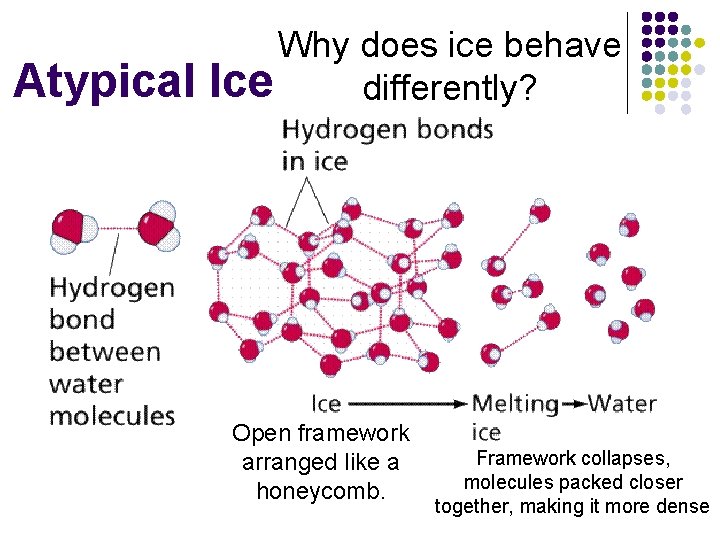
Atypical Why does ice behave Ice differently? Open framework arranged like a honeycomb. Framework collapses, molecules packed closer together, making it more dense
II. The Solution Process A. Solution - homogeneous mixture Solute - substance being dissolved Solvent - dissolving medium

II. The Solution Process B. Solvation– the process of dissolving 1 st solute particles are surrounded by solvent particles 2 nd solute particles are separated and pulled into solution

II. The Solution Process C. “Like Dissolves Like” Nonpolar solvents dissolve nonpolar compounds Polar solvents dissolve polar molecules and ionic compounds

III. Electrolytes A. Electrolytes – compounds that conduct an electric current in solutions l l All ionic compounds are electrolytes Compounds that don’t conduct an electric current are called nonelectrolytes – not composed of ions, includes many molecular compounds (covalent bonds)
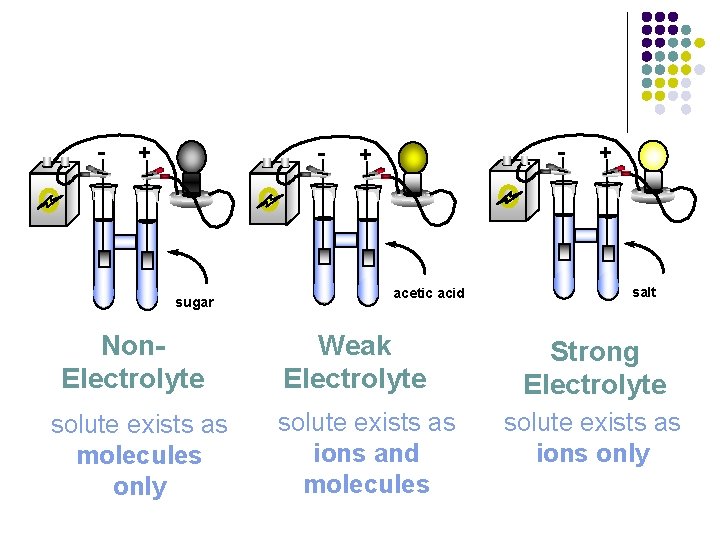
- + - sugar - + acetic acid Non. Electrolyte Weak Electrolyte soluteexistsas as molecules only soluteexistsas as ionsand molecules + salt Strong Electrolyte soluteexistsas as ionsonly
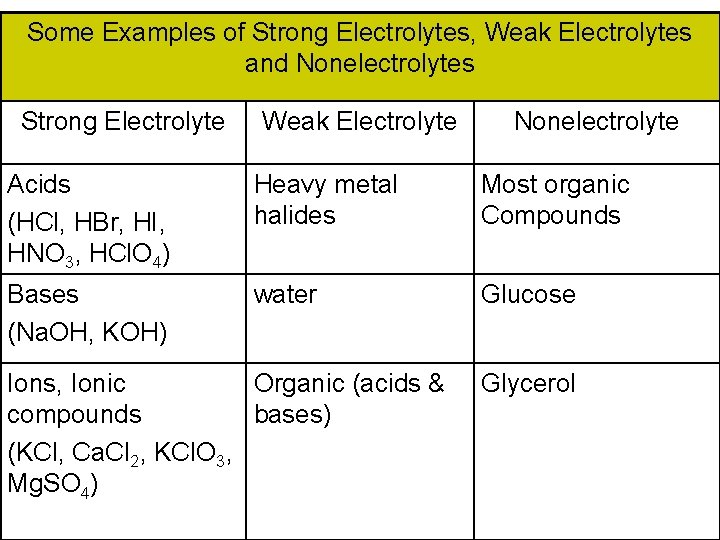
Some Examples of Strong Electrolytes, Weak Electrolytes and Nonelectrolytes Strong Electrolyte Weak Electrolyte Nonelectrolyte Acids (HCl, HBr, HI, HNO 3, HCl. O 4) Heavy metal halides Most organic Compounds Bases (Na. OH, KOH) water Glucose Ions, Ionic Organic (acids & compounds bases) (KCl, Ca. Cl 2, KCl. O 3, Mg. SO 4) Glycerol

IV. Heterogeneous Mixtures A. Suspensions – mixtures from which particles settle out upon standing and the average particle size is greater than 100 nm in diameter. l l Clearly identified as two substances Gravity or filtration will separate the particles B. Colloids – heterogeneous mixtures containing particles that are between 1 nm and 100 nm in diameter l l Appear to be homogeneous but particles are dispersed through medium Ex: paint, aerosol spray, smoke, marshmallow, whipped cream

IV. Heterogeneous Mixtures C. Tyndall Effect - phenomenon observed when beam of light passes through a colloid or suspension l Colloids exhibit the Tyndall effect Colloid Solution

V. Solubility • defined as the maximum grams of solute that will dissolve in 100 g of solvent at a given temperature • based on a saturated solution
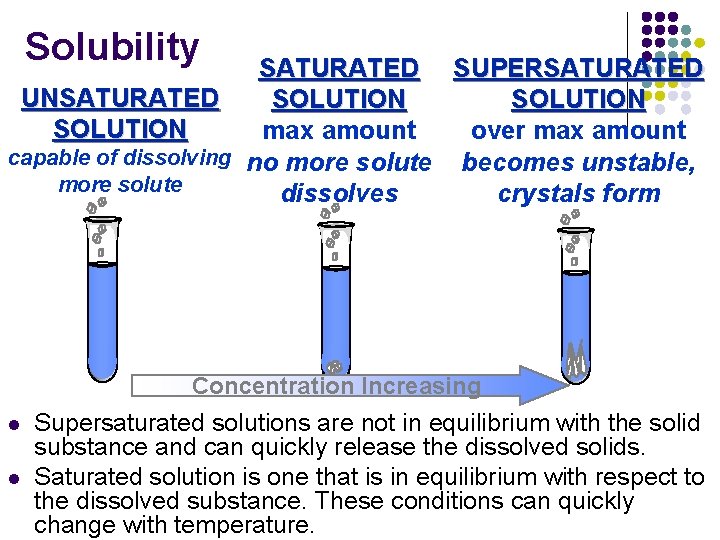
Solubility SATURATED SUPERSATURATED UNSATURATED SOLUTION over max amount capable of dissolving no more solute becomes unstable, more solute crystals form dissolves Concentration Increasing l l Supersaturated solutions are not in equilibrium with the solid substance and can quickly release the dissolved solids. Saturated solution is one that is in equilibrium with respect to the dissolved substance. These conditions can quickly change with temperature.

V. Solubility (cont. ) A. Factors Affecting Solubility 1. Stirring (agitation) l Increases solubility b/c if fresh solvent is brought in contact with the surface of the solute

V. Solubility (cont. ) 2. Temperature l Increases solubility by increasing kinetic energy, which increases the collisions between molecules of solvent and the surface of the solute

V. Solubility (cont. ) 3. Surface Area l A smaller particle size dissolves more rapidly than larger particles size l Surface phenomenon l More surface area exposed, faster rate of dissolving
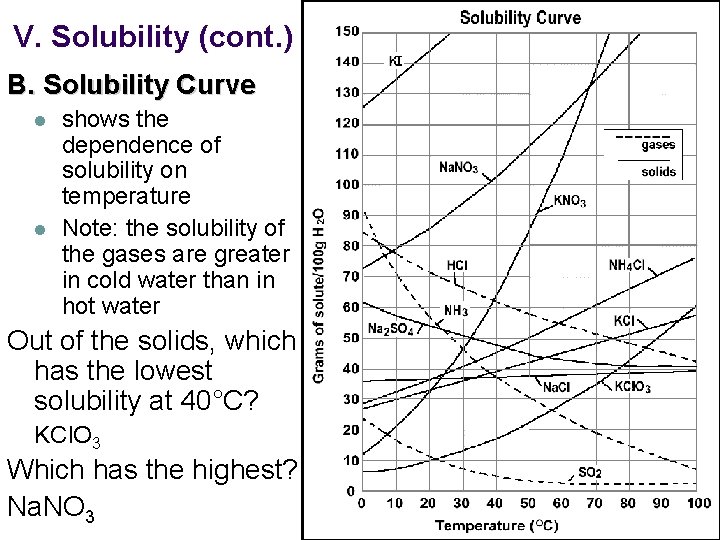
V. Solubility (cont. ) B. Solubility Curve l l shows the dependence of solubility on temperature Note: the solubility of the gases are greater in cold water than in hot water Out of the solids, which has the lowest solubility at 40°C? KCl. O 3 Which has the highest? Na. NO 3
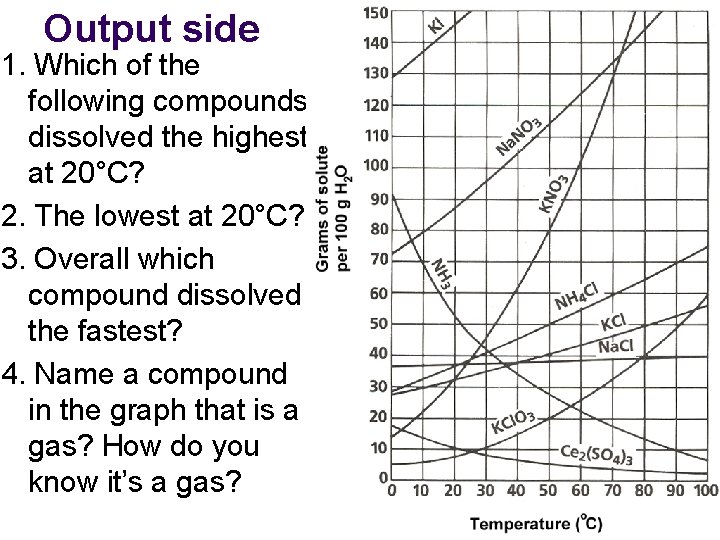
Output side 1. Which of the following compounds dissolved the highest at 20°C? 2. The lowest at 20°C? 3. Overall which compound dissolved the fastest? 4. Name a compound in the graph that is a gas? How do you know it’s a gas?

V. Solubility (cont. ) l Solids are more soluble at. . . l high temperatures. l Gases are more soluble at. . . l low temperatures & high pressures (Henry’s Law). EX: the “bends” & soda When opened partial pressure of CO 2 liquid decreases and the concentration of dissolved CO 2 decreases.

V. Solubility (cont. ) C. Henry’s Law – states that at a given temperature the solubility (S) of a gas in a liquid is directly proportional to the pressure (P) S 1 = S 2 P 1 P 2 If the solubility of a gas in water is 0. 77 g/L at 3. 5 atm of pressure, what is its solubility at 1. 0 atm of pressure? (The temperature is held at 25°C) S 1 = 0. 77 g/L = X 0. 22 g/L P 1 = 3. 5 atm P 2 = 1. 0 atm

VI. Concentrations of Solutions l l Concentration of a solution is a measure of the amount of solute that is dissolved in a given quantity of solution. Dilute solution – contains a low concentration of solute. Concentrated solution – contains a high concentration of solute. Molarity (M) – number of moles of a solute dissolved per liter of solution l a. k. a. molar concentration

A. Molarity l Calculate the number of moles in 1 L of the solution Molarity (M) = moles of solute liters of solution Example 1 Calculate the molarity when 2 mol of glucose is dissolved in 5 L of solution, divide the number of moles by the volume in liters. 2 mol glucose 5 L solution = 0. 4 mol/L = 0. 4 M
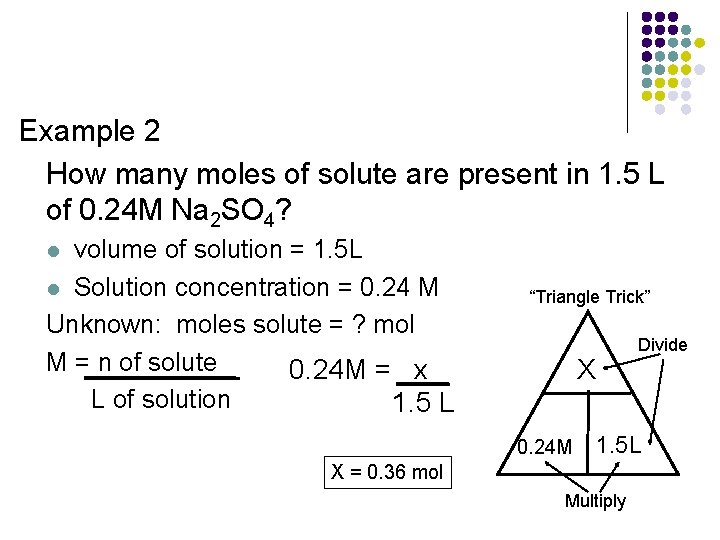
Example 2 How many moles of solute are present in 1. 5 L of 0. 24 M Na 2 SO 4? volume of solution = 1. 5 L l Solution concentration = 0. 24 M Unknown: moles solute = ? mol M = n of solute 0. 24 M = x L of solution 1. 5 L l “Triangle Trick” X 0. 24 M Divide 1. 5 L X = 0. 36 mol Multiply

Example 3 A saline solution contains 0. 90 g Na. Cl in exactly 100 m. L of solution. What is the molarity of the solution? 1. CONVERT GRAMS TO MOLES!! 2. CONVERT m. L to L. M = n of solute UNKNOWN: Molarity L of solution 0. 90 g Na. Cl 1 mol = molar 58 g mass Na 23 + Cl 35 0. 016 mol 100 m. L M = 0. 016 mol 0. 1 L 1 L = 0. 1 L 1000 m. L Molarity = 0. 16 M
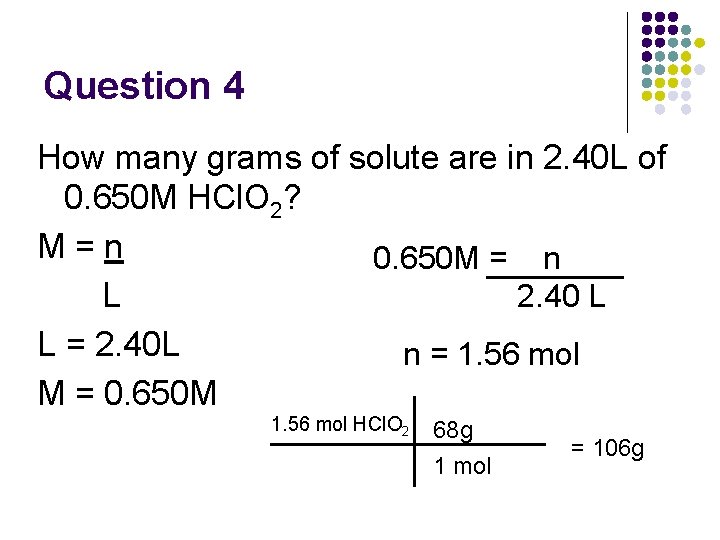
Question 4 How many grams of solute are in 2. 40 L of 0. 650 M HCl. O 2? M=n 0. 650 M = n 2. 40 L L L = 2. 40 L n = 1. 56 mol M = 0. 650 M 1. 56 mol HCl. O 2 68 g 1 mol = 106 g

Molarity Problems You do not have to write the problem. You MUST show your work. 1. A solution has a volume of 2. 0 L and contains 36. 0 g of glucose. If the molar mass of glucose is 180 g, what is the molarity of the solution? 2. A solution has a volume of 250 m. L and contains 0. 70 mol Na. Cl. What is its molarity? 3. How many moles of ammonium nitrate are in 335 m. L of 0. 425 M NH 4 NO 3? 4. How many moles of solute are in 250 m. L of 2. 0 M Ca. Cl 2? How many grams of Ca. Cl 2 is this?

VI. Concentrations of Solutions (cont. ) B. Making Dilutions l l You can make a solution less concentrated by diluting it with solvent. A dilution reduces the moles of solute per unit volume, however, the total moles of solute in solution does not change Moles of solute before dilution = moles of solute after dilution M 1 × V 1 = M 2 × V 2 l M 1 & V 1 are initial and M 2 & V 2 are final of the SAME substance l Volumes can be in L or m. L, as long as the same units are used for both V 1 & V 2

Example 1 How many milliliters of a stock solution of 2. 00 M Mg. SO 4 would you need to prepare 100. 0 m. L of 0. 400 M Mg. SO 4? M 1 = 2. 00 M Mg. SO 4 M 2 = 0. 400 M Mg. SO 4 V 2 = 100 m. L of 0. 400 M Mg. SO 4 Unknown = V 1 M 1 × V 1 = M 2 × V 2 2. 00 M × V 1 = 0. 400 M × 100 m. L V 1 = 20 m. L

Example 2 You need 150 m. L of 0. 40 M Na. Cl and you have a 1. 0 M of Na. Cl solution. Calculate the volume of the Na. Cl solution. V 1 = 150 m. L of 0. 40 M Na. Cl M 1 = 0. 400 M Na. Cl M 2 = 1. 0 M Na. Cl Unknown = V 2 M 1 × V 1 = M 2 × V 2 0. 400 M × 150 m. L = 1. 0 M × V 2 60. m. L = V 2

Example 3 What volume of 5. 00 M sulfuric acid is required to prepare 25. 00 L of 0. 400 M sulfuric acid? M 1 = 5. 00 M V 2 = 25. 00 L M 2 = 0. 400 M M 1 × V 1 = M 2 × V 2 5. 0 M × V 1 = 0. 40 M × V 1 = 2. 0 L 25 L

Title: Dilution Problems You do not have to write the problem. You MUST show your work. You have the following stock solutions available: 2. 00 M Na. Cl, 4. 00 M KNO 3 and 0. 50 M Mg. SO 4. Calculate the volumes you must dilute to make the following solution. 1. 250. 0 m. L of 0. 300 M Na. Cl 2. 75. 0 m. L of 0. 200 M KNO 3 3. 5. 0 L of 0. 2 M Mg. SO 4

VI. Concentrations of Solutions (cont. ) C. Percent Solutions l If both solute & solvent are liquids Must be the same unit: m. L or L Percent by volume (% v/v) = volume of solute × 100% solution volume l If a solid is dissolved in a liquid Percent (mass/volume) (%(m/v)) = mass of solute (g) × 100% solution volume (m. L) Must be this unit

Example 1 What is the percent by volume of ethanol (C 2 H 6 O) or ethyl alcohol, in the final solution when 85 m. L of ethanol is diluted to a volume of 250 m. L with water? Volume of solute = 85 m. L Volume of solution = 250 m. L % (v/v) = volume of solute × 100% volume of solution % (v/v) = 85 m. L ethanol × 100% 250 m. L solution = 34% ethanol

Example 2 How many grams of glucose (C 6 H 12 O 6) would you need to prepare 2. 0 L of 2. 8% glucose (m/v) solution? Solution volume = 2. 0 L → change to m. L Percent by mass = 2. 8% Percent (mass/volume) (%(m/v) = mass of solute (g) × 100% solution volume (m. L) 2. 0 L 1000 m. L = 2, 000 m. L 1 L 2. 8% = mass of solute (g) × 100% 2, 000 m. L 100% 0. 028 = X X = 56 g of solute 2, 000 m. L

Percent Solution Problems You do not have to write the problem. You MUST show your work. 1. What is the concentration, in percent (m/v), of a solution with 75 g K 2 SO 4 in 1500 m. L of solution? 2. A bottle of hydrogen peroxide antiseptic is labeled 3. 0% (v/v). How many m. L H 2 O 2 are in a 400. 0 m. L bottle of this solution? 3. Calculate the grams of solute required to make 250 m. L of 0. 10% Mg. SO 4 (m/v).
- Slides: 39