REGULATION OF FLUID AND ELECTROLYTE BALANCE Body Fluids

REGULATION OF FLUID AND ELECTROLYTE BALANCE • Body Fluids and Fluid Compartments • Body Fluid and Electrolyte Balance – fluid and electrolyte homeostasis Why do we care about this? üECF volume üOsmolarity

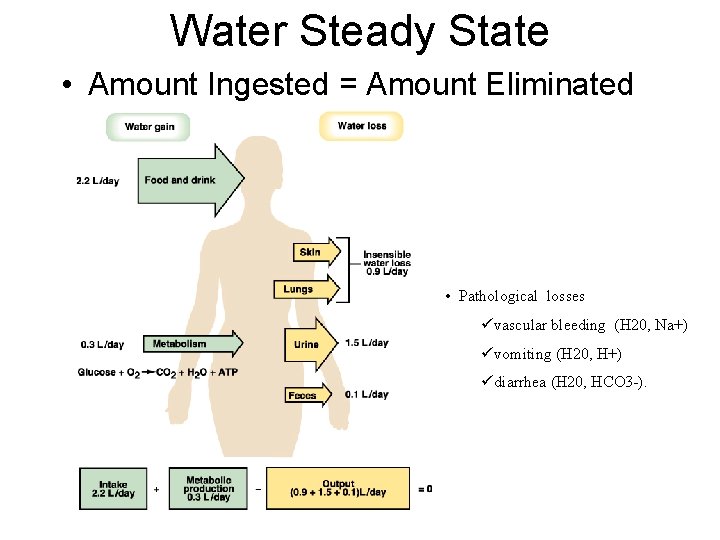
Water Steady State • Amount Ingested = Amount Eliminated • Pathological losses üvascular bleeding (H 20, Na+) üvomiting (H 20, H+) üdiarrhea (H 20, HCO 3 -).

Electrolyte (Na+, K+, Ca++) Steady State • Amount Ingested = Amount Excreted. • Normal entry: Mainly ingestion in food. • Clinical entry: Can include parenteral administration.

Electrolyte losses • • Renal excretion Stool losses Sweating Abnormal routes: e. g. . vomit and diarrhea

Body Fluids and Fluid Compartments • The percentage of total body water: 45 -75% • Intracellular compartment – 2/3 of body water (40% body weight) • Extracellular compartment – 1/3 of body water (20% body weight) – the blood plasma (water=4. 5% body weight) – interstitial fluid and lymph (water=15% body weight) – transcellular fluids: e. g. cerebrospinal fluid, aqueous humor (1. 5% BW) • Distribution of substances within the body is NOT HOMOGENEOUS.
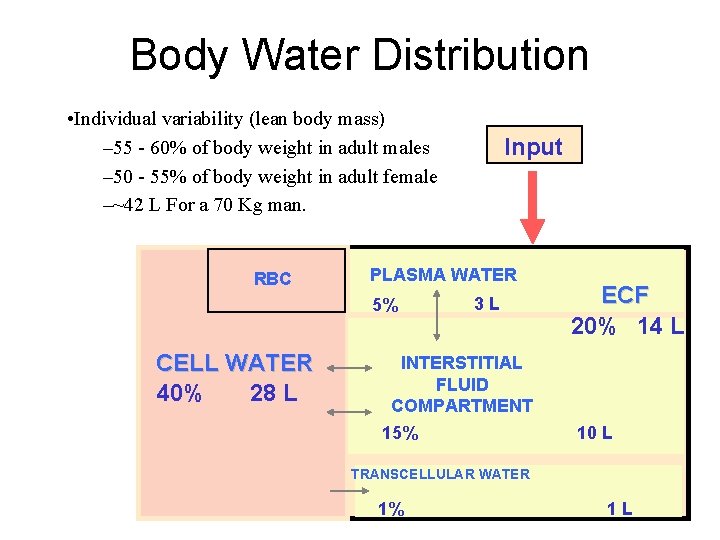
Body Water Distribution • Individual variability (lean body mass) – 55 - 60% of body weight in adult males – 50 - 55% of body weight in adult female –~42 L For a 70 Kg man. RBC PLASMA WATER 5% CELL WATER 40% 28 L Input 3 L INTERSTITIAL FLUID COMPARTMENT 15% ECF 20% 14 L 10 L TRANSCELLULAR WATER 1% 1 L

Electrochemical Equivalence • Equivalent (Eq/L) = moles x valence • Monovalent Ions (Na+, K+, Cl-): – 1 milliequivalent (m. Eq/L) = 1 millimole • Divalent Ions (Ca++, Mg++, and HPO 42 -) – 1 milliequivalent = 0. 5 millimole

Solute Overview: Intracellular vs. Extracellular • Ionic composition very different • Total ionic concentration very similar • Total osmotic concentrations virtually identical
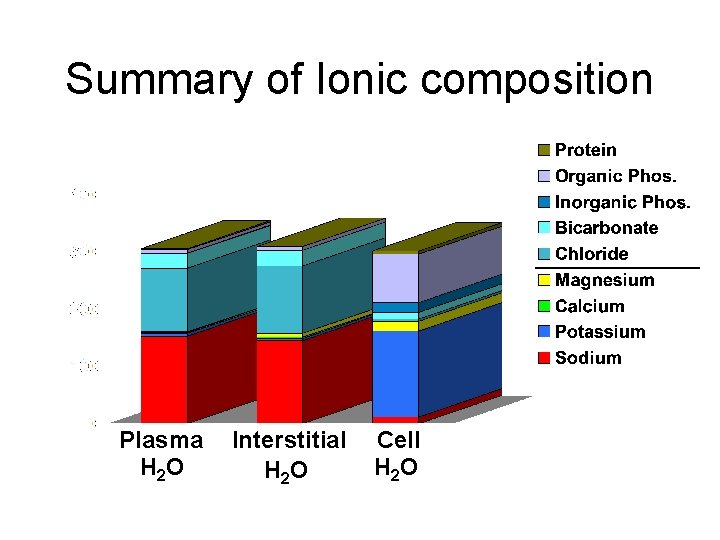
Summary of Ionic composition Plasma H 2 O Interstitial H 2 O Cell H 2 O

Net Osmotic Force Development • • Semipermeable membrane Movement some solute obstructed H 2 O (solvent) crosses freely End point: – Water moves until solute concentration on both sides of the membrane is equal – OR, an opposing force prevents further movement
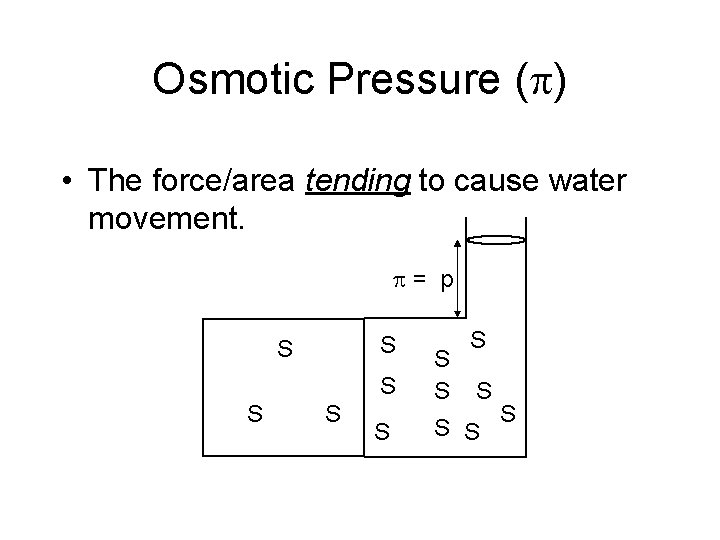
Osmotic Pressure ( ) • The force/area tending to cause water movement. = p S S S S
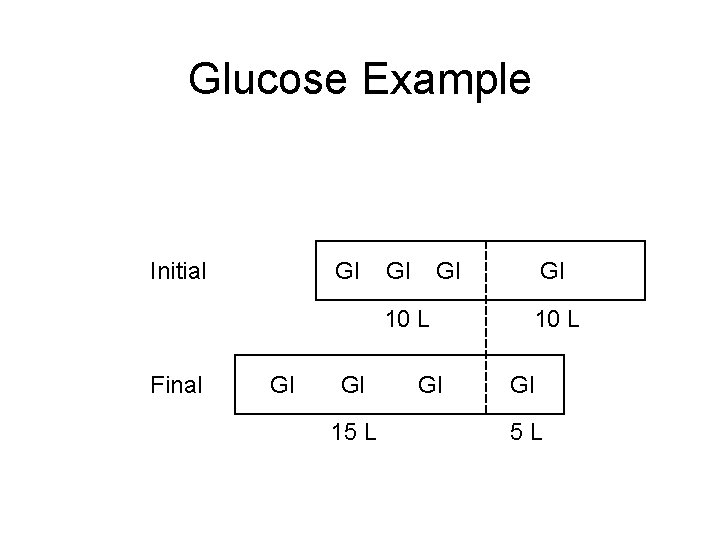
Glucose Example Initial Gl Gl 10 L Final Gl Gl 15 L Gl 10 L Gl 5 L

Osmotic Concentration • Proportional to the number of osmotic particles formed: Osm/L = moles x n (n, # of particles in solution) e. g. 1 M Na. Cl = 2 M Glu in Osm/L • Assuming complete dissociation: – 1 mole of Na. Cl forms a 2 osmolar solution in 1 L – 1 mole of Ca. Cl 2 forms a 3 osmolar solution in 1 L • Physiological concentrations: – milli. Osmolar units most appropriate – 1 m. OSM = 10 -3 osmoles/L

Principles of Body Water Distribution • Body control systems regulate ingestion and excretion: – constant total body water – constant total body osmolarity • Osmolarity is identical in all body fluid compartments (steady state conditions) – Body water will redistribute itself as necessary to accomplish this

Intra-ECF Water Redistribution Plasma vs. Interstitium • Balance of Starling Forces acting across the capillary membrane – osmotic forces – hydrostatic forces

Intracellular Fluid Volume • ICFV altered by: changes in extracellular fluid osmolarity. • ICFV NOT altered by: iso-osmotic changes in extracellular fluid volume. • ECF undergoes proportional changes in: – Interstitial water volume – Plasma water volume

Primary Disturbance: Increased ECF Osmolarity • Water moves out of cells – ICF Volume decreases (Cells shrink) – ICF Osmolarity increases • Total body osmolarity remains higher than normal

Primary Disturbance: Decreased ECF Osmolarity • Water moves into the cells – ICF Volume increases (Cells swell) – ICF Osmolarity decreases • Total body osmolarity remains lower than normal.
![Plasma Osmolarity Measures ECF Osmolarity • Plasma is clinically accessible • Dominated by [Na+] Plasma Osmolarity Measures ECF Osmolarity • Plasma is clinically accessible • Dominated by [Na+]](http://slidetodoc.com/presentation_image_h/9694f0c40f45561025d3afdb769ca42b/image-20.jpg)
Plasma Osmolarity Measures ECF Osmolarity • Plasma is clinically accessible • Dominated by [Na+] and the associated anions • Under normal conditions, ECF osmolarity can be roughly estimated as: POSM = 2 [Na+]p 270 -290 m. OSM

SOLUTIONS USED CLINICALLY FOR VOLUME REPLACEMENT THERAPY • Isotonic Solutions --> n. c. ICF • Hypertonic Solutions --> Decrease ICF • Hypotonic --> Increase ICF

Type of solutions • Saline solutions – Come in a variety of concentrations: hypotonic (eg. , 0. 2%), isotonic (0. 9%), and hypertonic (eg. 5%). • Dextrose in Saline – Glucose is rapidly metabolized to CO 2 + H 2 O – The volume therefore is distributed intracellularly as well as extracellularly – Again available in various concentrations – Used for simultaneous volume replacement and caloric supplement • Dextran, a long chain polysaccharide – Solutions are confined to the vascular compartment and preferentially expand this portion of the ECF

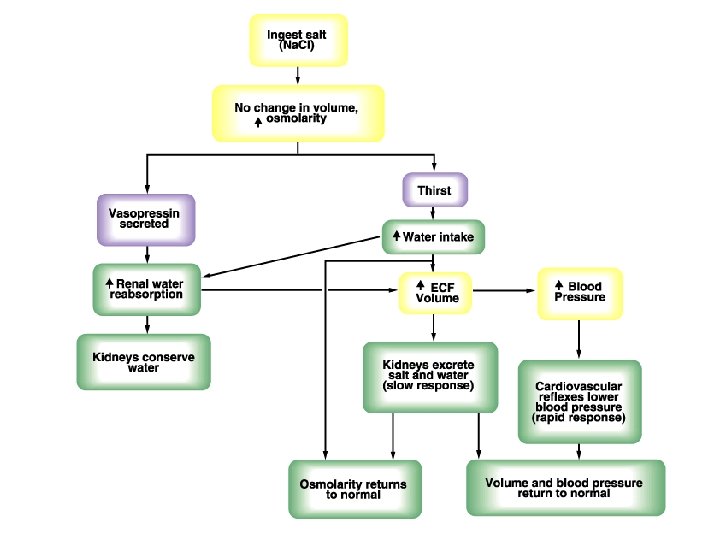
Body Fluid and Electrolyte Balance • Water input and output üThe role of the kidneys in maintaining balance of water and electrolytes üThe regulation of body water balance - thirst sensation - control of renal water excretion by ADH
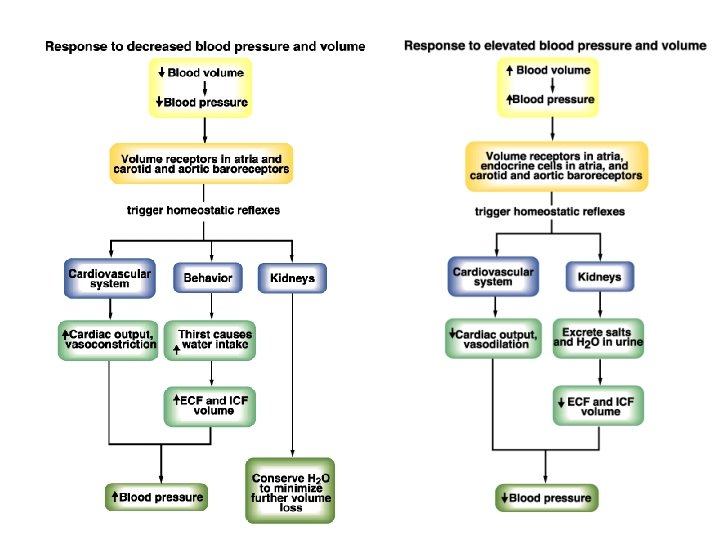
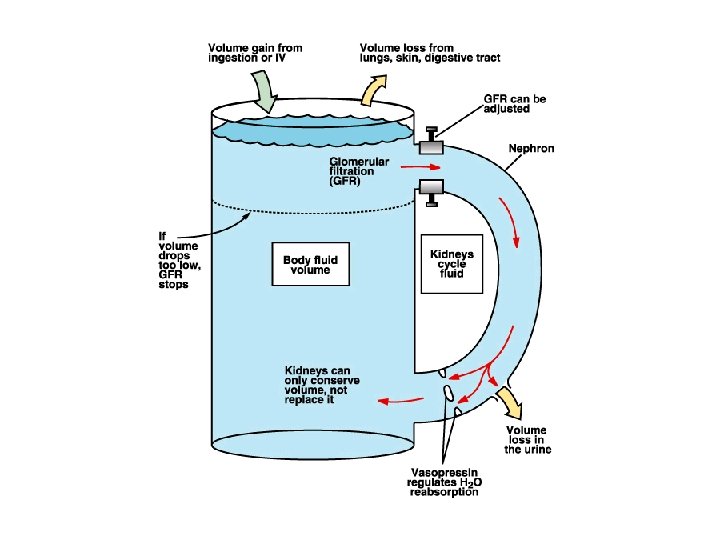

ü Thirst centers in the hypothalamus - relay information to the cerebral cortex where thirst becomes a conscious sensation - controls the release of ADH ü Stimuli for thirst sensation - Baroreceptors and stretch receptors as detectors - impulses sent to the thirst control centers in the hypothalamus ü Effect of ADH (vasopressin)
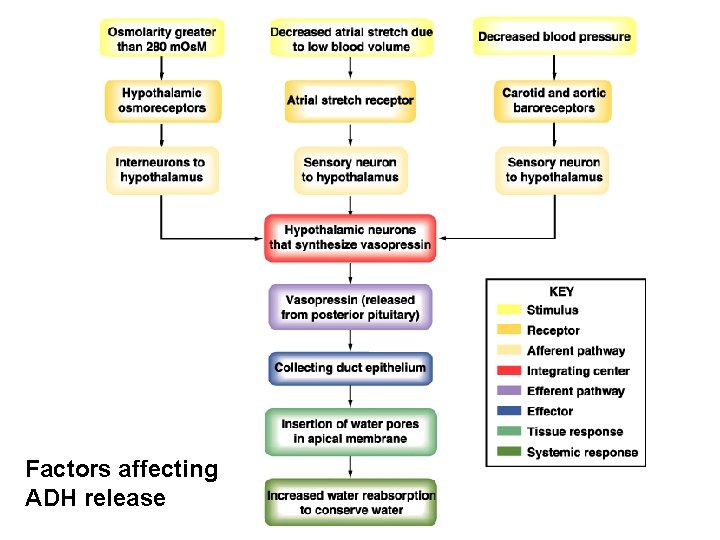
Factors affecting ADH release
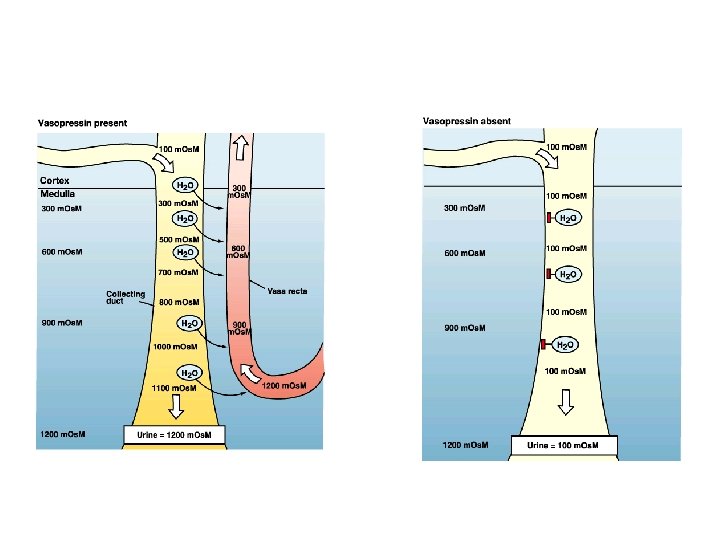
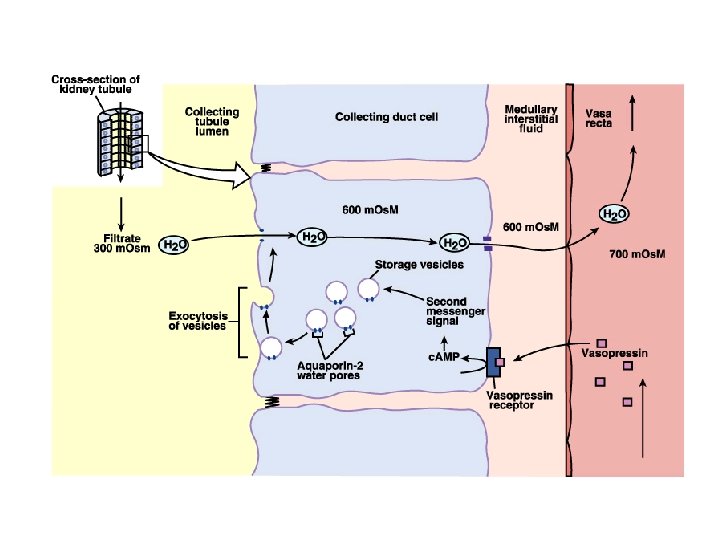

• Sodium balance ü The kidneys - the major site of control of sodium output ü Influence of dietary input on appropriate changes in sodium excretion by the kidneys ü Effector mechanisms include changes in: - glomerular filtration rate - plasma aldosterone levels - peritubular capillary Starling forces

- renal sympathetic nerve activity - intrarenal blood flow distribution - plasma atrial natriuretic factor (ANF ü Effects of aldosterone ü The renin-angiotensin system - release of renin - action of renin on the formation of angiotensin II - effects of angiotensin II: a. blood pressure; b. synthesis and release of aldosterone; c. stimulation of the hypothalamic thirst centers; d. release of ADH
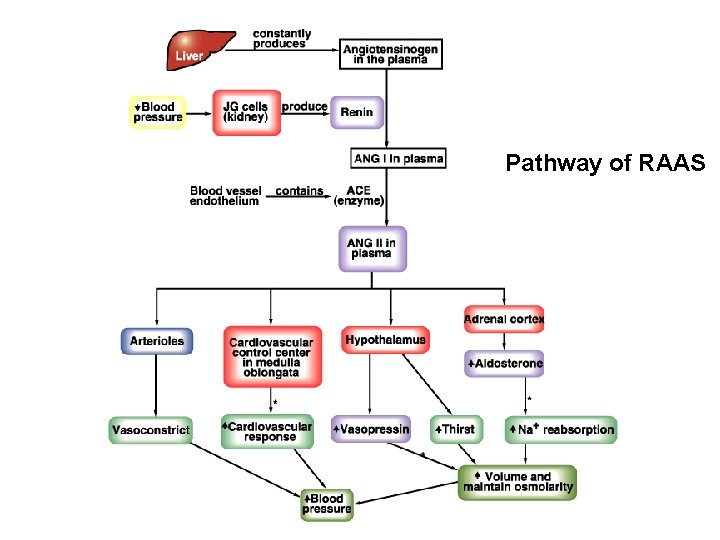
Pathway of RAAS
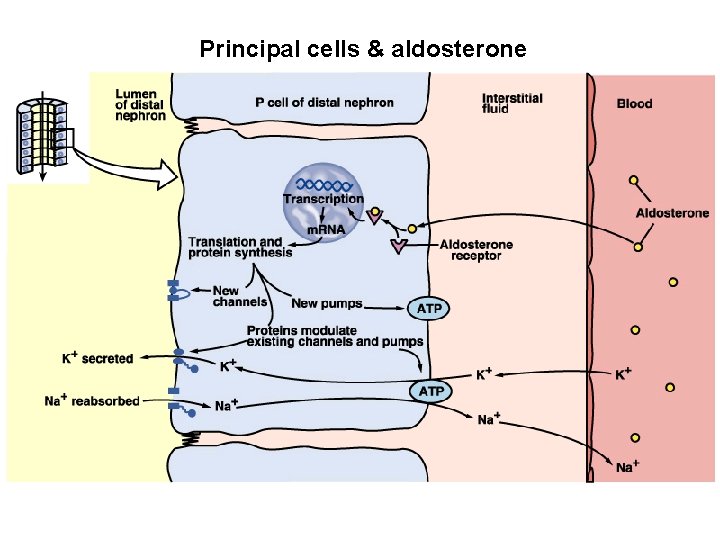
Principal cells & aldosterone

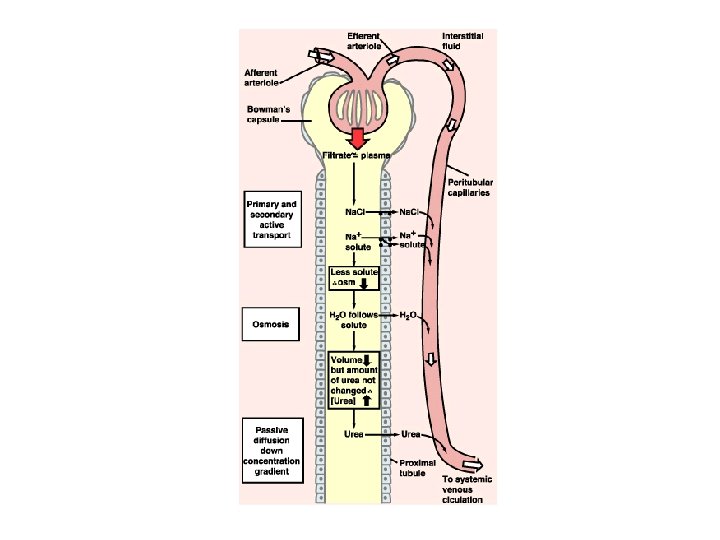
ü Net reabsorption of salt and water by the proximal convoluted tubule - peritubular capillary hydrostatic forces - colloid osmotic pressure ü Decrease in renal sodium excretion by stimulation of renal sympathetic nerves ü Release of Atrial natriuretic peptide (ANP) - in response to an increase in blood volume - increase sodium excretion by increasing GFR and inhibiting sodium reabsorption
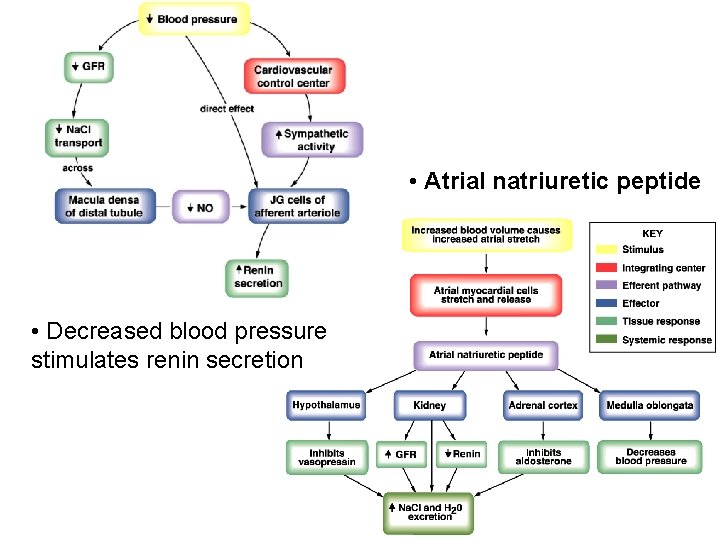
• Atrial natriuretic peptide • Decreased blood pressure stimulates renin secretion

ü The regulated variable affecting sodium excretion - effective arterial blood volume ü Changes in effective arterial blood volume can elicit the appropriate renal response by three possible mechanisms - a change in blood volume glomerular blood flow and capillary pressure GFR - a change in blood volume detected by an intrarenal baroreceptor release of renin - a change in blood volume could change peritubular capillary Starling forces

ü Other factors affecting sodium excretion include: - glucocorticoids - estrogen - osmotic diuretics - poorly reabsorbed anions - diuretic drugs
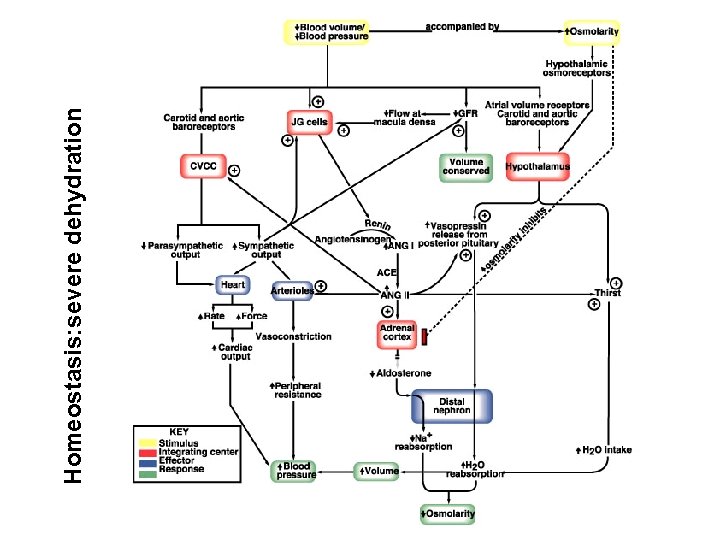
Homeostasis: severe dehydration

• Potassium balance ü Potassium plays a number of important roles in the body - electrical excitability of cells - major osmotically active solute in cells - acid-base balance - cell metabolism ü The kidneys are the major site in control of potassium balance

ü Factors affecting the distribution of potassium between cells and extracellular fluid include: - activity of the sodium-potassium pump - acid-base status of body fluids - availability of insulin - cellular breakdown due to trauma, infection, ischemia, and heavy exercise ü The regulation of plasma potassium by hormones - insulin - epinephrine - aldosterone,

ü Factors affecting potassium excretion include: - intracellular potassium concentration - aldosterone - excretion of anions - urine flow rate
- Slides: 43