CALCIUM METABOLISM Klinika nuklern medicny a endokrinologie UK

CALCIUM METABOLISM Klinika nukleární medicíny a endokrinologie UK 2. LF a FN Motol

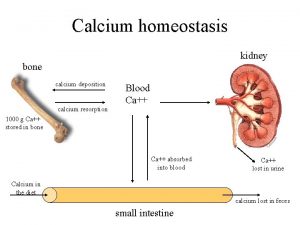
· A total of 1 to 2 kg of calcium is present in the average adult 98% of it in the skeleton. calcium of the mineral phase at the surface of the crystals is in equilibrium with that in the ECF about 0. 5% of the total pool is exchangeable. The calcium in ECF is critical for a variety of functions and is remarkably constant in range from 2. 2 to 2. 6 mmol/l.

Calcium n n n The calcium in plasma is present as three forms: free ions bound to plasma proteins diffusible complexes.

Calcium n n n The concentration of free calcium ions, averaging 1. 2 mmol/L, influences many cellular functions and is subjected to tight hormonal control, especially through PTH. The concentration of serum proteins is an important determinant of calcium ion concentration; most calcium ion is bound to albumin. Ionized calcium can be measured directly with the use of calcium-specific electrodes.

Calcium n n n Certain approximations can be used to distinguish the protein-bound and ionized fractions. Calcium metabolism controled by 3 basic systems: 1. Vitamin D 2. Parathyroid gland – Parathyroid hormone PTH 3. Renal functions Calcitonine, glucocorticoids, estrogens, thyroid hormons, GH


n n n · Calcium ions inside the cell mediate a variety of cellular functions. The level of free calcium in the cell, which is critical for functional regulation, is low, approximately 0. 1 umol/l · The gradient between plasma and intracellular free calcium is about 10, 000 to 1. This gradient is tightly regulated. · The concentration of calcium ions in the ECF is kept constant by processes that constantly add and remove calcium.

n n n · Calcium enters the plasma via absorption from the intestinal tract and resorption of ions from the bone mineral. · Calcium leaves the ECF via secretion into the gastrointestinal tract (~100 to 200 mg/d), urinary excretion (~50 to 300 mg/d), deposition in bone mineral, and losses in sweat (up to 100 mg/d). · Bone resorption and formation are tightly coupled, approximately 12 mmol (500 mg) calcium entering and leaving the skeleton daily.

n n n · The average dietary calcium intake for most adults is approximately 15 to 20 mmol/d (0. 6 to 0. 8 g/d). · Less than half of dietary calcium is absorbed in adults. · Calcium absorption increases during periods of rapid growth in children, in pregnancy, and in lactation and decreases with advancing age.

n n · Most of the calcium is absorbed in the proximal small intestine, and the efficiency of absorption decreases in the more distal intestinal segments. · Both active transport and diffusion-limited are involved; the former is more important in intestine and the latter in the lower intestine. · absorption the upper Both processes are influenced by vitamin D. · All forms of calcium in the diet may not be equally absorbed; calcium as the chloride is probably absorbed more efficiently than that in other preparations.

n n n · Calcium is also secreted into the lumen of the gastrointestinal tract. Secretion of calcium into the intestinal lumen is constant and independent of absorption. · If calcium availability in the diet is low [<12 mmol/d (500 mg/d)], a positive calcium balance requires an efficiency of absorption greater than 30 to 40 percent. · The urinary calcium excretion of normal adults having an average calcium intake ranges between 2. 5 and 10 mmol/d (100 and 400 mg/d).

n n · When the dietary calcium level is below 5 mmol/d (200 mg/d), urinary calcium excretion is usually less than 5 mmol/d (200 mg/d). · When the diet is low in calcium, the relative inefficiency of renal calcium conservation leads to a negative calcium balance unless calcium absorption is maximal.

Vitamin D n n · In the intestine, the net effect of 1, 25(OH)2 D is to stimulate calcium and phosphate transport from the lumen of the small intestine into the circulation. · The effect of 1, 25(OH)2 D on the enhancement of bone resorption is believed to be synergistic with that of PTH and 1, 25(OH)2 D may increase bone resorption activity by stimulating immature osteoclastic precursors to become mature osteoclasts and/or by interacting with osteoblasts and bone marrow stromal cells to produce cytokines that enhance the activity of mature osteoclasts.

· The role of 1, 25(OH)2 D in the renal handling of calcium and phosphorus remains uncertain. · Receptors for 1, 25(OH)2 D are present not only in intestine, bone, and kidney but too in cells not classically considered target organs for this hormone, including skin, breast, pituitary, parathyroids, pancreatic beta cells, gonads, brain, skeletal muscle, circulating monocytes, and activated B and T lymphocytes. So the role of vitamin. D in human metabolism is not only for Ca-P metabolism, but all consequences are unclear. · ·

Parathyroid hormone n n PTH is product of parathyroid glands. The function of PTH is to maintain the ECF calcium concentration. The hormone acts directly on bone and kidney and indirectly on intestine through its effects on synthesis of 1, 25(OH)2 D to increase serum calcium concentrations PTH production is closely regulated by the concentration of serum ionized calcium. This feedback system is the critical homeostatic mechanism for maintenance of ECF calcium.

This in turn: n 1. acts to increase the rate of dissolution of bone mineral, thereby increasing the flow of calcium from bone into blood n 2. reduces the renal clearance of calcium, returning more of the calcium filtered at the glomerulus into ECF n 3. increases the efficiency of calcium absorption in the intestine.

HYPOCALCEMIA n n n A decrease in the concentration of free calcium ions in plasma results in increased neuromuscular irritability and tetany. Diagnose: · calcium concentrations below 2. 1 mmol/L · hyperphosphatemia · low urine excretion calcium and phosphorus

This syndrome is characterized by n n n n peripheral and perioral paresthesias carpal spasm pedal spasm anxiety seizures bronchospasm and/or laryngospasm Chvostek's, Trousseau's, and Erb's signs lengthening of the QT interval of the ECG

n n In infants tetany may be manifested only by irritability and lethargy. The level of calcium ions that determines which features of tetany will be manifested varies among individuals. Tetany is also influenced by other components of the ECF. For example, hypomagnesemia and alkalosis lower whereas hypokalemia and acidosis raise threshold for tetany.

Treatment hypocalcemia n Acute hypocalcemia gluconicum i. v. with tetany: Calcium n n n Without tetany: If PTH absent: calcitriol or dyhydrotachysterol combined with a high oral calcium intake n n PTH ineffective: Vitamin D or calcitriol.

Hypercalcemia ↑ Increases in total serum calcium are usually accompanied by increases in free calcium levels and may be associated with: · · · · anorexia nausea vomiting constipation hypotonia depression occasionally lethargy and coma.

Hypercalcemia n n Persistent hypercalcemia, especially when accompanied by normal or elevated levels of serum phosphate, may cause ectopic deposition of a solid phase of calcium and phosphate in walls of blood vessels, connective tissue about the joints, gastric mucosa, cornea, and renal parenchyma. Hypercalcemia per se alters renal function in addition to the pathologic effects of calcium phosphate deposition.

Treatment hypercalcemia n n n Basic rules for treatment serious hypecalcemia: increase the renal excretion, decrease thi intestinal absorbtion and stop bone calcium turnover. 1. Rehydratation: iv infusion isotonic Na. Cl sol + 40 – 100 mg Furosemid ever 2 -4 hours. 2. Gluucocorticoids, which in doses of 100 - 150 mg of hydrocortisone per day (Vitamine D intoxication, NOT usefull in patient with sarcoidosis or malignancy-related hypercalcemia) 3. Calcitonin 50 -100 IU 3 -4 times per day (Miacalcic) 4. Bisphonats 5. Haemodialysis

PARATHYROID-RELATED HYPERCALCEMIA n · Primary hyperparathyroidism is a generalized disorder of calcium, phosphate, and bone metabolism due to an increased secretion of parathyroid hormone. n The elevation of circulating hormone usually leads to hypercalcemia and hypophosphatemia

There is great variation in the manifestations: recurrent nephrolithiasis n peptic ulcers n mental changes n less frequently: extensive bone resorption Etiology: n Solitary adenomas 80% of pacients n Multiple endocrine neoplasia MEN 1, MEN 2 A

Hyperparathyroidism n n n Symptoms: · Half or more of patients with hyperparathyroidism are asymptomatic. In series in which patients are followed without operation, as many as 80 percent are classified as without symptoms; the number may be smaller if mild manifestations, such as silent bone deterioration or reduced renal function, are included. Manifestations of hyperparathyroidism involve primarily the kidneys and the skeletal system. The diagnosis is made primarily on clinical and laboratory grounds. Hypercalcemia + elevated values of immunoreactive PTH

Hyperparthyroidism Treatment: 1. Medical treatment of hypercalcemia if present 2. Surgical exstirpation of adenoma

Secondary Hyperparathyroidism n n due renal insufficience increase urine excretion of calcium and then becauseoff hypocalcemia is going to parathyroid hypertrophy. It may cause paradox hypercalcemia. Therapy: calcitriol and, if medical teratment fail, surgery.

Phosphorus n is the most abundant intracellular anion and is critical for membrane structure, transport, and energy storage. · The role of phosphate ions in tissues explains the systemic nature of cellular injury consequent to phosphorus deficiency. n Hypophosphatemia is more important, has many causes. n n The finding of hypophosphatemia is not always a reliable indicator of deficiency, since a total-body deficit of phosphorus may exist in the face of hyperphosphatemia, such as, for example, in diabetic ketoacidosis.

n n Hypophosphatemia can be moderate or severe. Severe hypophosphatemia is defined as phosphorus levels in serum below 0. 3 mmol/L n n n n n The manifestations of phosphorus deficiency: · Rhabdomyolysis · Cardiomyopathy · Respiratory insufficiency · Erythrocyte dysfunction · Leukocyte dysfunction · Skeletal demineralization · Metabolic acidosis · Nervous system dysfunction: Such patients become irritable and apprehensive and hyperventilate sufficiently to cause paresthesia and numbness. Profound muscular weakness is followed by dysarthria, confusion, obtundation, convulsive seizures, coma, and death.

Osteoporosis n n is the term used for diseases that cause a reduction in the mass of bone per unit volume. It is used to define any degree of skeletal fragility sufficient to increase the risk of fracture. · Osteoporosis, the most common of the metabolic bone diseases, is an important cause of morbidity in the elderly.

n · The reduction in mass results from an imbalance in the processes that influence the acquisition and maintenance of skeletal mass and is not accompanied by a change in the ratio of the mineral phase to the organic phase or by any abnormality in bone mineral or organic matrix.

n n · Histologically, the disorder is characterized by a decrease in cortical thickness and in the number and size of the trabeculae of cancellous bone. Individual trabecular plates are abnormally perforated and may be fractured, and trabecular connectivity is reduced. The osteoid seams, however, are of normal width.


n The remodeling of bone (its formation and resorption) is a continuous process.


n n Resorption precedes formation and is probably more intense, but it does not last as long as formation. · Bone turnover is high when many units are active and low when few are active. · Unless formation compensates for resorption, bone mass decreases. · After age 40 to 50, cortical bone is lost at a rate of about 0. 3 to 0. 5 percent per year in both sexes.

n n Around the menopause in women, an accelerated loss of cortical bone is superimposed on the age-related loss. Loss of trabecular bone begins at an earlier age in both sexes but is probably greater in women. The cumulative losses of bone mass range from 20 to 30 percent in men and 40 to 50 percent in some women. · · Bone loss involves predominantly trabecular bone in the spine and distal radius in women and the spine and hip in both women and men.

Classification n n · In some instances, osteoporosis is a manifestation of another disease, such as Cushing's syndrome or osteogenesis imperfecta. This situation is called secondary osteoporosis. · In most cases of osteoporosis, however, no other disease is apparent. Than it is termed idiopathic osteoporosis, although most of the other forms are also of unknown pathogenesis.

n n n Although osteoporosis is a generalized disorder of the skeleton, the major sequelae result from fractures of the vertebrae, wrist, hip, humerus, and tibia. · The most frequent symptoms from vertebral body fractures are pain in the back and deformity of the spine. · Pain usually results from collapse of the vertebrae, especially in the dorsal and lumbar regions, is typically acute in onset, and often radiates around the flank into the abdomen.

n n n · Such episodes may occur after sudden bending, lifting, or jumping movements that may have seemed trivial; on some occasions they cannot be related to trauma · Some patients have an associated disease such as osteoarthritis of facet joints to account for chronic back pain. · Vertebral bodies usually collapse anteriorly, producing a wedge-shaped deformity and contributing to loss in height.

n n Hip fractures are the most severe complication. · They may be the consequence of trauma, most resulting from a fall from standing height. · The likelihood of fracture in a particular location is related in part to the site where the bond density is most reduced, such as the femoral neck or intertrochanteric region. · The incidence of hip fractures in both men and women increases with age.

Laboratory findings n n ·The concentrations of calcium and inorganic phosphorus in the blood are usually normal. ·Slight hyperphosphatemia occurs in women who are past the menopause. ·The alkaline phosphatase level in uncomplicated instances is normal, but it may increase after fractures · About 20 percent of postmenopausal women with osteoporosis have hypercalciuria.

n Biochemical indices of bone resorption are, on the average, increased. n · Urinary excretion of type I collagen-derived pyridinoline cross-links n n · Free and total pyridinolines and N-telopeptide crosslinks · hydroxyproline

Markers: n n Biochemical indices of bone formation are particularly useful in monitoring the effects of therapy. bone-specific alkaline phosphatase osteocalcin C-terminal procollagen peptides.

Treatment osteoporosis n n n Estrogens may have direct effects on osteoblasts and mononuclear phagocytes and can decrease the rate of bone resorption, but bone formation usually does not increase and eventually decreases. Calcium Supplements, Vitamin D metabolites are effective in maintaining bone mass and decreasing the incidence of hip fractures.

Treatment osteoporosis n n n Calcitonin decreases bone resorption, and the use of salmon calcitonin in established osteoporosis has been recommended in doses of 50 U subcutaneously every other day. Bisphonates as alendronate, pamidronate, and tiludronate are more potent inhibitors of bone resorption Fluoride ions are deposited in the skeleton, where they are incorporated into the crystal lattice of hydroxyapatite, substituting for hydroxyl ions. This process results in a mineral phase of greater crystallinity.
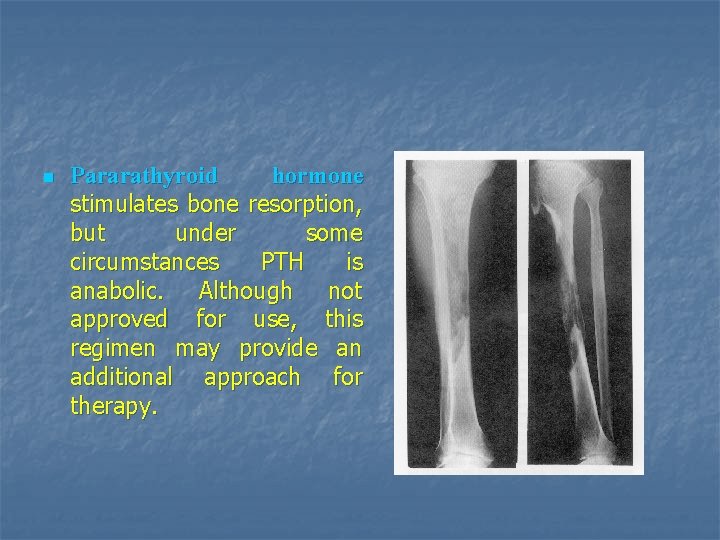
n Pararathyroid hormone stimulates bone resorption, but under some circumstances PTH is anabolic. Although not approved for use, this regimen may provide an additional approach for therapy.
- Slides: 48