MICROBIOLOGY Chapter 5 Microbial Metabolism Dr Abdelraouf A

MICROBIOLOGY Chapter 5 Microbial Metabolism Dr. Abdelraouf A. Elmanama Ph. D Microbiology Medical Technology Department, Faculty of Science, Islamic University-Gaza 2008

Microbial Metabolism • Metabolism is the sum of the chemical reactions in an organism. • Catabolism is the energy-releasing processes. • Anabolism is the energy-using processes. 2008
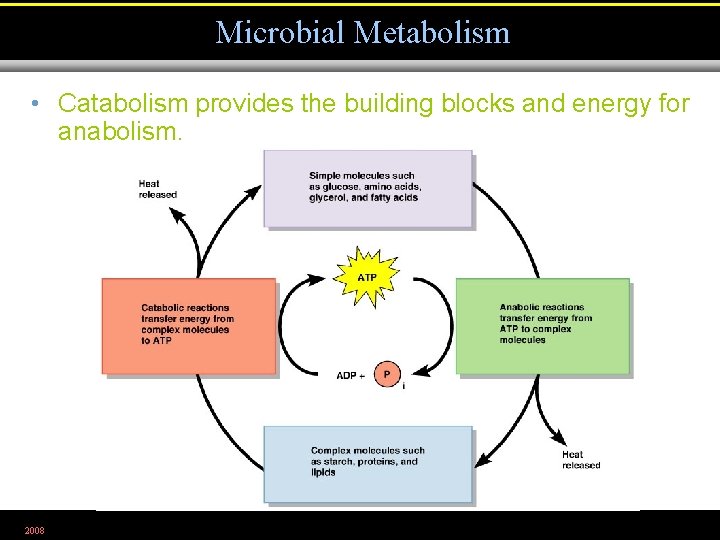
Microbial Metabolism • Catabolism provides the building blocks and energy for anabolism. 2008 Figure 5. 1

• A metabolic pathway is a sequence of enzymatically catalyzed chemical reactions in a cell. • Metabolic pathways are determined by enzymes. • Enzymes are encoded by genes. 2008

• The collision theory states that chemical reactions can occur when atoms, ions, and molecules collide. • Activation energy is needed to disrupt electronic configurations. • Reaction rate is the frequency of collisions with enough energy to bring about a reaction. • Reaction rate can be increased by enzymes or by increasing temperature or pressure. 2008
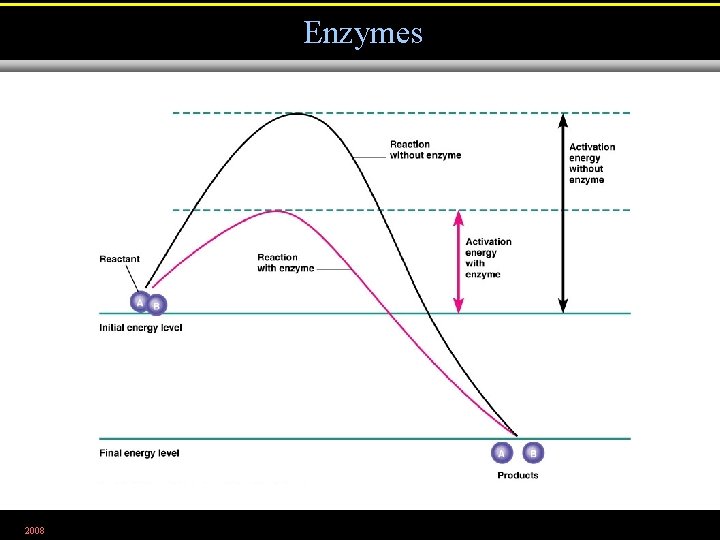
Enzymes 2008 Figure 5. 2

Enzymes • Biological catalysts • Specific for a chemical reaction; not used up in that reaction • Apoenzyme: protein • Cofactor: Non-protein component • Coenzyme: Organic cofactor • Holoenzyme: Apoenzyme + cofactor 2008
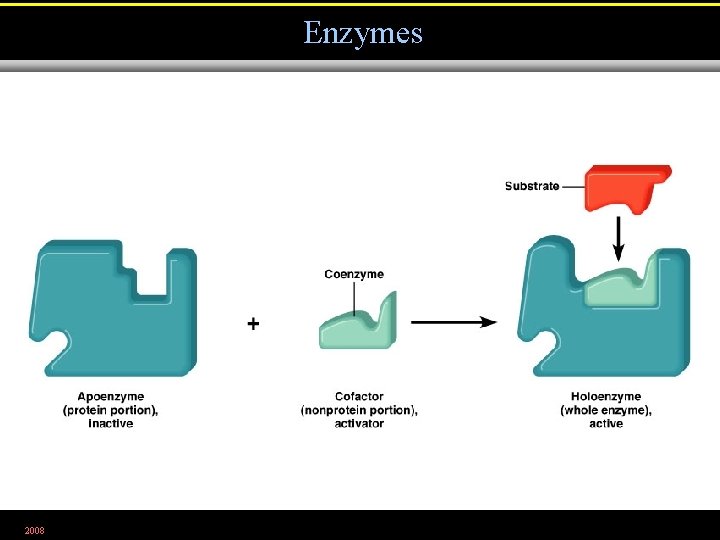
Enzymes 2008 Figure 5. 3

Important Coenzymes • NAD+ • NADP+ • FAD • Coenzyme A 2008
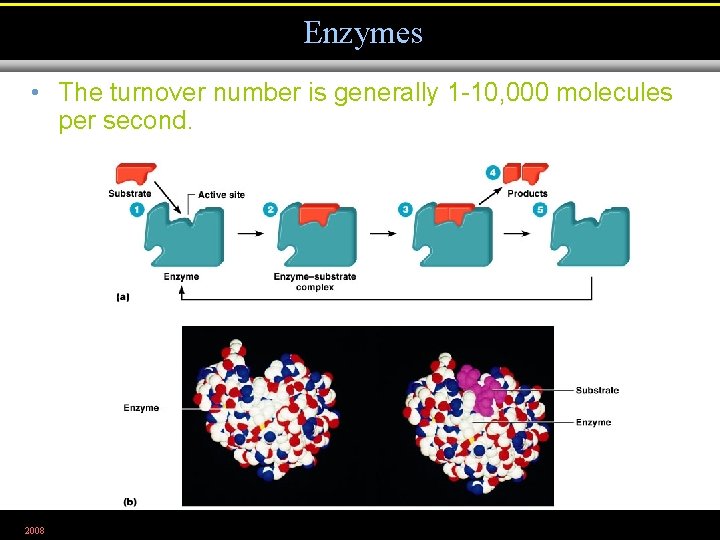
Enzymes • The turnover number is generally 1 -10, 000 molecules per second. 2008 Figure 5. 4

Enzyme Classification • Oxidoreductase Oxidation-reduction reactions • Transferase Transfer functional groups • Hydrolase Hydrolysis • Lyase Removal of atoms without hydrolysis • Isomerase Rearrangement of atoms • Ligase Joining of molecules, uses ATP 2008
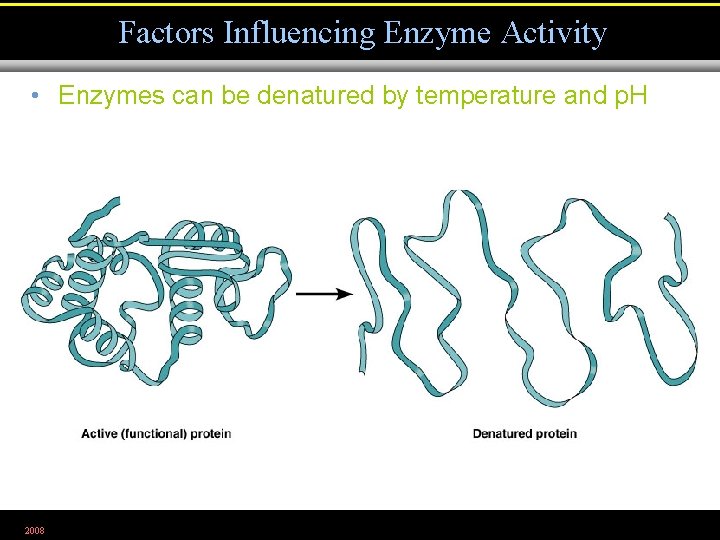
Factors Influencing Enzyme Activity • Enzymes can be denatured by temperature and p. H 2008 Figure 5. 6
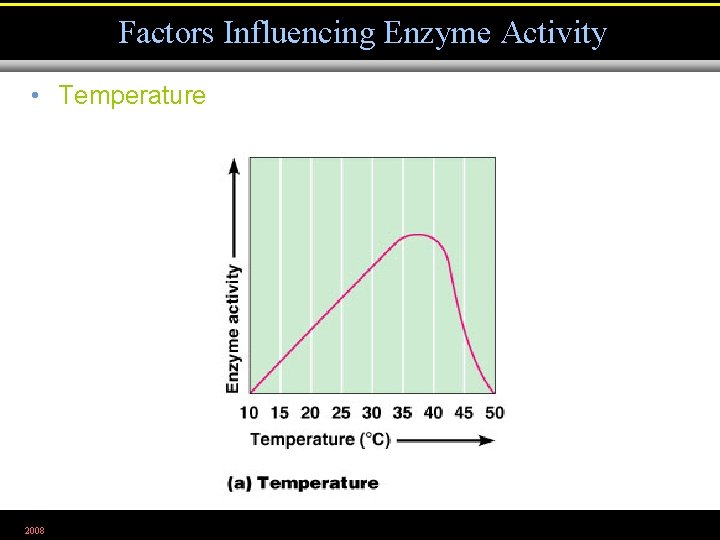
Factors Influencing Enzyme Activity • Temperature 2008 Figure 5. 5 a
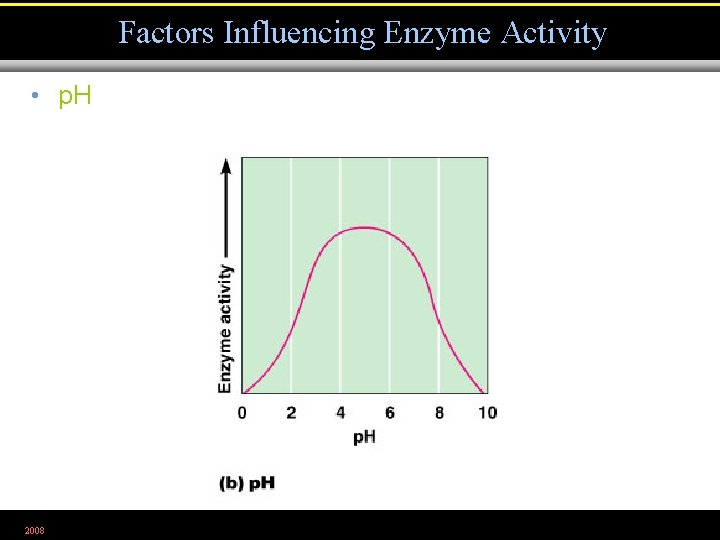
Factors Influencing Enzyme Activity • p. H 2008 Figure 5. 5 b

Factors Influencing Enzyme Activity • Substrate concentration 2008 Figure 5. 5 c

Factors Influencing Enzyme Activity • Competitive inhibition 2008 Figure 5. 7 a, b
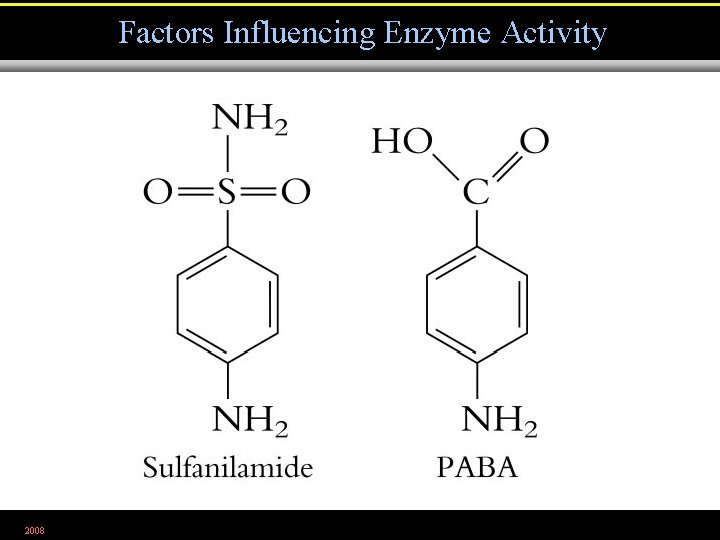
Factors Influencing Enzyme Activity 2008

Competitive inhibition 2008
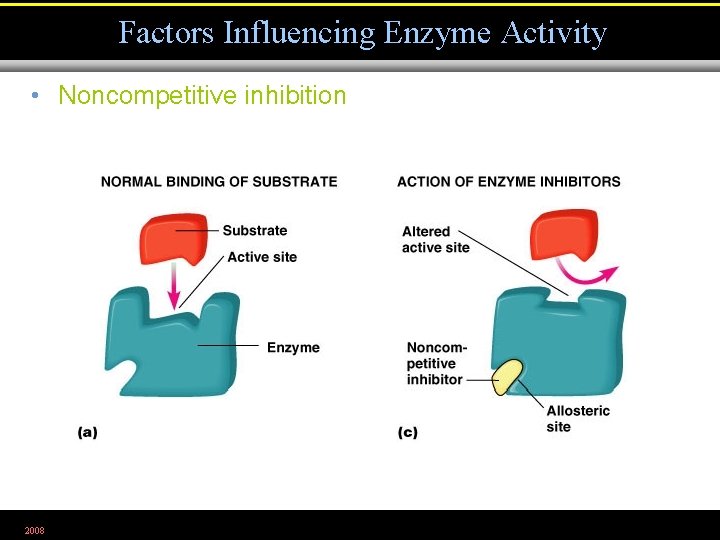
Factors Influencing Enzyme Activity • Noncompetitive inhibition 2008 Figure 5. 7 a, c

Factors Influencing Enzyme Activity • Feedback inhibition 2008 Figure 5. 8

Ribozymes • RNA that cuts and splices RNA 2008

Oxidation-Reduction • Oxidation is the removal of electrons. • Reduction is the gain of electrons. • Redox reaction is an oxidation reaction paired with a reduction reaction. 2008 Figure 5. 9
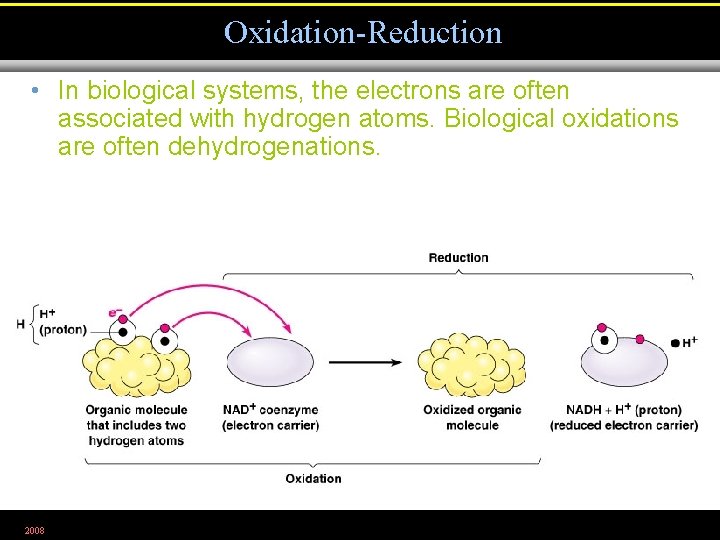
Oxidation-Reduction • In biological systems, the electrons are often associated with hydrogen atoms. Biological oxidations are often dehydrogenations. 2008 Figure 5. 10

The Generation of ATP • ATP is generated by the phosphorylation of ADP. 2008

The Generation of ATP • Substrate-level phosphorylation is the transfer of a high-energy PO 4 - to ADP. 2008

The Generation of ATP • Energy released from the transfer of electrons (oxidation) of one compound to another (reduction) is used to generate ATP by chemiosmosis. 2008

The Generation of ATP • Light causes chlorophyll to give up electrons. Energy released from the transfer of electrons (oxidation) of chlorophyll through a system of carrier molecules is used to generate ATP. 2008
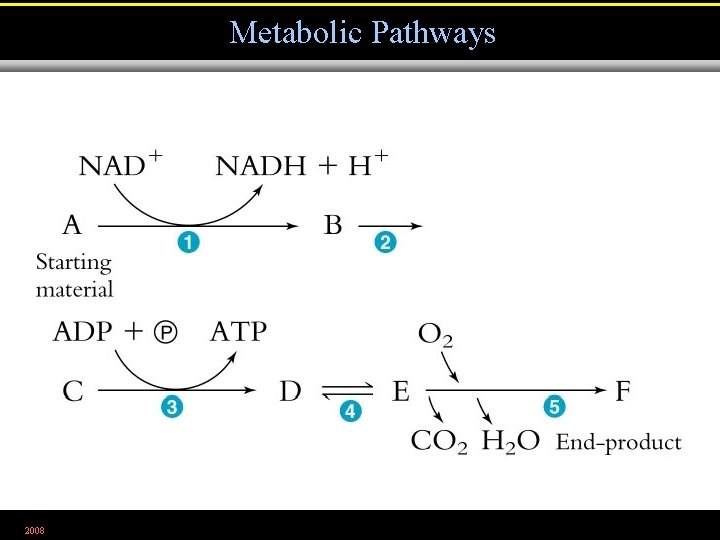
Metabolic Pathways 2008

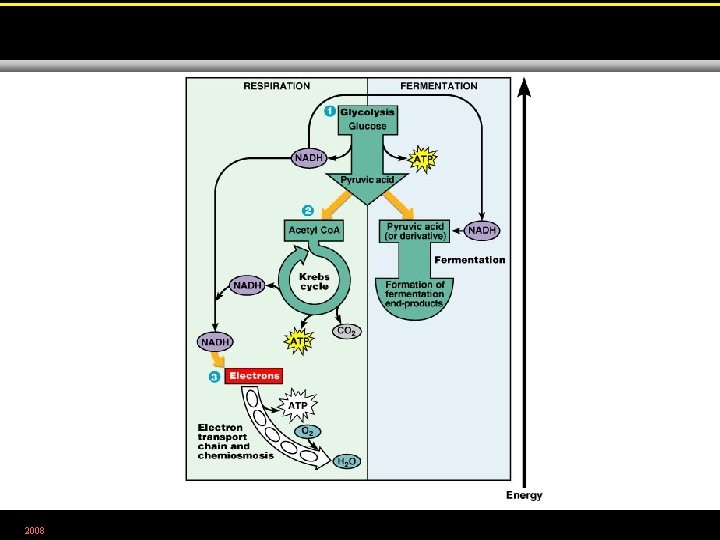
Carbohydrate Catabolism • The breakdown of carbohydrates to release energy • Glycolysis • Krebs cycle • Electron transport chain 2008

Glycolysis • The oxidation of glucose to pyruvic acid, produces ATP and NADH. 2008
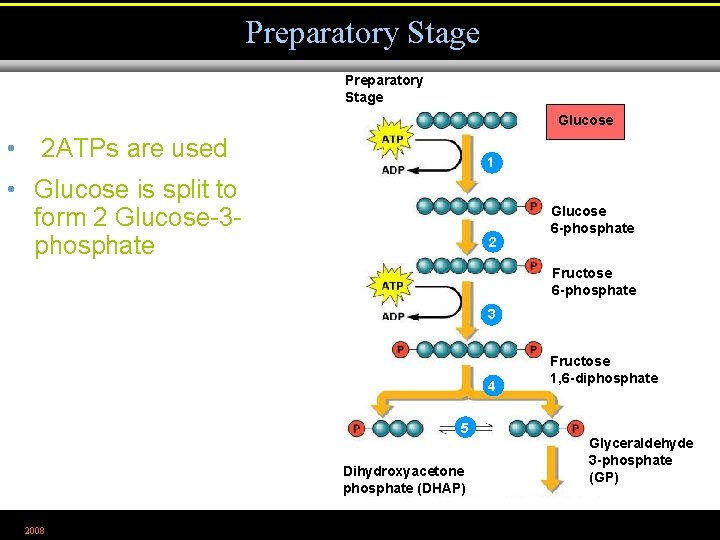
Preparatory Stage Glucose • 2 ATPs are used 1 • Glucose is split to form 2 Glucose-3 phosphate 2 Glucose 6 -phosphate Fructose 6 -phosphate 3 4 Fructose 1, 6 -diphosphate 5 Dihydroxyacetone phosphate (DHAP) 2008 Glyceraldehyde 3 -phosphate (GP)
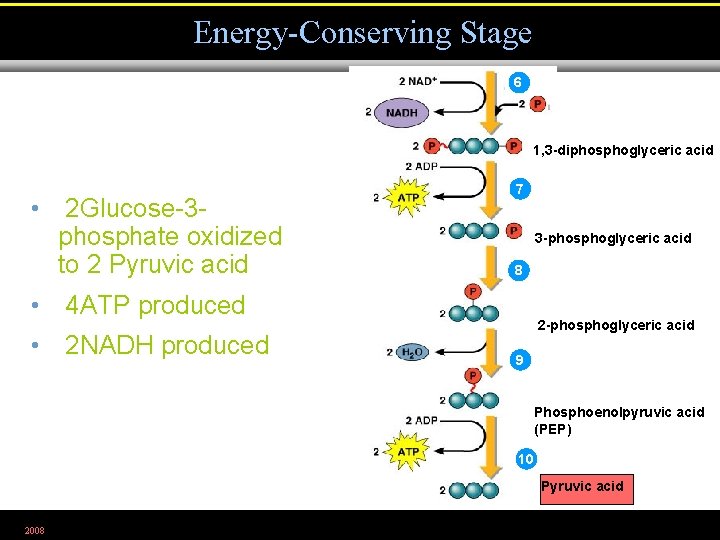
Energy-Conserving Stage 6 1, 3 -diphosphoglyceric acid • 2 Glucose-3 phosphate oxidized to 2 Pyruvic acid 7 3 -phosphoglyceric acid 8 • 4 ATP produced • 2 NADH produced 2 -phosphoglyceric acid 9 Phosphoenolpyruvic acid (PEP) 10 Pyruvic acid 2008

Glycolysis • Glucose + 2 ATP + 2 ADP + 2 PO 4– + 2 NAD+ 2 pyruvic acid + 4 ATP + 2 NADH + 2 H+ 2008

Alternatives to Glycolysis • Pentose phosphate pathway: • Uses pentoses and NADPH • Operates with glycolysis • Entner-Doudoroff pathway: • Produces NADPH and ATP • Does not involve glycolysis • Pseudomonas, Rhizobium, Agrobacterium 2008

Cellular Respiration • Oxidation of molecules liberates electrons for an electron transport chain • ATP generated by oxidative phosphorylation 2008
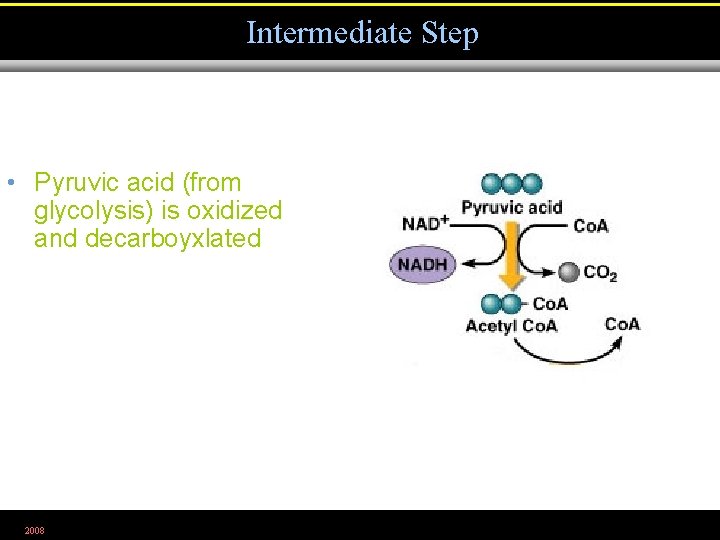
Intermediate Step • Pyruvic acid (from glycolysis) is oxidized and decarboyxlated 2008 Figure 5. 13. 1

Krebs Cycle • Oxidation of acetyl Co. A produces NADH and FADH 2 2008
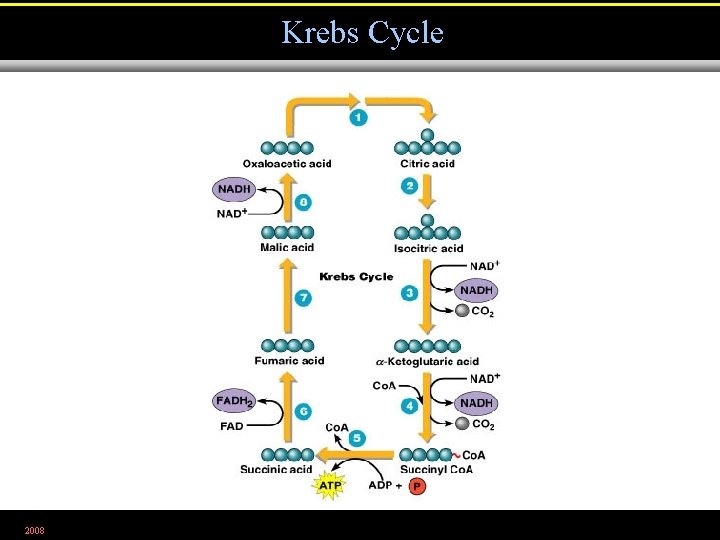
Krebs Cycle 2008 Figure 5. 13. 2

The Electron Transport Chain • A series of carrier molecules that are, in turn, oxidized and reduced as electrons are passed down the chain. • Energy released can be used to produce ATP by chemiosmosis. 2008
2008 Figure 5. 14
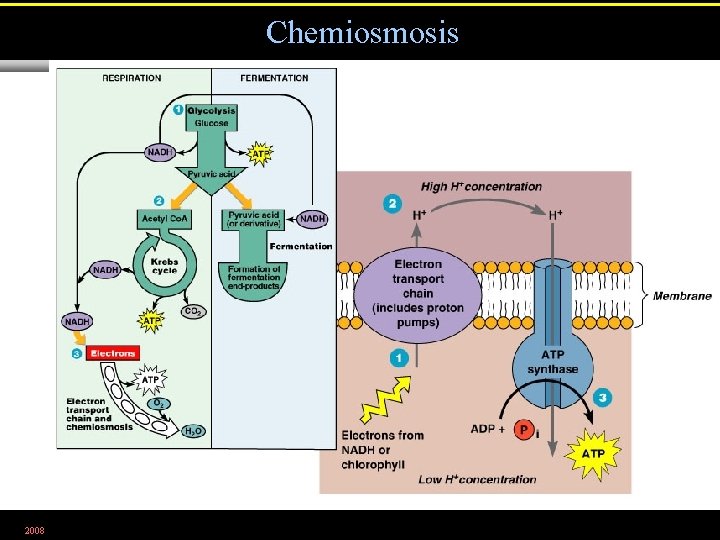
Chemiosmosis 2008 Figure 5. 15
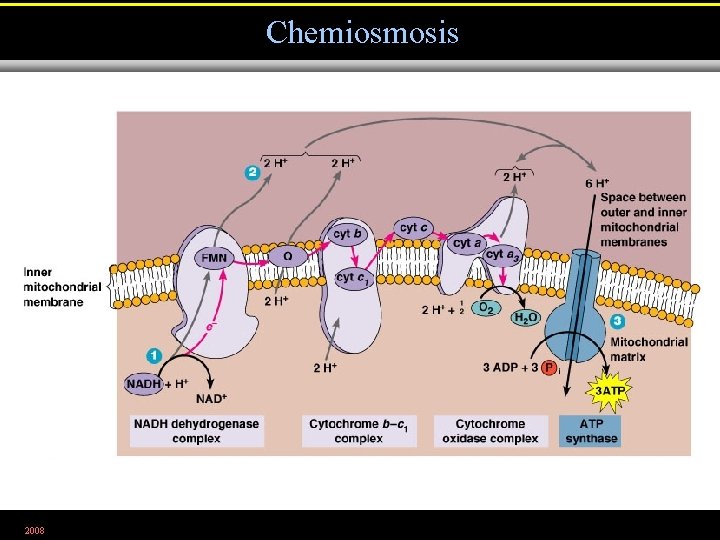
Chemiosmosis 2008 Figure 5. 16. 2

Respiration • Aerobic respiration: The final electron acceptor in the electron transport chain is molecular oxygen (O 2). • Anaerobic respiration: The final electron acceptor in the electron transport chain is not O 2. Yields less energy than aerobic respiration because only part of the Krebs cycles operations under anaerobic conditions. 2008

Anaerobic respiration Electron acceptor Products NO 3– NO 2–, N 2 + H 2 O SO 4– H 2 S + H 2 O CO 32 – CH 4 + H 2 O 2008
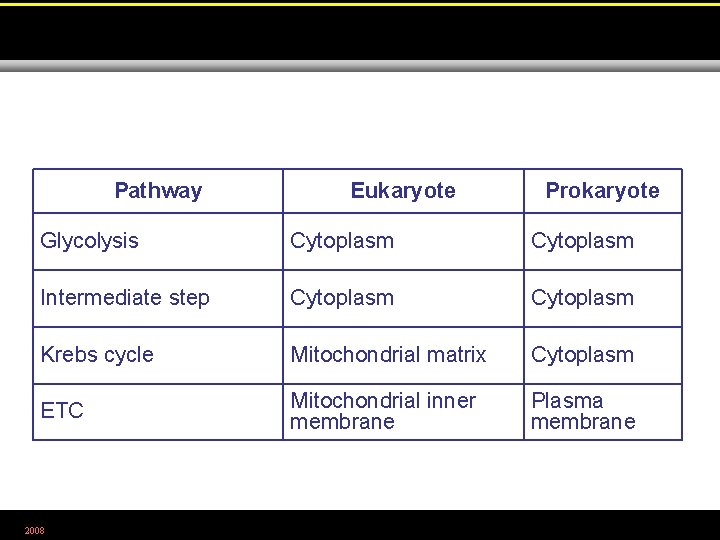
Pathway Eukaryote Prokaryote Glycolysis Cytoplasm Intermediate step Cytoplasm Krebs cycle Mitochondrial matrix Cytoplasm ETC Mitochondrial inner membrane Plasma membrane 2008

• Energy produced from complete oxidation of 1 glucose using aerobic respiration ATP produced NADH produced FADH 2 produced Glycolysis 2 2 0 Intermediate step 0 2 Krebs cycle 2 6 2 Total 4 10 2 Pathway 2008

• ATP produced from complete oxidation of 1 glucose using aerobic respiration Pathway By substrate-level phosphorylation By oxidative phosphorylation From NADH FADH Glycolysis 2 6 Intermediate step 0 6 Krebs cycle 2 18 4 Total 4 30 4 • 36 ATPs are produced in eukaryotes. 2008 0

Fermentation • Releases energy from oxidation of organic molecules • Does not require oxygen • Does not use the Krebs cycle or ETC • Uses an organic molecule as the final electron acceptor 2008
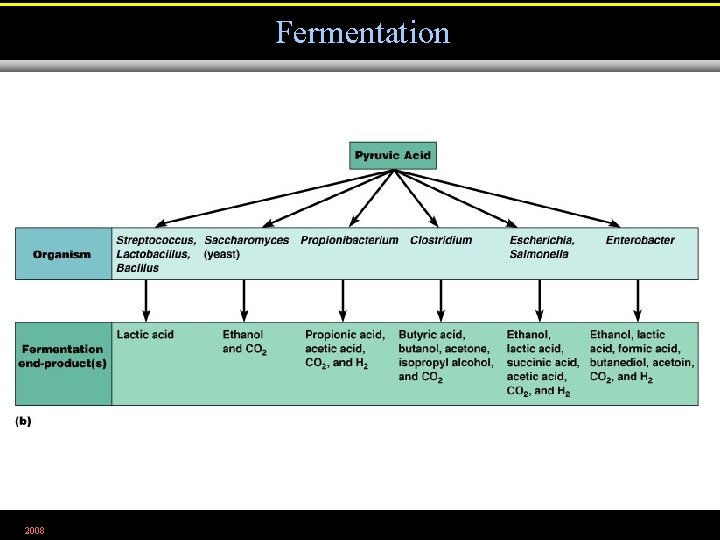
Fermentation 2008 Figure 5. 18 b

Fermentation • Alcohol fermentation. Produces ethyl alcohol + CO 2 • Lactic acid fermentation. Produces lactic acid. • Homolactic fermentation. Produces lactic acid only. • Heterolactic fermentation. Produces lactic acid and other compounds. 2008
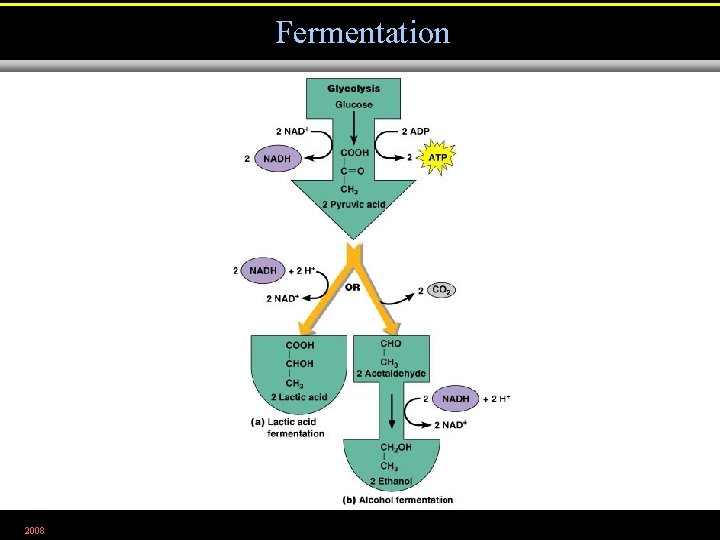
Fermentation 2008 Figure 5. 19

Fermentation 2008 Figure 5. 23
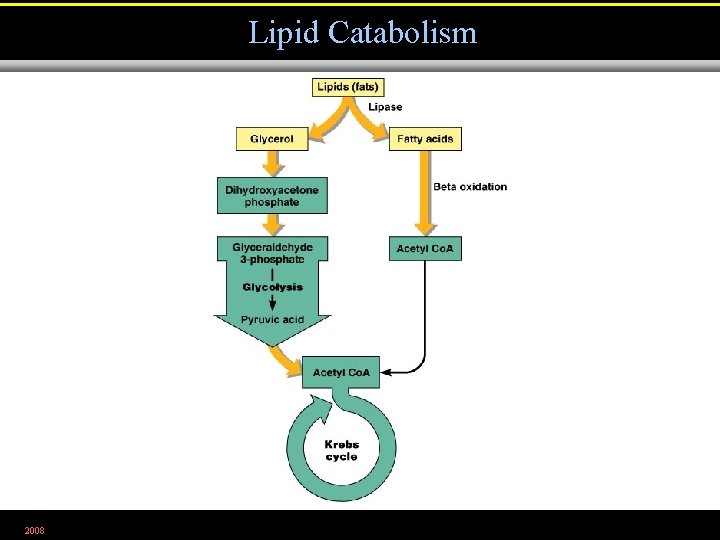
Lipid Catabolism 2008 Figure 5. 20
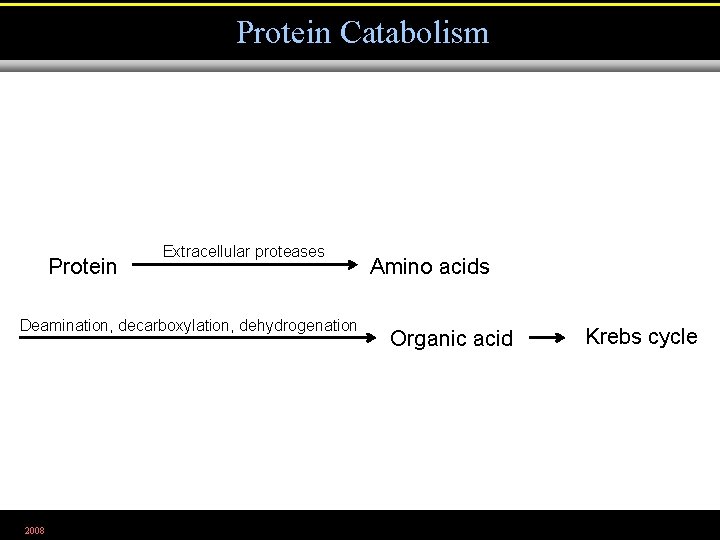
Protein Catabolism Protein Extracellular proteases Deamination, decarboxylation, dehydrogenation 2008 Amino acids Organic acid Krebs cycle

Protein Catabolism 2008 Figure 5. 22

Biochemical tests • Used to identify bacteria. 2008 Figure 10. 8

Photosynthesis • Photo: Conversion of light energy into chemical energy (ATP) • Light-dependent (light) reactions • Synthesis: Fixing carbon into organic molecules • Light-independent (dark) reaction, Calvin-Benson cycle • Oxygenic: 6 CO 2 + 12 H 2 O + Light energy C 6 H 12 O 6 + 6 O 2 + 6 H 2 O • Anoxygenic: CO 2 + 2 H 2 S + Light energy [CH 2 O] + 2 A + H 2 O 2008
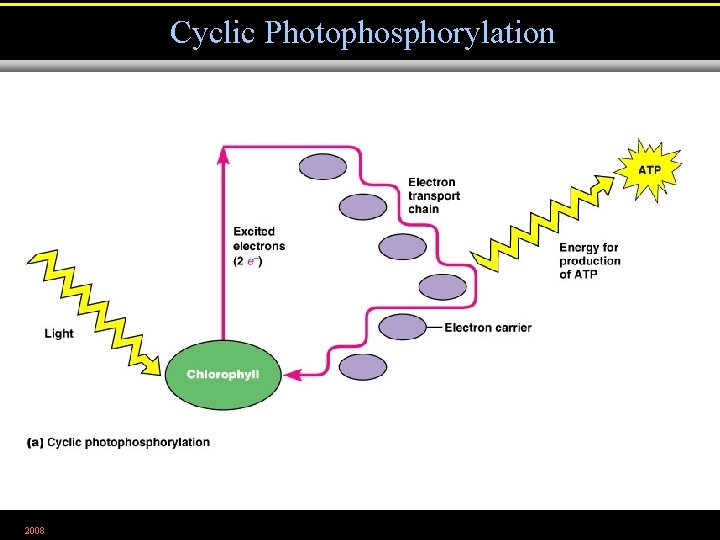
Cyclic Photophosphorylation 2008 Figure 5. 24 a

Noncyclic Photophosphorylation 2008 Figure 5. 24 b
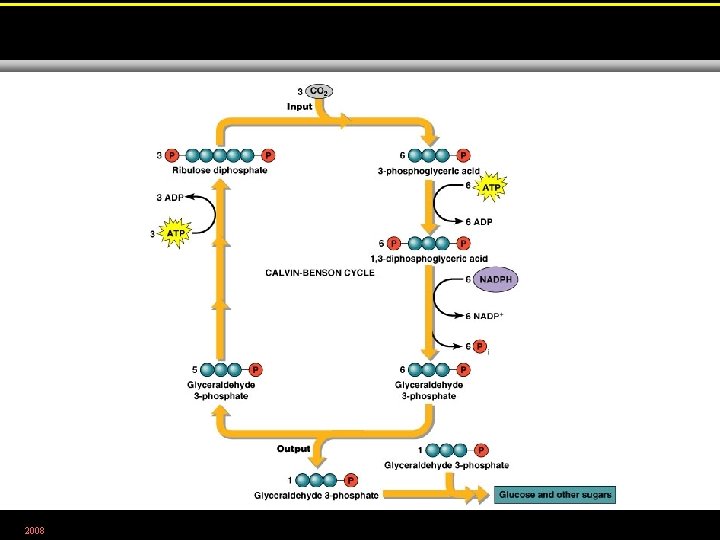
2008 Figure 5. 25
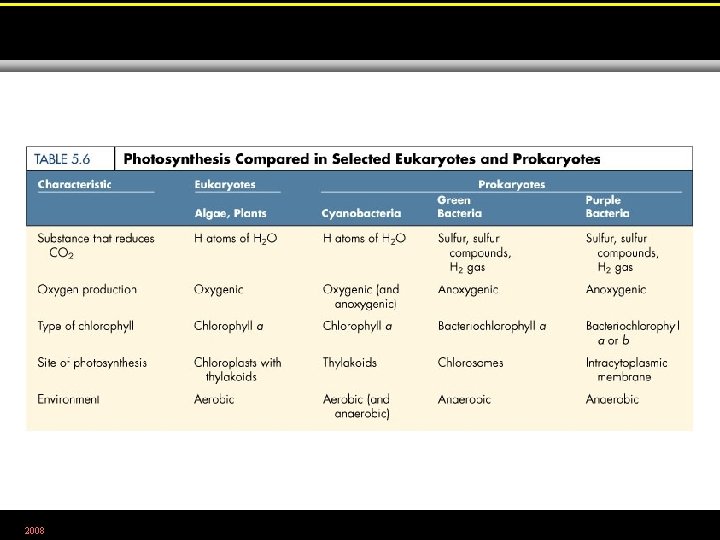
2008 Table 5. 6

• Halobacterium uses bacteriorhodopsin, not chlorophyll, to generate electrons for a chemiosmotic proton pump. 2008
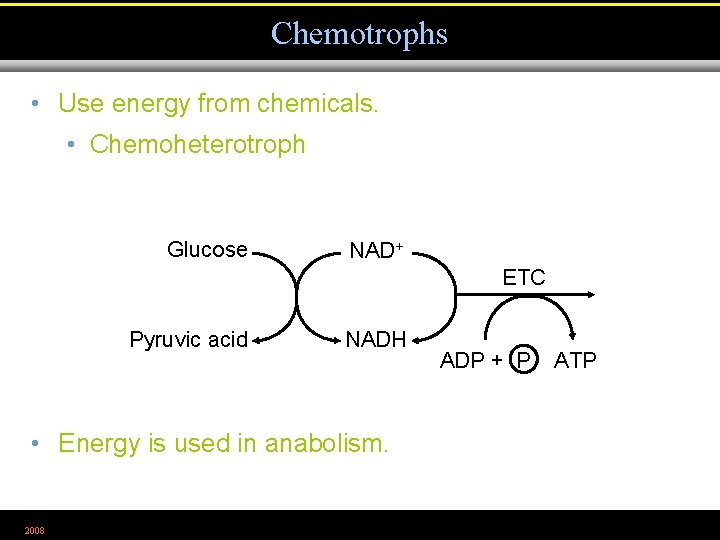
Chemotrophs • Use energy from chemicals. • Chemoheterotroph Glucose NAD+ ETC Pyruvic acid NADH • Energy is used in anabolism. 2008 ADP + P ATP
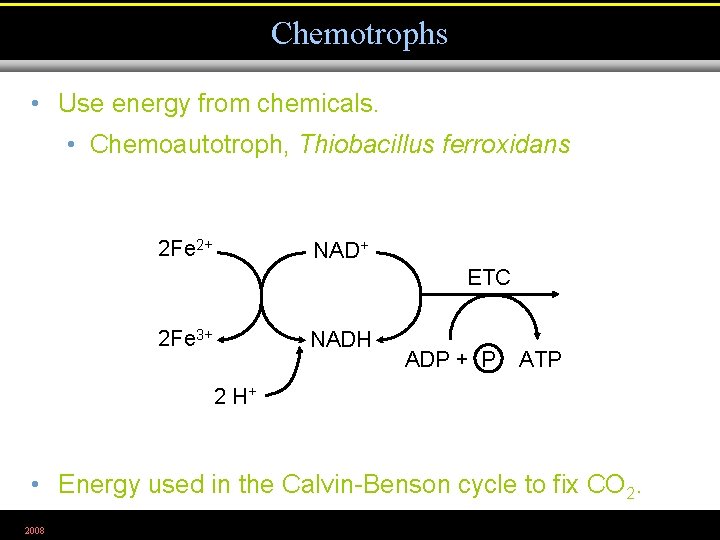
Chemotrophs • Use energy from chemicals. • Chemoautotroph, Thiobacillus ferroxidans 2 Fe 2+ NAD+ ETC 2 Fe 3+ NADH ADP + P ATP 2 H+ • Energy used in the Calvin-Benson cycle to fix CO 2. 2008

Phototrophs • Use light energy. Chlorophyll ETC Chlorophyll oxidized ADP + P ATP • Photoautotrophs use energy in the Calvin-Benson cycle to fix CO 2. • Photoheterotrophs use energy. 2008
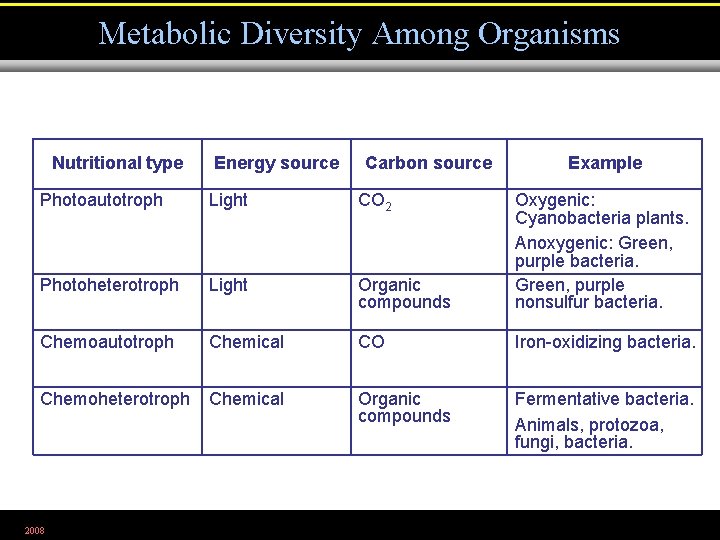
Metabolic Diversity Among Organisms Nutritional type Energy source Carbon source Example Photoautotroph Light CO 2 Photoheterotroph Light Organic compounds Oxygenic: Cyanobacteria plants. Anoxygenic: Green, purple bacteria. Green, purple nonsulfur bacteria. Chemoautotroph Chemical CO Iron-oxidizing bacteria. Chemoheterotroph Chemical Organic compounds Fermentative bacteria. Animals, protozoa, fungi, bacteria. 2008
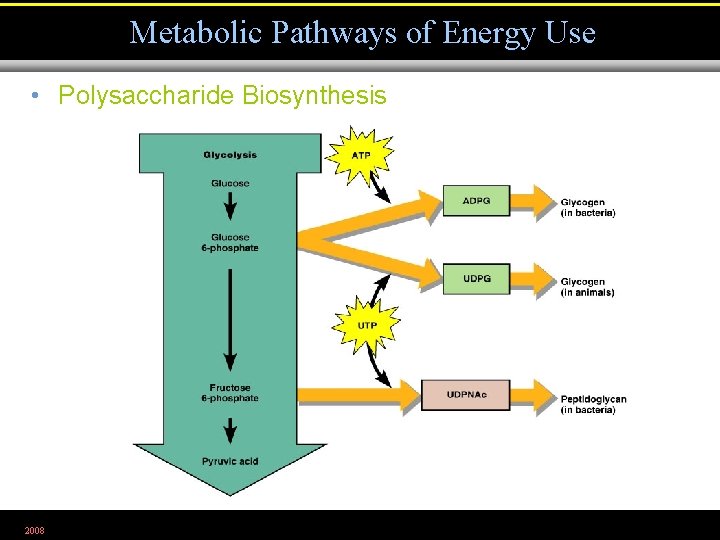
Metabolic Pathways of Energy Use • Polysaccharide Biosynthesis 2008 Figure 5. 28
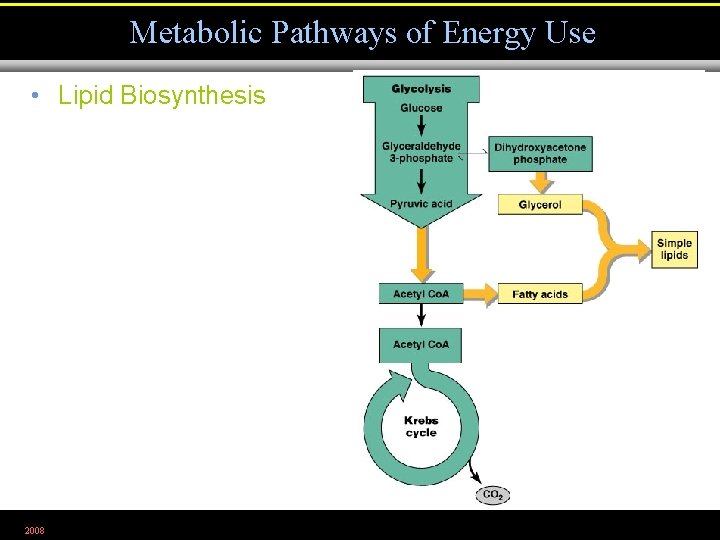
Metabolic Pathways of Energy Use • Lipid Biosynthesis 2008 Figure 5. 29

Metabolic Pathways of Energy Use • Amino Acid and Protein Biosynthesis 2008 Figure 5. 30 a
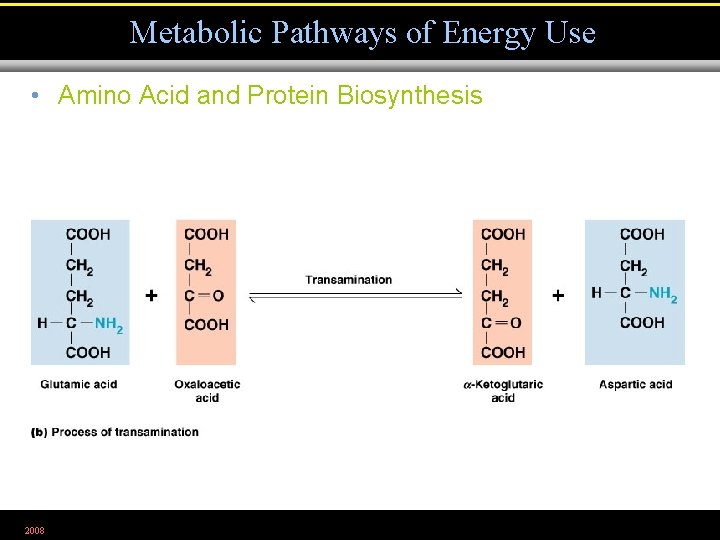
Metabolic Pathways of Energy Use • Amino Acid and Protein Biosynthesis 2008 Figure 5. 30 b
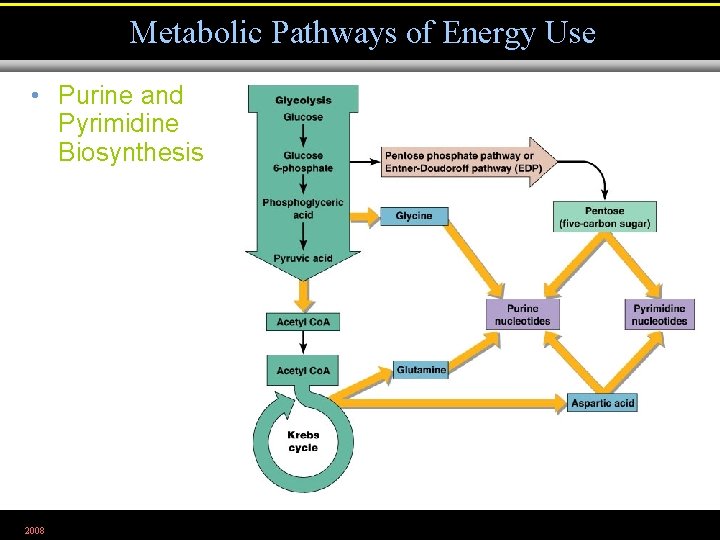
Metabolic Pathways of Energy Use • Purine and Pyrimidine Biosynthesis 2008 Figure 5. 31

Amphibolic pathways • Are metabolic pathways that have both catabolic and anabolic functions. 2008 Figure 5. 32. 1
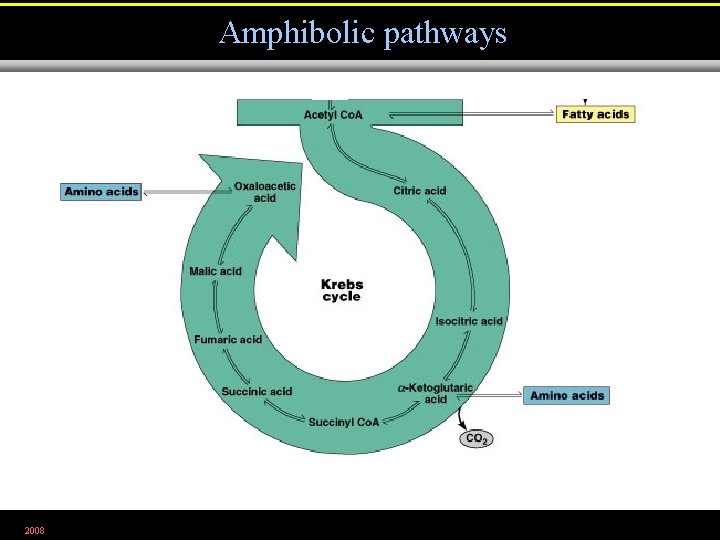
Amphibolic pathways 2008 Figure 5. 32. 2
- Slides: 73