Nucleotide Metabolism BasesNucleosidesNucleotides Base Base Adenine Base Sugar

Nucleotide Metabolism
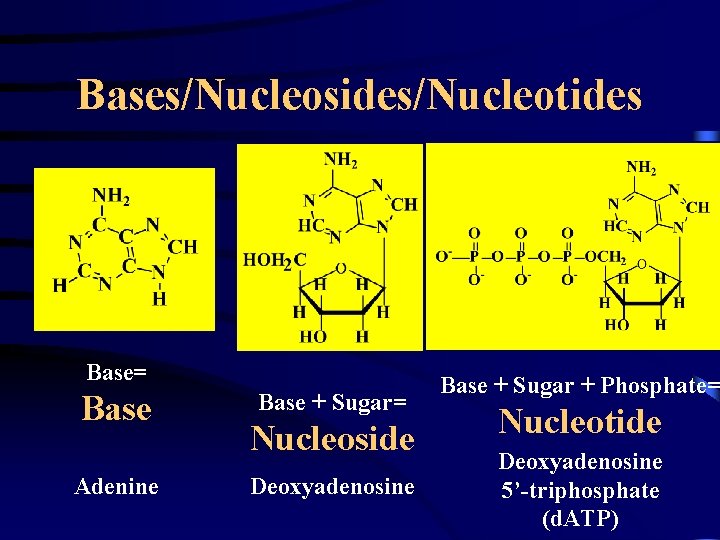
Bases/Nucleosides/Nucleotides Base= Base Adenine Base + Sugar= Nucleoside Deoxyadenosine Base + Sugar + Phosphate= Nucleotide Deoxyadenosine 5’-triphosphate (d. ATP)

Cellular Roles of Nucleotides • Energy metabolism (ATP)* • Monomeric units of nucleic acids* • Regulation of physiological processes – Adenosine controls coronary blood flow – c. AMP and c. GMP serve as signaling molecules • • Precursor function-GTP to tetrahydrobiopternin Coenzyme components- 5’-AMP in FAD/NAD+ Activated intermediates- UDP Glucose Allosteric effectors- regulate themselves and others

How I hope to make this at least bearable if not mildly interesting • Purines and Pyrimidines – Synthesis (de novo and salvage pathways) – Degradation – Relevant disease states – Relevant clinical applications (Friday) You are not responsible for any structures
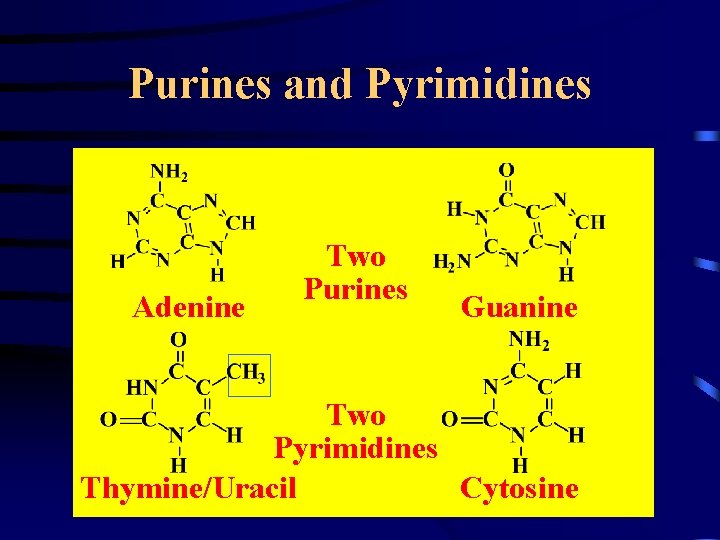
Purines and Pyrimidines Adenine Two Purines Guanine Two Pyrimidines Thymine/Uracil Cytosine

Synthesis Pathways • For both purines and pyrimidines there are two means of synthesis (often regulate one another) – de novo (from bits and parts) – salvage (recycle from pre-existing nucleotides) de novo Pathway Salvage Pathway
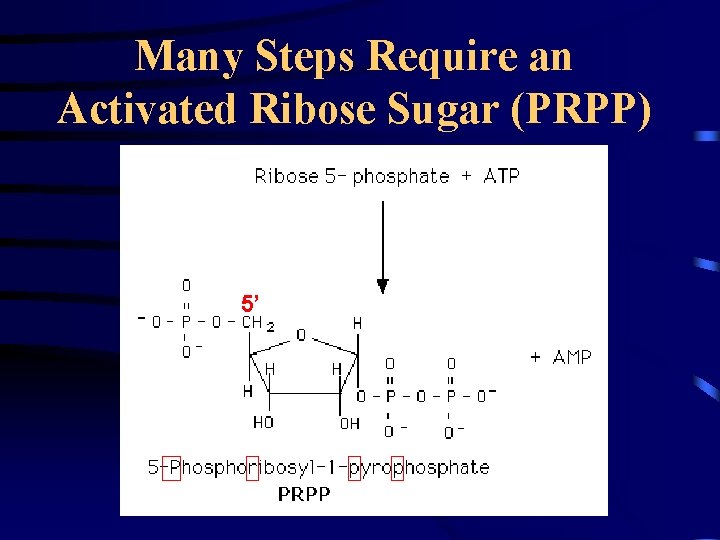
Many Steps Require an Activated Ribose Sugar (PRPP) 5’

de novo Synthesis • Committed step: This is the point of no return – Occurs early in the biosynthetic pathway – Often regulated by final product (feedback inhibition) X

Purine Biosynthesis (de novo) • Atoms derived from: – – – Aspartic acid Glycine Glutamine CO 2 Tetrahydrofolate Inhibited by AMP, GMP, IMP X • Also requires – 4 ATP’s Committed Step Purines are synthesized on the Ribose ring
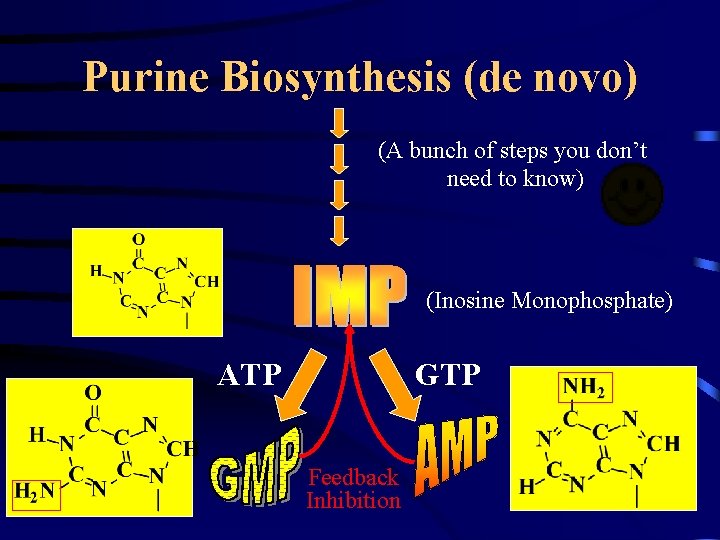
Purine Biosynthesis (de novo) (A bunch of steps you don’t need to know) (Inosine Monophosphate) ATP GTP Feedback Inhibition

Purine Degradation • Sequential removal of bits and pieces • End product is uric acid • Uric acid is primate-specific Other species further metabolize uric acid Excreted in Urine Xanthine Oxidase

Excess Uric Acid Causes Gout • Primary gout (hyperuricemia) – Inborn errors of metabolism that lead to overproduction of Uric Acid • Overactive de novo synthesis pathway – Leads to deposits of Uric Acid in the joints – Causes acute arthritic joint inflammation Xanthine Oxidase X Allopurinol Avoid: Offal foods such as liver, kidneys, tripe, sweetbreads and tongue

Immunodeficiency Diseases Associated with Purine Degradation • Defect in adenosine deaminase – Removes amine from adenosine • SCID- severe combined immunodeficiency • “Bubble Boy” Disease • Defect in both B-cells and Tcells (Disease of Lymphocytes) • Patients extremely susceptible to infection - hence the Bubble Lymphocyte

Therapies for SCID • Can be diagnosed in infants through a simple blood test (white cell count) • Bone marrow transplant for infants – Familial donor • Continued administration of adenosine deaminase (ADA-PEG) • Gene therapy- repair defective gene in Tcells or blood stem cells

Salvage Pathway for Purines Hypoxanthine + PRPP = IMP or GMP + PPi or Hypoxanthineguanosylphosphoribosyl transferase Guanine (HGPRTase) Adenine + PRPP = AMP + PPi Adeninephosphoribosyl transferase (APRTase)

Lesch-Nyhan Syndrome • Absence of HGPRTase • X-linked (Gene on X) – Occurs primarily in males • Characterized by: – – – Increased uric acid Spasticity Neurological defects Aggressive behavior Self-mutilation

Total Aside on X-linked Diseases • Why are X-linked diseases generally found only in males? • Females have two X chromosomes - would need to mutate both copies to see a recessive phenotype • Males have a single X chromosome XY XX Think about Fragile X Syndrome
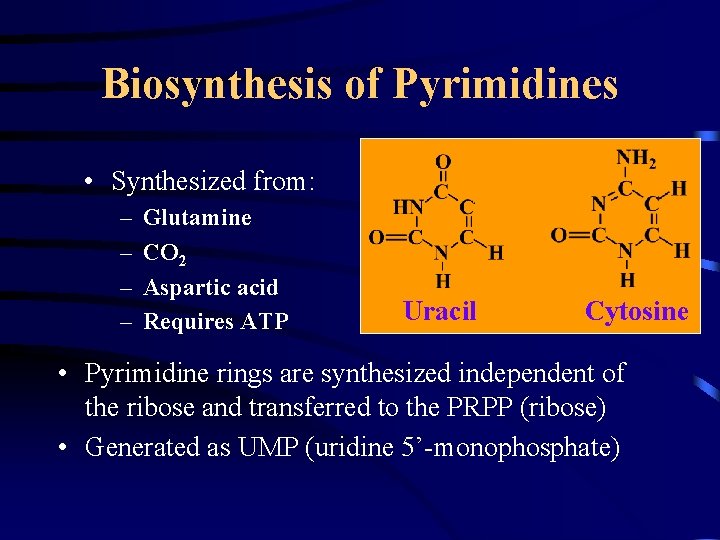
Biosynthesis of Pyrimidines • Synthesized from: – – Glutamine CO 2 Aspartic acid Requires ATP Uracil Cytosine • Pyrimidine rings are synthesized independent of the ribose and transferred to the PRPP (ribose) • Generated as UMP (uridine 5’-monophosphate)

Regulation of Pyrimidine Biosynthesis • Regulation occurs at first step in the pathway (committed step) X • 2 ATP + CO 2 + Glutamine = carbamoyl phosphate Inhibited by UTP If you have lots of UTP around this means you won’t make more that you don’t need

Hereditary Orotic Aciduria • Defect in de novo synthesis of pyrimidines • Loss of functional UMP synthetase – Gene located on chromosome III • • Characterized by excretion of orotic acid Results in severe anemia and growth retardation Extremely rare (15 cases worldwide) Treated by feeding UMP
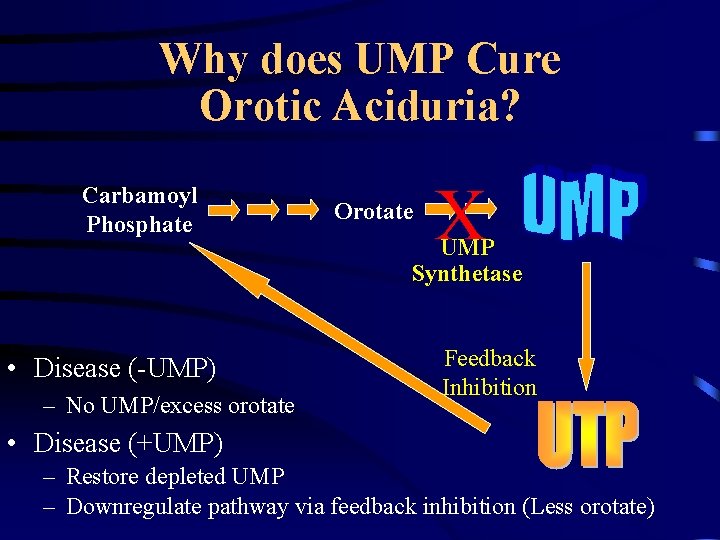
Why does UMP Cure Orotic Aciduria? Carbamoyl Phosphate • Disease (-UMP) – No UMP/excess orotate Orotate X UMP Synthetase Feedback Inhibition • Disease (+UMP) – Restore depleted UMP – Downregulate pathway via feedback inhibition (Less orotate)

Biosynthesis: Purine vs Pyrimidine • Synthesized on PRPP • Regulated by GTP/ATP • Generates IMP • Requires Energy • Synthesized then added to PRPP • Regulated by UTP • Generates UMP/CMP • Requires Energy Both are very complicated multi-step process which your kindly professor does not expect you to know in detail

Pyrimidine Degradation/Salvage • Pyrimindine rings can be fully degraded to soluble structures (Compare to purines that make uric acid) • Can also be salvaged by reactions with PRPP – Catalyzed by Pyrimidine phosphoribosyltransferase Degradation pathways are quite distinct for purines and pyrimidines, but salvage pathways are quite similar

Wait a minute: So far we’ve only made GMP, AMP, and UMP We need the d. NTPs according to the Know-it-All Professor who taught us that a couple of months ago

Two Problems • These are monophosphates (i. e. GMP)- we need triphosphates (i. e. GTP) for both DNA and RNA synthesis • These are ribonucleotides- that’s fine for RNA but we also need to make DNA Synthesis of ribonucleotides first supports the RNA world theory

Specific Kinases Convert NMP to NDP Nucleoside Monophosphates Nucleoside Diphosphates Monophosphate Kinases • Monophosphate kinases are specific for the bases Adenylate Kinase AMP + ATP 2 ADP Guanylate Kinase GMP + ATP GDP + ADP
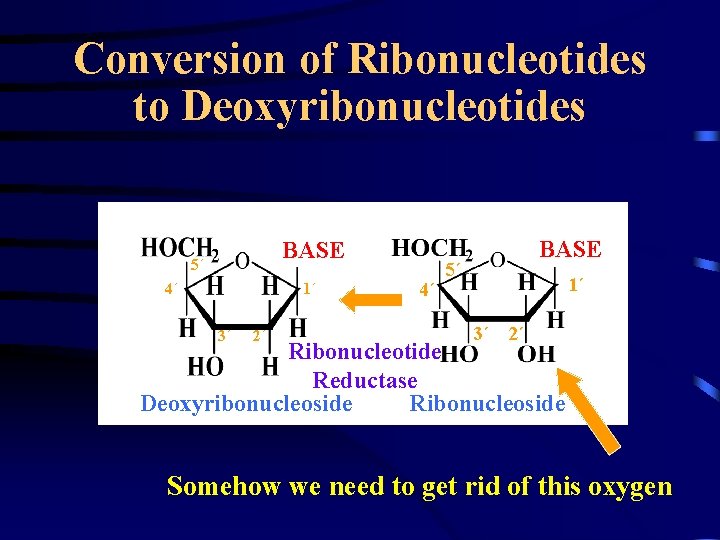
Conversion of Ribonucleotides to Deoxyribonucleotides BASE 5´ 1´ 4´ 3´ 2´ 4´ BASE 5´ 1´ 3´ 2´ Ribonucleotide Reductase Deoxyribonucleoside Ribonucleoside Somehow we need to get rid of this oxygen

Ribonucleotide Reductase • • • Catalyzes conversion of NDP to d. NDP Highly regulated enzyme Regulates the level of cellular d. NTPs Activated prior to DNA synthesis Controlled by feedback inhibition

d. NDP to d. NTP (the final step) • Once d. NDPs are generated by ribonucleotide reductase a general kinase can phosphorylate to make the d. NTP’s • So far we’ve made GTP, ATP, and UTP (which can be aminated to form CTP) • What about TTP? You’ll have to tune in tomorrow

Plan for Tomorrow • Brief Explanation of how d. UMP is converted to d. TMP • Some clinically relevant treatments based on these pathways that are used to combat: – Cancer – Bacterial Infections – Viral Infections

Take Home Concepts from Today’s Lecture • Nucleotides can be made through two pathways – (de novo and salvage) • • Pathways are regulated by feedback inhibition Specific degradation pathways exist Molecular basis of metabolic diseases mentioned What steps are necessary to generate a d. NTP from the initial NMP made
- Slides: 31